Abstract
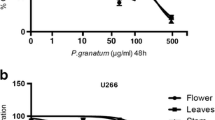
Multiple myeloma (MM) is an incurable B cell neoplasm causing significant morbidity and mortality. Despite recent advances in the MM-treatment, the disease still remains incurable; necessitating a search for new therapeutic agents. Therefore, Strychnos nux-vomica L. root extract (SN) was screened using the human MM-cell line, U266B1. SN-extract demonstrated anti-proliferative effect in a time- and dose-dependent manner. Morphological studies, cell cycle analysis by FACS indicate apoptosis. Furthermore, assessment of signaling molecules like IL-6 (interleukin-6) and CD-138 (Sydencan-1) confirmed apoptosis. The anti-proliferative activity of SN-extract is associated with apoptosis, which is likely due to the presence of alkaloids, strychnine and brucine, which have been identified by MALDI-TOF-MS and RP-HPLC analysis



Similar content being viewed by others
References
Kovacs, E. (2010). Investigation of the proliferation, apoptosis/necrosis, and cell cycle phases in several human multiple myeloma cell lines. Comparison of Viscum album QuFrF extract with vincristine in an in vitro model. Scientific World Journal, 10, 311–320.
Ishii, Y., Hsiao, H.-H., Sashida, G., Ito, Y., Miyazawa, K., Kodama, A., et al. (2006). Derivative (1;7)(q10;p10) in multiple myeloma. A sign of therapy-related hidden myelodysplastic syndrome. Cancer Genetics and Cytogenetics, 167, 131–137.
Kovacs, E. (2006). Multiple Myeloma and B cell lymphoma. Investigation of IL-6, IL-6 receptor antagonist (IL-6RA), and GP130 antagonist (GP130A) using various parameters in an in vitro model. The Scientific World Journal, 6, 888–898.
Ghias, K., Ma, C., Gandhi, V., Platanias, L. C., Krett, N. L., & Rosen, S. T. (2005). 8-Amino-adenosine induces loss of phosphorylation of p38 mitogen-activated protein kinase, extracellular signal-regulated kinase 1/2, and Akt kinase: Role in induction of apoptosis in multiple myeloma. Molecular Cancer Therapeautics, 4, 569–577.
Hideshima, T., Podar, K., Chauhan, D., & Anderson, K. C. (2005). Cytokines and signal transduction. Best Practice & Research Clinical Haematology, 18, 509–524.
Mahtouk, K., Tjin, E. P. M., Spaargaren, M., & Pals, S. T. (2010). The HGF/MET pathway as target for the treatment of multiple myeloma and B-cell lymphomas. Biochimica et Biophysica Acta, 1806, 208–219.
Sporn, M. B., & Suh, N. (2000). Chemoprevention of cancer. Carcinogenesis, 21, 525–530.
Liu, H. P., Shi, X. F., Zhang, Y. C., Li, Z. X., Zhang, L., & Wan, Z. Y. (2011). Quantitative analysis of quercetin in Euphorbia helioscopia L by RP-HPLC. Cell Biochemistry and Biophysics, 61, 59–64.
Gacche, R. N., Shegokar, H. D., Gond, D. S., Yang, Z., & Jadhav, A. D. (2011). Evaluation of selected flavonoids as antiangiogenic, anticancer, and radical scavenging agents: An experimental and in silico analysis. Cell Biochemistry and Biophysics, 61, 651–663.
Prasad, M. N. V., Padmalatha, K., Jayaram, K., Raju, N. L., & Teixeira da Silva, J. (2007). Medicinal plants from Deccan ecoregion, India—Traditional knowledge, ethnophamacology, cultivation, utilization, biotechnology and conservation. Medicinal and Aromatic Plant Science and Biotechnology, 1, 1155–1208.
Bandopadhyay, J., & De, B. (1997). Seasonal variation of strychnine and brucine in vegetative parts of Strychnos nux-vomica. International Journal of Pharmacognosy, 35, 349–353.
Philippe, G., Angenot, L., Tits, M., & Frederich, M. (2004). About the toxicity of some Strychnos species and their alkaloids. Toxicon, 44, 405–416.
Rao, P. S., & Prasad, M. N. V. (2008). Extraction, Purification and Characterization of Indole Alkaloids from Strychnos wallichiana L.—An endangered medicinal plant from India. Medicinal and Aromatic Plant Science and Biotechnology, 2, 63–67.
Cai, B. C., Wang, T. S., Krokawa, M., Shiraki, K., & Hattori, M. (1998). Cytotoxicities of alkaloids from processed and unprocessed seeds of Strychnos nux-vomica. Acta Pharmacologica Sinica, 19, 425–428.
Deng, X. K., Yin, W., Li, W. D., Yin, F. Z., Lu, X. Y., Zang, X. C., et al. (2006). The anti-tumor effects of alkaloids from the seeds of Strychnos nux-vomica on HepG2 cells and its possible mechanism. Journal of Ethnopharmacology, 106, 179–186.
Yin, W., Deng, X. K., Li, W. D., Yin, F. Z., Lu, X. Y., Zang, X. C., et al. (2007). The cytotoxicity induced by Strychnos nux-vomica proceeds via apoptosis by Cox 2 and caspase-3 in SMMC 7221 cells. Food and Chemical Toxicology, 45, 1700–1708.
Rao, P. S., Ramanadham, M., & Prasad, M. N. V. (2009). Anti-proliferative and cytotoxic effects of Strychnos nux-vomica root extract on the human multiple myeloma cell line—RPMI 8226. Food and Chemical Toxicology, 47, 283–288.
Wu, W., Qiao, C., Liang, Z., Xu, H., Zhao, Z., & Cai, Z. (2007). Alkaloid profiling in crude and processed Strychnos nux-vomica seeds by matrix-assisted laser desorption/ionization-time of flight mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis, 45, 430–436.
Ramanadham, M., & Nageshwari, B. (2010). Anti-proliferative effect of levamisole on human myeloma cell lines in vitro. Journal of Immunotoxicology, 7, 327–332.
Subhashini, J., Mahipal, S. V. K., Reddy, M. C., Reddy, M., Rachamallu, A., & Reddanna, P. (2004). Molecular mechanisms in C-Phcocyanin induced apoptosis in human chronic myeloid leukemia cell line K562. Biochemical Pharmacology, 68, 453–462.
Pitchakarn, P., Suzuki, S., Ogawa, K., Pompimon, W., Takahashi, S., Asamoto, M., et al. (2011). Induction of G1 arrest and apoptosis in androgen-dependent human prostate cancer by Kuguacin J, a triterpenoid from Momordica charantia leaf. Cancer Letters, 306(2), 142–150.
Catlett-Falcone, R., Landowski, T. H., Oshiro, M. M., Turkson, J., Levitzki, A., Savino, R., et al. (1999). Constitutive activation of Stat3 signaling confers resistance to apoptosis in human U266 myeloma cells. Immunity, 10, 105–115.
Mahtouk, K., Hose, D., Raynaud, P., Hundemer, M., Jourdan, M., Jourdan, E., et al. (2007). Heparanase influences expression and shedding of syndecan-1, and its expression by the bone marrow environment is a bad prognostic factor in multiple myeloma. Blood, 109, 4914–4923.
Khotskaya, Y. B., Dai, Y., Ritchie, J. P., MacLeod, V., Yang, Y., Zinn, K., et al. (2009). Syndecan-1 is required for robust growth, vascularization, and metastasis of myeloma tumors in vivo. Journal of Biological Chemistry, 284, 26085–26095.
Reijmers, R. M., Groen, R. W., Rozemuller, H., Kuil, A., de Haan-Kramer, A., Csikós, T., et al. (2010). Targeting EXT-1 reveals a crucial role for heparan sulfate in the growth of multiple myeloma. Blood, 115, 601–604.
Jourdan, M., Ferlin, M., Legouffe, E., Horvathova, M., Liautard, J., Rossi, J. F., et al. (1998). The myeloma cell antigen syndecan-1 is lost by apoptotic myeloma cells. British Journal of Haematology, 100(1998), 637.
Acknowledgments
Thanks are due to the Principal Chief Conservator of Forests, officers of the Andhra Pradesh forest Department (India) for giving us permission to collect plant samples from reserve forest divisions (ref. 15698/02/U2). PSR wishes to acknowledge Prof. M Ramanadham (Dept. of Biochemistry, University of Hyderabad) for his critical guidance, valuable suggestions and also providing access to the cell culture facility. PSR thankfully acknowledges the award of the research fellowship by the “UGC University with potential for excellence.” Part of the work received financial support from the Department of Biotechnology, Govt. of India, New Delhi (to MNVP vide ref. BT/PR2273/PBD/17/117/2000) and the Ministry of Environment and Forest, GOI ref No.10/03/2003-CS/BG.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, P.S., Prasad, M.N.V. Strychnos nux-vomica Root Extract Induces Apoptosis in the Human Multiple Myeloma Cell Line—U266B1. Cell Biochem Biophys 66, 443–450 (2013). https://doi.org/10.1007/s12013-012-9492-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-012-9492-5