Abstract
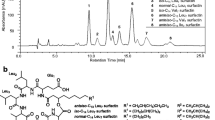
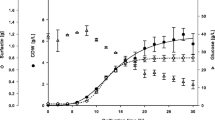
Physical and chromatographic characterization of the surfactin biosurfactant produced by Bacillus subtilis isolate BS5 has been conducted to study its potentiality for industrial application. The crude extract of test surfactin appeared as off-white to buff flake-like amorphous residue with bad odor similar to sour pomegranate. Test surfactin showed solubility in aqueous solution at pH > 5 with optimum solubility at pH 8–8.5. It was also soluble in organic solvents like ethanol, acetone, methanol, butanol, chloroform, and dichloromethane. Surfactin crystals appeared rectangular with blunt corners and were arranged perpendicular to each other making a plus sign. Extracted surfactin showed high surface activity, as it could lower the surface tension of water from about 70 to 36 mN/m at ~15.6 mg/l. Moreover, test surfactin exhibited excellent stabilities at high temperatures (100°C for up to 1 h at and autoclaving at 121°C for 10 min), salinities (up to 6% NaCl), and over a wide range of pH (5–13). Test surfactin in the cell-free supernatant or crude culture broth forms showed high emulsification indices against kerosene (62.5% and 59%, respectively), diesel (62.5% and 66%, respectively), and motor oil (62% and 66%, respectively). These characters can effectively make test surfactin, in its crude forms, a potential candidate for the use in bioremediation of hydrocarbon-contaminated sites or in the petroleum industry. Chromatographic characterization of test surfactin, using high-performance liquid chromatography technique, revealed that the extracted surfactin contained numerous isoforms, of which six were found in the standard surfactin preparation (Fluka). Additional peaks appeared in the test surfactin and not in the standard one. These peaks may correspond to new surfactin isoforms that may be present in the test surfactin produced by B. subtilis isolate BS5.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.








Similar content being viewed by others
Abbreviations
- BS:
-
biosurfactant
- CFS:
-
cell-free supernatant
- CMC:
-
critical micelle concentration
- E 24 :
-
emulsification index after 24 h
- HPLC:
-
high-performance liquid chromatography
- RP-HPLC:
-
reversed-phase HPLC
- MSM:
-
mineral slats medium
- MMSM:
-
molasses–MSM
- ODS:
-
octadecyl silane
- SF:
-
surfactin
- ST:
-
surface tension
- TPA:
-
total peak area
- tr :
-
retention time
- Wt:
-
weight
References
Makkar, R. S., & Cameotra, S. S. (1998). Production of biosurfactant at mesophilic and thermophilic conditions by a strain of Bacillus subtilis. Journal of Industrial Microbiology and Biotechnology, 20(1), 48–52.
Desai, J. D., & Banat, I. M. (1997). Microbial production of surfactants and their commercial potential. Microbiology and Molecular Biology Reviews, 61(1), 47–64.
Cooper, D. G., Macdonald, T. C. R., Duff, S. J. B., & Kosaric, N. (1981). Enhanced production of surfactin from Bacillus subtilis by continuous product removal and metal cation additions. Applied and Environmental Microbiology, 42(3), 408–412.
Dufoura, S., Deleua, M., Notta, K., Watheleta, B., Thonartb, P., & Paquot, M. (2005). Hemolytic activity of new linear surfactin analogs in relation to their physico-chemical properties. Biochimica et Biophysica Acta, 1726, 87–95.
Arima, K., Kakinuma, A., & Tamura, G. (1968). Surfactin, a crystalline peptidelipid surfactant produced by Bacillus subtilis: Isolation, characterization and its inhibition of fibrin clot formation. Biochemical and Biophysical Research Communications, 31, 488–494.
Vollenbroich, D., Ozel, M., Vater, J., Kamp, R. M., & Pauli, G. (1997a). Mechanism of inactivation of enveloped viruses by the biosurfactant surfactin from Bacillus subtilis. Biologicals, 25, 289–297.
Vollenbroich, D., Pauli, G., Ozel, M., & Vater, J. (1997b). Antimycoplasma properties and application in cell culture of surfactin, a lipopeptide antibiotic from Bacillus subtilis. Applied and Environmental Microbiology, 63(1), 44–49.
Nitschke, M., Ferraz, C., & Pastore, G. M. (2004). Selection of microorganisms for biosurfactant production using agroindustrial wastes. Brazilian Journal of Microbiology, 35, 81–85.
Abdel-Mawgoud, A. M., Aboulwafa, M. M., & Hassouna, N. A.-H. (2007). Microbial production of surfactants: Screening and identification of two promising isolates and their biosurfactants. Egyptian Journal of Biotechnology, 27, 166–185.
Abdel-Mawgoud, A. M., Aboulwafa, M. M., & Hassouna, N. A.-H. (2008). Optimization of surfactin production by Bacillus subtilis isolate BS5. Applied Biochemistry and Biotechnology. DOI 10.1007/s12010-008-8155-x.
Bodour, A. A., Drees, K. P., & Maier, R. M. (2003). Distribution of biosurfactant-producing bacteria in undisturbed and contaminated arid southwestern soils. Applied and Environmental Microbiology, 69(6), 3280–3287.
Hsieh, F.-C., Li, M.-C., Lin, T.-C., & Kao, S.-S. (2004). Rapid detection and characterization of surfactin-producing Bacillus subtilis and closely related species based on PCR. Current Microbiology, 49, 186–191.
Ozdemir, G., Peker, S., & Helvaci, S. S. (2004). Effect of pH on the surface and interfacial behavior of rhamnolipids R1 and R2. Colloids and Surfaces A, 234, 135–143.
Cooper, D. G., & Goldenberg, B. G. (1987). Surface active agents from two Bacillus species. Applied and Environmental Microbiology, 53, 224–229.
Benincasa, M., Abalos, A., Oliveira, I., & Manresa, A. (2004). Chemical structure, surface properties and biological activities of the biosurfactant produced by Pseudomonas aeruginosa LBI from soapstock. Antonie van Leeuwenhoek, 85, 1–8.
Dubey, K., & Juwarkar, A. (2001). Distillery and curd whey wastes as viable alternative sources for biosurfactant production. World Journal of Microbiology & Biotechnology, 17, 61–69.
Clifford, J. S., Ioannidis, M. A., & Legge, R. L. (2007). Enhanced aqueous solubilization of tetrachloroethylene by a rhamnolipid biosurfactant. Journal of Colloid and Interface Science, 305, 361–365.
Fox, S. L., & Bala, G. A. (2000). Production of surfactant from Bacillus subtilis ATCC 21332 using potato substrates. Bioresource Technology, 75, 235–240.
Sen, R., & Swaminathan, T. (2005). Characterization of concentration and purification parameters and operating conditions for the small-scale recovery of surfactin. Process Biochemistry, 40, 2953–2958.
Turkovskaya, O. V., Dmitrieva, T. V., & Muratova, A. Y. (2001). A Biosurfactant-producing Pseudomonas aeruginosa strain. Applied Biochemistry and Microbiology, 37(1), 71–75.
Bonilla, M., Olivaro, C., Corona, M., Vazquez, A., & Soubes, M. (2004). Production and characterization of a new bioemulsifier from Pseudomonas putida ML2. Journal of Applied Microbiology, 98, 456–463.
Helvaci, S. S., Peker, S., & Ozdemir, G. (2004). Effect of electrolytes on the surface behavior of rhamnolipids R1 and R2. Colloids and Surfaces B: Biointerfaces, 35, 225–233.
Zhang, Y., & Miller, R. M. (1992). Enhanced octadecane dispersion and biodegradation by a Pseudomonas rhamnolipid surfactant (biosurfactant). Applied and Environmental Microbiology, 58(10), 3276–3282.
Wei, Y.-H., Wang, L.-F., Chang, J.-S., & Kung, S.-S. (2003). Identification of induced acidification in iron-enriched cultures of Bacillus subtilis during biosurfactant fermentation. Journal of Bioscience and Bioengineering, 96(2), 174–178.
Kim, H.-S., Yoon, B.-D., Lee, C.-H., Suh, H.-H., Oh, H.-M., Katsuragi, T., & Tani, Y. (1997). Production and properties of a lipopeptide biosurfactant from Bacillus subtilis C9. Journal of Fermentation and Bioengineering, 84(1), 41–46.
Wei, Y.-H., & Chu, I.-M. (1998). Enhancement of surfactin production in iron-enriched media by Bacillus subtilis ATCC 21332. Enzyme and Microbial Technology, 22, 724–728.
Haba, E., Pinazo, A., Jauregui, O., Espuny, M. J., Infante, M. R., & Manresa, A. (2003). Physicochemical characterization and antimicrobial properties of rhamnolipids produced by Pseudomonas aeruginosa 47T2 NCBIM 40044. Biotechnology and Bioengineering, 81(3), 316–322.
Parkinson, M. (1985). Bio-surfactants. Biotechnology Advances, 3(1), 65–83.
Mulligan, C. N. (2005). Environmental applications for biosurfactants. Environmental Pollution, 133, 183–198.
Bognolo, G. (1999). Biosurfactants as emulsifying agents for hydrocarbons. Colloids and Surfaces A, 152, 41–52.
Carrillo, C., Teruel, J. A., Aranda, F. J., & Ortiz, A. (2003). Molecular mechanism of membrane permeabilization by the peptide antibiotic surfactin. Biochimica et Biophysica Acta, 1611, 91–97.
Kikuchi, T., & Hasumi, K. (2002). Enhancement of plasminogen activation by surfactin C: Augmentation of fibrinolysis in vitro and in vivo. Biochimica et Biophysica Acta, 1596, 234–245.
Maier, R. M., & Soberon-Chavez, G. (2000). Pseudomonas aeruginosa rhamnolipids: Biosynthesis and potential applications. Applied Microbiology and Biotechnology, 54, 625–633.
Takeyama, H., Wada, M., & Matsunaga, T. (2002). Screening of soil bacteria for production of biocleaner. Applied Biochemistry and Biotechnology, 98-100, 319–326.
Lin, S.-C., & Jiang, H.-J. (1997). Recovery and purification of the lipopeptide biosurfactant of Bacillus subtilis by ultrafiltration. Biotechnology Techniques, 11(6), 413–416.
Vater, J., Kablitz, B., Wilde, C., Franke, P., Mehta, N., & Cameotra, S. S. (2002). Matrix-assisted laser desorption ionization–time of flight mass spectrometry of lipopeptide biosurfactants in whole cells and culture filtrates of Bacillus subtilis C-1 isolated from petroleum sludge. Applied and Environmental Microbiology, 68(12), 6210–6219.
Kluge, B., Vater, J., Sainikow, J., & Eckart, K. (1988). Studies on the biosynthesis of surfactin, a lipopetide antibiotic from Bacillus subtilis ATCC 21332. FEBS Letters, 231(1), 107–110.
Nitschke, M., & Pastore, G. M. (2006). Production and properties of a surfactant obtained from Bacillus subtilis grown on cassava wastewater. Bioresource Technology, 97, 336–341.
Kowall, M., Vater, J., Kluge, B., Stein, T., Franke, P., & Ziessow, D. (1998). Separation and characterization of surfactin isoforms produced by Bacillus subtilis OKB 105. Journal of Colloid and Interface Science, 204, 1–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel-Mawgoud, A.M., Aboulwafa, M.M. & Hassouna, N.AH. Characterization of Surfactin Produced by Bacillus subtilis Isolate BS5. Appl Biochem Biotechnol 150, 289–303 (2008). https://doi.org/10.1007/s12010-008-8153-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8153-z