Abstract
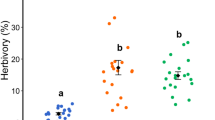
Plant development is the main factor that determines the insect-ontogeny interaction, since it leads to variations in resource quality and availability. The aim of this study was to test the hypothesis that plant development and varying tannin concentration leads to changes in species richness, abundance and composition of ants, free-feeding herbivores and galling insects associated with Copaifera langsdorffii (Fabaceae). The plant ontogeny and tannin concentration effects on insects were tested on 60 individuals with height varying from 0.9 to 11.0 m. A positive correlation was observed for tree height and species richness and abundance of ants, free-feeding and galling insects. In contrast, we did not find a significant relation between leaf tannin concentration and plant height, or richness and abundance of the different insect guilds. The assemblage of ants (composition of species) did not change between saplings and adults of C. langsdorffii. However, the assemblage of free-feeding herbivores and galling insects varied between the two development stages studied. The present study reveals an ontogenetic succession pattern for herbivore insects along the C. langsdorffii growth, probably due to both indirect and direct benefits from the host plant architecture and quality. Those plants with more complex architectures should support a wider diversity of insects, since they present higher number of sites for egg laying, housing, feeding and better environmental conditions. This is the first work to investigate the host plant ontogeny effect on insects in Cerrado “Savanna” vegetation. The pattern described, along with other previous studies, suggests a vast occurrence of ontogenetic succession in tropical areas.



Similar content being viewed by others
References
Almeida CIM, Leite GLD, Rocha SL, Machado MML, Maldonado WCH (2006) Fenologia e artrópodes de Copaifera langsdorffii Desf. no Cerrado. Rev Bras Plant Med 8:64–70
Basey JM, Jenkings SW, Busher PE (1988) Optimal central place foraging by beavers: tree size selection in relation to defensive chemicals of quaking aspen. Oecologia 76:278–282
Basset Y (1999) Diversity and abundance of insect herbivores collected on Castanopsis acuminatissima (Fabaceae) in New Guinea: relationships with leaf production and surrounding vegetation. Eur J Entomol 96:381–391
Basset Y, Aberlench HP, Delvare G (1992) Abundance and stratification of foliage arthropods in a lowland rain forest of Cameroon. Ecol Entomol 17:310–318
Basset Y, Aberlenc HP, Barrios H, Curletti G, Berenger JM, Vesco JP, Causse P, Haug A, Hennion AS, Lesobre L, Marques F, O’Meara R (2001) Stratification and diel activity of arthropods in a lowland rainforest in Gabon Biological. J Linn Soc 72:585–607
Basset Y, Novovtny V, Miller SE, Kitching RL (2003) Arthropods of tropical forests. Spatio-temporal dynamics and resource use in the canopy. Cambridge University Press, Cambridge
Blüthgen N, Verhaagh M, Goitía W, Jaffé K, Morawetz W, Barthlott W (2000) How plants shape the ant community in the Amazonian rainforest canopy: the key role of extrafloral nectaries and homopteran honeydew. Oecologia 125:229–240
Blüthgen N, Stork NG, Fiedler F (2004) Bottom-up control and co-occurrence in complex communities: honeydew and nectar determine a rainforest ant mosaic. Oikos 106:344–358
Boege K (2005) Herbivore attack in Casearia nitida influenced by plant ontogenetic variation in foliage quality and plant architecture. Oecologia 143:117–125
Boege K, Marquis RJ (2005) Facing herbivory as you grow up: the ontogeny of resistance in plants. Trends Ecol Evol 20:441–448
Boege K, Marquis RJ (2006) Plant quality and predation risk mediated by plant ontogeny: consequences for herbivores and plants. Oikos 115:559–572
Bolton B (1994) Identification guide to the ant genera of the world. Harvard University Press, Cambridge
Borror DJ, Triplehorn CA, Johnson NF (2002) An introduction to the study of insects. Saunders College Publing, Philadelphia
Bowers MD, Stamp NE (1993) Effects of plant age, genotype and herbivory on Plantago performance and chemistry. Ecology 76:1778–1791
Bryant JP, Chapin FS III, Klein DR (1983) Carbon/nutrient balance of boreal plants in relation to vertebrate herbivory. Oikos 40:357–368
Campos RI, Vasconcelos HL, Ribeiro SP, Neves FS, Soares JP (2006) Relationship between tree size and insect assemblages associated with Anadenanthera macrocarpa. Ecography 29:442–450
Carneiro MAA, Branco CSA, Braga CED, Almada ED, Costa MBM, Maia VC, Fernandes GW (2009) Are gall midge species (Diptera, Cecidomyiidae) host-plant specialists? Rev Bras Entomol 53:365–378
Carvalho PER (2003) Espécies arbóreas brasileiras. Embrapa Informação Tecnológica, Embrapa Florestas, Brasília
Clarke KR (1993) Nonparametric analyses of changes in community structure. Aust J Ecol 18:117–143
Coley PD, Bryant JP, Chapin FS III (1985) Resource availability and plant anti-herbivore defense. Science 230:895–899
Costa FV, Fagundes MF, Neves FS (2010) Arquitetura da planta e diversidade de galhas associadas à Copaifera langsdorffii (Fabaceae). Ecol Austral 20:9–17
Crawley M (2002) Statistical computing: an introduction to data analysis using S-Plus. Wiley, Imperial College, Silwood Park
Cuevas-Reyes P, Hanson P, Dardo R, Oyama K (2004) Diversity of gall-inducing insects in a Mexican tropical dry forest: the importance of plant richness, life-forms, host plant age and plant density. J Ecol 92:707–716
Davidson DW, Cook SC, Snelling RR, Chua TH (2003) Explaining the abundance of ants in lowland tropical rainforest canopies. Science 300:969–972
Dejean A, Djiéto-Lordon C, Céréghino R, Leponce M (2008) Ontogenetic succession and the ant mosaic: an empirical approach using pioneer trees. Basic Appl Ecol 9:316–323
Espírito-Santo MM, Fernandes GW (2007) How many species of gall-inducing insects are there on earth, and where are they? Ann Entomol Soc Am 100:95–99
Espírito-Santo MM, Neves FS, Andrade-Neto FR, Fernandes GW (2007) Plant architecture and meristem dynamics as the mechanisms determining the diversity of gall-inducing insects. Oecologia 153:353–364
Fagundes M, Neves FS, Fernandes GW (2005) Direct and indirect interactions involving ants, insect herbivores, parasitoids, and the host plant Baccharis dracunculifolia (Asteraceae). Ecol Entomol 30:28–35
Fernandes GW, Price PW (1991) Comparisons of tropical and temperate galling species richness: the roles of environmental harshness and plant nutrient status. In: Price PW, Lewinsohn MT, Benson WW (eds) Plant-animal interactions: evolutionary ecology in tropical and temperate regions. Wiley, New York, pp 91–115
Fernandes GW, Fagundes M, Woodman RL, Price PW (1999) Ants effects on three-trophic level interactions: plant, gall and parasitoids. Ecol Entomol 24:411–415
Ferraz FF, Monteiro RF (2003) Complex interaction envolving a gall midge Myrciamia maricaensis Maia (Diptera, Cecidomyiidae), phytophagous modifiers and parasitoids. Rev Bras Zool 3:433–437
Fonseca CR, Benson WW (2003) Ontogenetic succession in Amazonian ant trees. Oikos 102:407–412
Fonseca CR, Fleck T, Fernandes GW (2006) Processes driving ontogenetic succession of galls in a canopy tree. Biotropica 38:514–521
Fowler SV (1984) Foliage value, apparency and defence investment in birch seedlings and trees. Oecologia 62:387–392
Fritz RS, Hochwender CG, Lewkiewicz DA, Bothwell S, Orians CM (2001) Seedling herbivory by slugs in a willow hybrid system: developmental changes in damage, chemical defense, and plant performance. Oecologia 129:87–97
Fuchs A, Bowers MD (2004) Patterns of iridoid glycoside production and induction in Plantago lanceolata and the importance of plant age. J Chem Ecol 30:1723–1741
Hagerman AE (1987) Radial diffusion method for determining tannin in plant extracts. J Chem Ecol 13:437–449
Hammer Ø, Harper DAT, Ryan PD (2001) Past: paleontological statistics software package for education and data analysis. Palaeontol Electronica 4:1–9
Hartley SE (1998) The chemical composition of plant galls: are levels of nutrients and secondary compounds controlled by the gall-former? Oecologia 113:492–501
Hölldobler B, Wilson EO (1990) The ants. Harvard University Press, Cambridge
Hunter MD, Price PW (1992) Playing chutes and ladders: heterogeneity and the relative roles of bottom-up and top-down forces in natural communities. Ecology 73:724–732
Izaguirre MM, Mazza CA, Svatos A, Baldwin IT, Ballare CL (2007) Solar ultraviolet-B radiation and insect herbivory trigger partially overlapping phenolic responses in Nicotiana attenuate and Nicotiana longiflora. Ann Bot 99:103–109
Lawton JH (1983) Plant architecture and the diversity of phytophagous insect. Annu Rev Entomol 28:23–39
Mattson JMJ (1980) Herbivory in relation to plant nitrogen content. Annu Rev Ecol Syst 11:119–161
Medianero E, Barrios H (2001) Riqueza de insectos cecidógenos en el dosel y sotobosque e dos zonas ecológicas en panamá. Scientia (Panamá) 16:17–42
Medianero E, Valderrama A, Barrios H (2003) Diversidad de insectos minadores de hojas y formadores de agallas en el dosel y sotobosque del bosque tropical. Acta Zool Mex 89:153–168
Moran CV, Southwood TRE (1982) The guild composition of arthropod communities in trees. J Anim Ecol 51:289–306
Neves FS, Araújo LS, Fagundes M, Espírito-Santo MM, Fernandes GW, Sánchez-Azofeifa GA, Quesada M (2010) Canopy herbivory and insect herbivore diversity in a dry forest-savanna transition in Brazil. Biotropica 42:112–118
Oliveira PS, Brandão CRF (1991) The ant community associated with extrafloral nectaries in the Brazilian cerrados. In: Huxley CR, Cutler DF (eds) Ant/plant interactions. Oxford University Press, Oxford, pp 198–212
Parker GG (1995) Structure and microclimate of forest canopies. In: Lowman MD, Nadkarni NM (eds) Forest canopies. Academic Press, San Diego, pp 431–455
Price PW (1991) The plant vigor hypothesis and herbivore attack. Oikos 62:244–251
Price PW, Bouton CE, Gross P, McPheron BA, Thompson JN, Weis AE (1980) Interactions among three trophic levels: influence of plants on interactions between insect herbivores and natural enemies. Annu Rev Ecol Syst 11:41–65
R Development Core Team (2008) R: A language and environment for statistical computing. http://www.R-project.org. Accessed 21 Oct, 2009
Ribas CR, Schoereder JH (2004) Determining factors of arboreal ant mosaics in Cerrado vegetation (Hymenoptera: Formicidae). Sociobiology 43:49–68
Ribeiro SP, Braga OA, Silva CHL, Fernandes GW (1999) Leaf polyphenols in brazilian melastomataceae: sclerophylly, habitats and insect herbivores. Ecotropic 5:137–146
Ribeiro JF, Fonseca CEL, Sousa-Silva JC (2001) Cerrado: Caracterização e Recuperação de Matas de Galeria. Planaltina: Embrapa Cerrados, Brasília
Richards LA, Coley FD (2008) Combined effects of host plant quality and predation on a tropical lepidopteran: a comparison between treefall Gaps and the Understory in Panama. Biotropica 40:736–741
Riihimäki J, Vehvilänen H, Kaitaniemi P, Koricheva J (2006) Host tree architecture mediates the effect of predators on herbivore survival. Ecol Entomol 31:227–235
Rizzini CT (1997) Tratado de fitogeografia do Brasil: aspectos ecológicos, sociológicos e florísticos. Âmbito Cultural, Rio de Janeiro
Roslin T, Salminen JP (2008) Specialization pays off: contrasting effects of two types of tannin on oak specialist and generalist moth species. Oikos 117:1560–1568
Santos RM, Vieira FA, Fagundes M, Nunes YRF, Gusmão E (2007) Riqueza e similaridade florística de oito remanescentes florestais no norte de Minas Gerais. Rev Árvore 31:135–144
Silva JO, Jesus FM, Fagundes M, Fernandes GW (2009) Esclerofilia, taninos e insetos herbívoros associados a Copaifera langsdorffii Desf. (Fabaceae: Caesalpinioideae) em área de transição Cerrado-Caatinga no Brasil. Ecol Austral 19:197–206
Sterck FJ, Bongers F (1998) Ontogenetic changes in size, allometry, and mechanical design of tropical rain forest trees. Am J Bot 85:266–272
Stone GN, Schönrogge K (2003) The adaptive significance of insect gall morphology. Trends Ecol Evol 18:512–522
Strong DR, Lawton JH, Southwood TR (1984) Insects on plants: community patterns and mechanisms. Blackwell Scientific Publication, London
Styrsky JD, Eubanks MD (2007) Ecological consequences of interactions between ants and honeydew-producing insects. P Roy Soc Lond B Bio 274:151–164
Summerville KS, Crist TO, Kahn JK, Gering JC (2003) Community structure of arboreal caterpillars within and among four tree species of the eastern deciduous forest. Ecol Entomol 28:747–757
Varanda EM, Pais MP (2006) Insect folivory in Didymopanax vinosum (Apiaceae) in a vegetation mosaic of Brazilian Cerrado. Braz J Biol 66:671–680
Waltz AM, Whitham TG (1997) Plant development affects arthropod communities: opposing impacts of species removal. Ecology 78:2133–2144
Weis AE, Berenbaum MR (1989) Herbivorous insects and green plants. In: Abrahamson WG (ed) Plant-animal interactions. Mc Graw-Hill, New York, pp 123–162
Wilson EO (1987) The arboreal ant fauna of Peruvian Amazon forests: a first assessment. Biotropica 19:245–251
Acknowledgments
We are grateful to Queiroz ACM, Mota-Souza JG and Dantas KQ for helping in the field and laboratory work. The author Costa FV acknowledges a grant provided by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes). Neves FS, Silva JO and Fagundes M greatly acknowledge a scholarship from Fundação de Amparo a Pesquisa de Minas Gerais (FAPEMIG). This work was carried out with financial support from FAPEMIG (APQ-01231-09).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Joseph Dickens.
Rights and permissions
About this article
Cite this article
da Costa, F.V., de Siqueira Neves, F., de Oliveira Silva, J. et al. Relationship between plant development, tannin concentration and insects associated with Copaifera langsdorffii (Fabaceae). Arthropod-Plant Interactions 5, 9–18 (2011). https://doi.org/10.1007/s11829-010-9111-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-010-9111-6