Abstract
Over 90,000 legal claims for complications related to transvaginal mesh have been filed to date. Safety issues specifically related to the use of transvaginal mesh for pelvic organ prolapse have not been appropriately differentiated from the use of mesh for stress urinary incontinence by either attorneys or the media. This litigious environment and nebulous communication on the safety issues have led to patient and physician concern regarding mesh slings for the treatment of stress urinary incontinence. The ultimate result is likely to be a decrease in use of the gold standard of care for stress urinary incontinence—the mid-urethral synthetic sling.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Reynolds WS, Dmochowski RR, Penson DF. Epidemiology of stress urinary incontinence in women. Curr Urol Rep. 2011;12(5):370–6.
Wu JM et al. Predicting the number of women who will undergo incontinence and prolapse surgery, 2010 to 2050. Am J Obstet Gynecol. 2011;205(3):230 e1–5.
Cox A, Herschorn S, Lee L. Surgical management of female SUI: is there a gold standard? Nat Rev Urol. 2013;10(2):78–89.
Anger JT et al. Trends in surgical management of stress urinary incontinence among female Medicare beneficiaries. Urology. 2009;74(2):283–7.
Rogo-Gupta L et al. Trends in the surgical management of stress urinary incontinence among female Medicare beneficiaries, 2002–2007. Urology. 2013;82(1):38–41.
DeLancey JO. Structural support of the urethra as it relates to stress urinary incontinence: the hammock hypothesis. Am J Obstet Gynecol. 1994;170(6):1713–20.
Delancey JO, Ashton-Miller JA. Pathophysiology of adult urinary incontinence. Gastroenterology. 2004;126(1 Suppl 1):S23–32.
FDA. Surgical mesh for the treatment of women with pelvic organ prolapse and stress urinary incontinence: FDA executive summary. 2011; Obstetrics & Gynecology Devices Advisory Committee Meeting. Available from: http://www.fda.gov/downloads/UCM270402.pdf.
Delorme E. Transobturator urethral suspension: mini-invasive procedure in the treatment of stress urinary incontinence in women. Prog Urol. 2001;11(6):1306–13 [Abstract].
Rogo-Gupta L et al. Trends in surgical mesh use for pelvic organ prolapse from 2000 to 2010. Obstet Gynecol. 2012;120(5):1105–15.
Mucowski SJ, Jurnalov C. Use of vaginal mesh in the face of recent FDA warnings and litigation. Am J Obstet Gynecol. 2010;203(2):103 e1–4.
Feeley, J. Coloplast said to pay $16 million to settle mesh lawsuits. Bloomberg News March 4, 2014; Available from: http://www.bloomberg.com/news/2014-03-04/coloplast-said-to-pay-16-million-to-settle-mesh-lawsuits.html.
FDA. Public health notification: serious complications associated with transvaginal placement of surgical mesh in repair of pelvic organ prolapse and stress urinary incontinence. 2008 October 20, 2008; Available from: http://www.fda.gov/medicaldevices/safety/alertsandnotices/publichealthnotifications/ucm061976.htm. The FDA’s 2008 Public Health Notification was the first of two communications that initially raised public concern about the use of transvaginal mesh in POP and SUI.
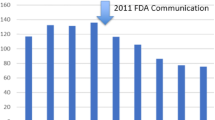
FDA. Safety communications: update on serious complications associated with transvaginal placement of surgical mesh for pelvic organ prolapse. 2011 July 13, 2011; Available from: http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm262435.htm. In 2011, the FDA reiterated and strengthened their concerns about transvaginal mesh, particularly for the treatment of POP; however, the use of mesh for SUI was not included in this heightened warning.
National Association for Continence. Surgical treatment for female stress urinary incontinence: FDA communication regarding surgery for SUI. 2012http://www.nafc.org/bladder-bowel-health/types-of-incontinence/stress-incontinence/surgical-treatment-for-female-stress-urinary-incontinence/%239.
FDA. FDA’s role and activities. 2014; Available from: http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/UroGynSurgicalMesh/ucm262301.htm.
FDA. Considerations about surgical mesh for stress urinary incontinence. 2013; Available from: http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/UroGynSurgicalMesh/ucm345219.htm. After a review of literature and the MAUDE database, the FDA determined that the use of multi-incision mesh slings for SUI was both safe and effective.
AUGS. Pelvic floor disorders registry fact sheet. 2014; Available from: http://pfdregistry.augs.org/.
Johnson and Johnson. Form 10-K Annual Report. Filed February 23, 2011; Available from: http://www.investor.jnj.com/governance/sec-filings.cfm?DocType=Annual.
Johnson and Johnson. Form 10-Q Quarterly Report. Filed August 1, 2014; Available from: http://www.investor.jnj.com/secfiling.cfm?filingID=200406-14-89.
Boston Scientific Corporation. Form 10-K Annual Report. Filed February 17, 2012; Available from: http://phx.corporate-ir.net/phoenix.zhtml?c=62272&p=irol-sec&control_selectgroup=AnnualFilings.
Boston Scientific Corporation. Form 10-Q Quarterly Report. Filed August 6, 2014; Available from: http://phx.corporate-ir.net/phoenix.zhtml?c=62272&p=irol-sec&control_selectgroup=QuarterlyFilings.
Endo Health Solutions, Inc. Form 10-K Annual Filling. Filed February 29, 2012; Available from: http://www.endo.com/investors/sec-filings.
Endo Health Solutions, Inc. Form 10-Q Quarterly Report. Filed August 4, 2014; Available from: http://phx.corporate-ir.net/phoenix.zhtml?c=123046&p=irol-SECText&TEXT=aHR0cDovL2FwaS50ZW5rd2l6YXJkLmNvbS9maWxpbmcueG1sP2lwYWdlPTk3MzI3OTUmRFNFUT0wJlNFUT0wJlNRREVTQz1TRUNUSU9OX0VOVElSRSZzdWJzaWQ9NTc%3d.
C.R. Bard, Inc. Form 10-K Annual Filing. Filed February 23, 2012; Available from: http://investorrelations.crbard.com/phoenix.zhtml?c=91501&p=irol-sec&secCat01.1_rs=11&secCat01.1_rc=10&control_searchbox=&control_selectgroup=1.
C.R. Bard, I. Form 10-Q Quarterly Report. Filed July 25, 2014; Available from: http://investorrelations.crbard.com/phoenix.zhtml?c=91501&p=irol-reportsOther.
Judicial Panel on Multidistrict Litigation. MDL Statistics Report - Number of Pending MDL Dockets by District. 2014; Available from: http://www.jpml.uscourts.gov/sites/jpml/files/Pending_MDL_Dockets_By_District-August-15-2014.pdf.
Chapple CR et al. Mesh sling in an era of uncertainty: lessons learned and the way forward. Eur Urol. 2013;64(4):525–9.
Feeley, J. Bard, Doctor ordered to pay $5.5 million over implant. Bloomberg News 2012; Available from: http://www.bloomberg.com/news/2012-07-24/bard-must-pay-5-5-million-over-vaginal-mesh-implants.html.
Scott v. Kannapan. 2012, Superior Court for Kern County, California.
Bloomberg Bureau of National Affairs. Medical Devices Law and Industry Report. 8 MELR, Issue No 18 2014; Available from: http://www.bna.com/medical-devices-law-p6787/.
Gross v. Ethicon 2013, New Jersey Superior Court, Atlantic County.
Cisson et al. v. C.R. Bard Inc. 2013, West Virginia Southern Court District.
Lewis, et al. v. Johnson & Johnson, et al. 2014, West Virginia Southern Court District.
Batiste v. McNabb. 2014, District Court for the 95th Judicial District, Dallas County Texas (Dallas).
Feeley, J. Boston Scientific wins first vaginal-mesh injury trial. Bloomberg News, 2014.
Albright v. Boston Scientific Corp. 2014, Middlesex County Superior Court, Massachusetts.
Feeley, J. Boston Scientific wins second trial over vaginal mesh. Bloomberg News, 2014.
Cardenas v. Boston Scientific Corp. 2014, Middlesex County Superior Court, Massachusetts.
Huskey v. Ethicon, Inc. 2014, West Virginia Southern Court District.
Salazar v. Lopez 2014, District Court for Dallas County, 95th Judicial District of Texas (Dallas).
McCarty, D. Boston Scientific mesh verdict halved by judge to $34 Million. Bloomberg, 2014.
Tan, G., Endo puts medical-device unit AMS on the auction block. The Wall Street Journal, 2014.
Feeley, J. Endo said to pay $400 million plus in vaginal-mesh accord. Bloomberg, 2014; Available from: http://www.bloomberg.com/news/2014-10-01/endo-said-to-pay-400-million-plus-in-vaginal-mesh-accord.html.
AUGS. Patient privacy overview fact sheet. 2014; Available from: http://www.augs.org/p/cm/ld/fid=572.
Nosti PA, Iglesia CB. Medicolegal issues surrounding devices and mesh for surgical treatment of prolapse and incontinence. Clin Obstet Gynecol. 2013;56(2):221–8.
Brown LK et al. Defining patients’ knowledge and perceptions of vaginal mesh surgery. Female Pelvic Med Reconstr Surg. 2013;19(5):282–7.
Silverstein, A. In a Dallas courtroom, it’s big pharma vs. women over vaginal mesh. 2014; Available from: http://blogs.dallasobserver.com/unfairpark/2014/09/vaginal_mesh_dallas_trial_boston_scientific.php.
Koski ME et al. Patient perception of transvaginal mesh and the media. Urology. 2014;84(3):575–82.
Koski ME, Rovner ES. Implications of the FDA statement on transvaginal placement of mesh: the aftermath. Curr Urol Rep. 2014;15(2):380.
Rice NT et al. Pelvic mesh complications in women before and after the 2011 FDA public health notification. Female Pelvic Med Reconstr Surg. 2013;19(6):333–8.
AUA. AUA position statement on the use of vaginal mesh for the surgical treatment of stress urinary incontinence. 2013; Available from: http://www.auanet.org/about/vaginal-mesh-for-sui.cfm. In 2013, the AUA released a position statement on the use of mid-urethral slings, again reaffirming the safety and efficacy of mesh MUS in the treatment of SUI. The statement concurred with the FDA that a thorough informed consent should be performed and surgeons should be rigorously trained in the use of MUS.
Wideman, N. Surgical mesh and autoimmune disease connection. 2013 April 2013; Available from: http://tvtno.org/the-facts/surgical-mesh-and-autoimmune-disease-connection/.
Walker-Journey, J. Transvaginal mesh may cause or worsen autoimmune diseases. 2013; Available from: http://www.rightinginjustice.com/news/2013/02/12/transvaginal-mesh-may-cause-or-worsen-autoimmune-diseases/.
Nager C et al. Position statement on mesh midurethral slings for stress urinary incontinence. Female Pelvic Med Reconstr Surg. 2014;20(3):123–5.
SUFU and AUGS. Frequently asked questions by providers mid-urethral slings for stress urinary incontinence. cited 2014; Available from: http://www.sufuorg.com/docs/news/Provider-FAQs-MUS-for-posting.aspx.
IUGA. Position statement on mid-urethral slings for stress urinary incontinence. 2014 [cited 2014 September 19]; Available from: http://www.iuga.org/?page=mus.
Clemons JL et al. Impact of the 2011 FDA transvaginal mesh safety update on AUGS members’ use of synthetic mesh and biologic grafts in pelvic reconstructive surgery. Female Pelvic Med Reconstr Surg. 2013;19(4):191–8.
Nabhani J, G.D., The utilization of mesh in prolapse and incontinence surgery before and after the 2008 FDA public health notification in a private insurer population, in American Urological Association. 2014: Orlando.
AUGS. AUGS transvaginal mesh informed consent toolkit. 2012; Available from: http://www.augs.org/p/cm/ld/fid=174.
Myers E et al. Estimating the early impact of the FDA safety communication on the use of surgical mesh. South Med J. 2013;106(12):684–8.
McFadden BL et al. Patient recall 6 weeks after surgical consent for midurethral sling using mesh. Int Urogynecol J. 2013;24(12):2099–104.
Compliance with Ethics Guidelines
Conflict of Interest
C. Perkins, K. Warrior, and L. McClelland declare that they have no conflict of interest.
K. Eilber and J. Anger declare that they are investigators for FDA 522 studies for Boston Scientific Corporation and American Medical Systems. J. Anger declares that she is an expert witness for Boston Scientific Corporation.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Stress Incontinence and Prolapse
Rights and permissions
About this article
Cite this article
Perkins, C.E., Warrior, K., Eilber, K.S. et al. The Role of Mid-urethral Slings in 2014: Analysis of the Impact of Litigation on Practice. Curr Bladder Dysfunct Rep 10, 39–45 (2015). https://doi.org/10.1007/s11884-014-0278-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11884-014-0278-z