Abstract
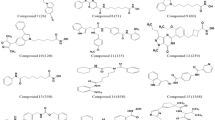
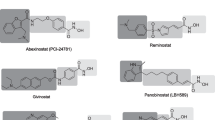
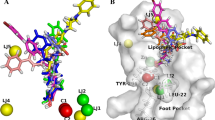
We describe the conformational behavior of histone deacetylase 8 (HDAC8) using molecular dynamics (MD) simulations. HDAC8 conformers were used for the docking studies using some known HDAC inhibitors (HDACi) suberoylanilide hydroxamic acid (SAHA), valproic acid (VPA), aroyl-pyrrole-hydroxy-amide (APHA-8) and tubacin to explore their interactions, binding modes, free energy values. The MD simulation show that HDAC8 make important surface changes at the catalytic site (CS) entrance as well as at two entrances locations in the 14-Å tunnel. In addition, we identify an alternate entrance to the 14-Å tunnel named adjacent to the catalytic site pocket (ACSP). By using docking studies, it was possible to elucidate the importance of hydrophobic and π–π interactions that are the most important for the ligand–HDAC8 complex structural stabilization. In conclusion, the ligand flexibility, molecular weight and chemical moieties (hydroxamic acid, aryl and aliphatic moieties) are the principal properties required to increase the binding affinity on HDAC8.









Similar content being viewed by others
References
Li, E. (2002). Nature Reviews. Genetics, 3, 662–673.
Berger, S. L. (2002). Current Opinion in Genetics & Development, 12, 142–148.
Zhang, Y., & Reinberg, D. (2001). Genes & Development, 15, 2343–2360.
Bolden, J. E., Peart, M. J., & Johnstone, R. W. (2006). Nature Reviews. Drug Discovery, 5, 769–784.
Hildmann, C., Riester, D., & Schwienhorst, A. (2007). Applied Microbiology and Biotechnology, 75, 487–497.
Riester, D., Hildmann, C., & Schwienhorst, A. (2007). Applied Microbiology and Biotechnology, 75, 499–514.
Gregoretti, I. V., Lee, Y. M., & Goodson, H. V. (2004). Journal of Molecular Biology, 338, 17–31.
de Ruijter, A. J., van Gennip, A. H., Caron, H. N., Kemp, S., & van Kuilenburg, A. B. (2003). The Biochemical Journal, 370, 737–749.
Minucci, S., & Pelicci, P. G. (2006). Nature Reviews. Cancer, 6, 38–51.
Shuttleworth, S. J., Bailey, S. G., & Townsend, P. A. (2010). Current Drug Targets, 11, 1430–1438.
Yoo, C. B., & Jones, P. A. (2006). Nature Reviews. Drug Discovery, 5, 37–50.
Oehme, I., Deubzer, H. E., Wegener, D., Pickert, D., Linke, J. P., Hero, B., et al. (2009). Clinical Cancer Research, 15, 91–99.
Krennhrubec, K., Marshall, B. L., Hedglin, M., Verdin, E., & Ulrich, S. M. (2007). Bioorganic & Medicinal Chemistry Letters, 17, 2874–2878.
Vannini, A., Volpari, C., Filocamo, G., Casavola, E. C., Brunetti, M., Renzoni, D., et al. (2004). Proceedings of the National Academy of Sciences of the United States of America, 101, 15064–15069.
Balasubramanian, S., Ramos, J., Luo, W., Sirisawad, M., Verner, E., & Buggy, J. J. (2008). Leukemia, 22, 1026–1034.
Estiu, G., Greenberg, E., Harrison, C. B., Kwiatkowski, N. P., Mazitschek, R., Bradner, J. E., et al. (2008). Journal of Medicinal Chemistry, 51, 2898–2906.
Bieliauskas, A. V., & Pflum, M. K. (2008). Chemical Society Reviews, 37, 1402–1413.
Estiu, G., West, N., Mazitschek, R., Greenberg, E., Bradner, J. E., & Wiest, O. (2010). Bioorganic & Medicinal Chemistry, 18, 4103–4110.
Nussinov, R., & Tsai, C.-J. (2012). Current Pharmaceutical Design, 18, 1311–1316.
Knegtel, R. M., Kuntz, I. D., & Oshiro, C. M. (1997). Journal of Molecular Biology, 266, 424–440.
Alonso, H., Bliznyuk, A. A., & Gready, J. E. (2006). Medicinal Research Reviews, 26, 531–568.
Altschul, S. F., Gish, W., Miller, W., Myers, E. W., & Lipman, D. J. (1990). Journal of Molecular Biology, 215, 403–410.
Gille, C., & Frommel, C. (2001). Bioinformatics, 17, 377–378.
Mai, A., Massa, S., Ragno, R., Cerbara, I., Jesacher, F., Loidl, P., et al. (2003). Journal of Medicinal Chemistry, 46, 512–524.
Mai, A., Massa, S., Valente, S., Simeoni, S., Ragno, R., Bottoni, P., et al. (2006). ChemMedChem, 1, 225–237.
Blackwell, L., Norris, J., Suto, C. M., & Janzen, W. P. (2008). Life Sciences, 82, 1050–1058.
Gottlicher, M., Minucci, S., Zhu, P., Kramer, O. H., Schimpf, A., Giavara, S., et al. (2001). The EMBO Journal, 20, 6969–6978.
Khan, N., Jeffers, M., Kumar, S., Hackett, C., Boldog, F., Khramtsov, N., et al. (2008). The Biochemical Journal, 409, 581–589.
Wong, J. C., Hong, R., & Schreiber, S. L. (2003). Journal of the American Chemical Society, 125, 5586–5587.
Haggarty, S. J., Koeller, K. M., Wong, J. C., Grozinger, C. M., & Schreiber, S. L. (2003). Proceedings of the National Academy of Sciences of the United States of America, 100, 4389–4394.
Butler, K. V., Kalin, J., Brochier, C., Vistoli, G., Langley, B., & Kozikowski, A. P. (2010). Journal of the American Chemical Society, 132, 10842–10846.
Mai, A., Massa, S., Rotili, D., Cerbara, I., Valente, S., Pezzi, R., et al. (2005). Medicinal Research Reviews, 25, 261–309.
Miller, T. A., Witter, D. J., & Belvedere, S. (2003). Journal of Medicinal Chemistry, 46, 5097–5116.
Wang, D. F., Helquist, P., Wiech, N. L., & Wiest, O. (2005). Journal of Medicinal Chemistry, 48, 6936–6947.
Moradei, O. M., Mallais, T. C., Frechette, S., Paquin, I., Tessier, P. E., Leit, S. M., et al. (2007). Journal of Medicinal Chemistry, 50, 5543–5546.
Methot, J. L., Chakravarty, P. K., Chenard, M., Close, J., Cruz, J. C., Dahlberg, W. K., et al. (2008). Bioorganic & Medicinal Chemistry Letters, 18, 973–978.
Bermudez-Lugo, J. A., Perez-Gonzalez, O., Rosales-Hernandez, M. C., Ilizaliturri-Flores, I., Trujillo-Ferrara, J., & Correa-Basurto, J. (2012). Journal of Molecular Modeling, 18, 2301–2310.
Somoza, J. R., Skene, R. J., Katz, B. A., Mol, C., Ho, J. D., Jennings, A. J., et al. (2004). Structure, 12, 1325–1334.
Trott, O., & Olson, A. J. (2010). Journal of Computational Chemistry, 31, 455–461.
Sali, A., & Blundell, T. L. (1993). Journal of Molecular Biology, 234, 779–815.
Phillips, J. C., Braun, R., Wang, W., Gumbart, J., Tajkhorshid, E., Villa, E., et al. (2005). Journal of Computational Chemistry, 26, 1781–1802.
Pettersen, E. F., Goddard, T. D., Huang, C. C., Couch, G. S., Greenblatt, D. M., Meng, E. C., et al. (2004). Journal of Computational Chemistry, 25, 1605–1612.
Frisch, M. J. T. G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Montgomery, J. A., et al. (2003). Gaussian 03, revision A, 9th ed. Pittsburgh, PA: Gaussian Inc.
Frisch, M. J. T. G., & Schlegel, H. B. (1998). Gaussian 98. Pittsburgh: Gaussian, Inc.
Varetto, U. (2000). MOLEKEL Version 4.3. Lugano (Switzerland): Swiss National Supercomputing Centre.
Gasteiger, J., & Marsili, M. (1980). Tetrahedron, 36, 3219–3228.
Singh, U. C., & Kollman, P. A. (1984). Journal of Computational Chemistry, 5, 129–145.
Kollman, P. A., Grootenhuis, P. D. J., & Lopez, M. A. (1989). Pure and Applied Chemistry, 61, 593–596.
Goodford, P. J. (1985). Journal of Medicinal Chemistry, 28, 849–857.
Wang, R., Lai, L., & Wang, S. (2002). Journal of Computer-Aided Molecular Design, 16, 11–26.
Brooks, B. R., Bruccoleri, R. E., Olafson, B. D., States, D. J., Swaminathan, S., & Karplus, M. (1983). Journal of Computational Chemistry, 4, 187–217.
Humphrey, W., Dalke, A., & Schulten, K. (1996). VMD: Visual molecular dynamics. Journal of Molecular Graphics, 14, 33–38.
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W., & Klein, M. L. (1983). Comparison of simple potential functions for simulating liquid water. The Journal of Chemical Physics, 79, 926.
Batcho, P. F., Case, D. A., & Schlick, T. (2001). The Journal of Chemical Physics, 115, 4003.
Ryckaert, J.-P., Ciccotti, G., & Berendsen, H. J. C. (1977). Journal of Computational Physics, 23, 327–341.
Glykos, N. M. (2006). Journal of Computational Chemistry, 27, 1765–1768.
Lee, H., Rezai-Zadeh, N., & Seto, E. (2003). Molecular and Cellular Biology, 24, 765–773.
Dowling, D. P., Gantt, S. L., Gattis, S. G., Fierke, C. A., & Christianson, D. W. (2008). Biochemistry, 47, 13554–13563.
Laskowski, R. A., MacArthur, M. W., Moss, D. S., & Thornton, J. M. (1993). Journal of Applied Crystallography, 26, 283–291.
Colovos, C. Y. T. (1993). Protein Science, 2, 1511–1519.
Wiederstein, M., & Sippl, M. J. (2007). Nucleic Acids Research, 35, W407–W410.
Dale, G. E., Oefner, C., & D’Arcy, A. (2003). Journal of Structural Biology, 142, 88–97.
Vannini, A., Volpari, C., Gallinari, P., Jones, P., Mattu, M., Carfi, A., et al. (2007). EMBO Reports, 8, 879–884.
Whitehead, L., Dobler, M. R., Radetich, B., Zhu, Y., Atadja, P. W., Claiborne, T., et al. (2011). Bioorganic & Medicinal Chemistry, 19, 4626–4634.
Thangapandian, S., John, S., Lee, Y., Kim, S., & Lee, K. W. (2011). International Journal of Molecular Sciences, 12, 9440–9462.
Saha, S., Banerjee, S., & Ganguly, S. (2010). International Journal of ChemTech Research, 2, 932–936.
Kroemer, R. T. (2007). Current Protein & Peptide Science, 8, 312–328.
Bora Tatar, G. T. T. D., Yelekc, K.¸., & Yurter, H. (2011). Turkish Journal of Chemistry, 35, 861–870.
Finnin, M. S., Donigian, J. R., Cohen, A., Richon, V. M., Rifkind, R. A., Marks, P. A., et al. (1999). Nature, 401, 188–193.
Acknowledgments
The work was supported by grants from CONACYT (132353), CYTED: 214RT0482, PIFI-SIP-COFAA, grant SIP20140252, from IPN, and scholarships to YSL from CONACYT. Finally, we thank to the referee for their fruitful suggestions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 3045 kb)
Rights and permissions
About this article
Cite this article
Sixto-López, Y., Gómez-Vidal, J.A. & Correa-Basurto, J. Exploring the Potential binding Sites of Some Known HDAC Inhibitors on Some HDAC8 Conformers by Docking Studies. Appl Biochem Biotechnol 173, 1907–1926 (2014). https://doi.org/10.1007/s12010-014-0976-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-0976-1