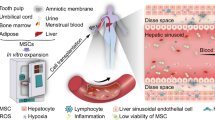
Abstract
Orthotopic liver transplant (OLT) remains the standard of care for end stage liver disease. To circumvent allo-rejection, OLT subjects receive gluococorticoids (GC). We investigated the effects of GC on endogenous mesenchymal stem (stromal) cells (MSCs) in OLT. This question is relevant because MSCs have regenerative potential and immune suppressor function. Phenotypic analyses of blood samples from 12 OLT recipients, at pre-anhepatic, anhepatic and post-transplant (2 h, Days 1 and 5) indicated a significant decrease in MSCs after GC injection. The MSCs showed better recovery in the blood from subjects who started with relatively low MSCs as compared to those with high levels at the prehepatic phase. This drop in MSCs appeared to be linked to GC since similar change was not observed in liver resection subjects. In order to understand the effects of GC on decrease MSC migration, in vitro studies were performed in transwell cultures. Untreated MSCs could not migrate towards the GC-exposed liver tissue, despite CXCR4 expression and the production of inflammatory cytokines from the liver cells. GC-treated MSCs were inefficient with respect to migration towards CXCL12, and this correlated with retracted cytoskeleton and motility. These dysfunctions were partly explained by decreases in the CXCL12/receptor axis. GC-associated decrease in MSCs in OLT recipients recovered post-transplant, despite poor migratory ability towards GC-exposed liver. In total, the study indicated that GC usage in transplant needs to be examined to determine if this could be reduced or avoided with adjuvant cell therapy.







Similar content being viewed by others
References
Wald, O., Pappo, O., Safadi, R., et al. (2004). Involvement of the CXCL12/CXCR4 pathway in the advanced liver disease that is associated with hepatitis C virus or hepatitis B virus. European Journal of Immunology, 34, 1164–1174.
Fausto, N., & Campbell, J. S. (2003). The role of hepatocytes and oval cells in liver regeneration and repopulation. Mechanisms of Development, 120, 117–130.
Russo, F. P., & Parola, M. (2011). Stem and progenitor cells in liver regeneration and repair. Cytotherapy, 13, 135–144.
Friedman, S. L. (2000). Molecular regulation of hepatic fibrosis, an integrated cellular response to tissue injury. The Journal of Biological Chemistry, 275, 2247–2250.
Valenti, L., Fracanzani, A. L., & Fargion, S. (2009). The immunopathogenesis of alcoholic and nonalcoholic steatohepatitis: Two triggers for one disease? Seminars in Immunopathology, 31, 359–369.
McCullough, A. J., O'Shea, R. S., & Dasarathy, S. (2011). Diagnosis and management of alcoholic liver disease. Journal of Digestive Diseases, 12, 257–262.
Kim, H. J., & Lee, H. W. (2013). Important predictor of mortality in patients with end-stage liver disease. Clinical and Molecular Hepatology, 19, 105–115.
Starzl, T. E., Groth, C. G., Brettschneider, L., et al. (1968). Orthotopic homotransplantation of the human liver. Annals of Surgery, 168, 392.
O'Mahony, C. A., & Goss, J. A. (2012). The future of liver transplantation. Texas Heart Institute Journal, 39, 874.
Lucey, M. R., Terrault, N., Ojo, L., et al. (2013). Long-term management of the successful adult liver transplant: 2012 practice guideline by the American Association for the Study of Liver Diseases and the American Society of Transplantation. Liver Transplantation, 19, 3–26.
Forbes, S. J. (2008). Stem cell therapy for chronic liver disease--choosing the right tools for the job. Gut, 57, 153–155.
Zhou, J., & Cidlowski, J. A. (2005). The human glucocorticoid receptor: One gene, multiple proteins and diverse responses. Steroids, 70, 407–417.
Coutinho, A. E., & Chapman, K. E. (2011). The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Molecular and Cellular Endocrinology, 335, 2–13.
Raimondo, M. L., & Burroughs, A. K. (2002). Single-agent immunosuppression after liver transplantation. Drugs, 62, 1587–1597.
Hass, R., Kasper, C., Böhm, S., & Jacobs, R. (2011). Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Communication and Signaling: CCS, 9, 1.
Pisciotta, A., Carnevale, G., Meloni, S., et al. (2015). Human dental pulp stem cells (hDPSCs): Isolation, enrichment and comparative differentiation of two sub-populations. BMC Developmental Biology, 15, 1.
Margini, C., Vukotic, R., Brodosi, L., Bernardi, M., & Andreone, P. (2014). Bone marrow derived stem cells for the treatment of end-stage liver disease. World Journal of Gastroenterology, 9098–9105.
Sherman, L. S., Shaker, M., Mariotti, V., & Rameshwar, P. (2017). Mesenchymal stromal/stem cells in drug therapy: New perspective. Cytotherapy, 19, 19–27.
Salem, H. K., & Thiemermann, C. (2010). Mesenchymal stromal cells: Current understanding and clinical status. Stem Cells, 28, 585–596.
Kean, T. J., Lin, P., Caplan, A. I., & Dennis, J. E. (2013). MSCs: Delivery routes and engraftment, cell-targeting strategies, and immune modulation. Stem Cells International, 2013, 732742.
Castillo, M., Liu, K., Bonilla, L., & Rameshwar, P. (2007). The immune properties of mesenchymal stem cells. International Journal of Biological Sciences, 3, 76.
Cho, K.-A., Ju, S.-Y., Cho, S. J., et al. (2009). Mesenchymal stem cells showed the highest potential for the regeneration of injured liver tissue compared with other subpopulations of the bone marrow. Cell Biology International, 33, 772–777.
Popp, F. C., Renner, P., Eggenhofer, E., et al. (2009). Mesenchymal stem cells as immunomodulators after liver transplantation. Liver Transplantation, 15, 1192–1198.
Sato, K., Ozaki, K., Mori, M., Muroi, K., & Ozawa, K. (2010). Mesenchymal stromal cells for graft-versus-host disease: Basic aspects and clinical outcomes. Journal of Clinical and Experimental Hematopathology, 50, 79–89.
Kim, N., Im, K.-I., Lim, J.-Y., et al. (2013). Mesenchymal stem cells for the treatment and prevention of graft-versus-host disease: Experiments and practice. Annals of Hematology, 92, 1295–1308.
Kapoor, S., Patel, S. A., Kartan, S., Axelrod, D., Capitle, E., & Rameshwar, P. (2012). Tolerance-like mediated suppression by mesenchymal stem cells in patients with dust mite allergy–induced asthma. The Journal of Allergy and Clinical Immunology, 129, 1094–1101.
Corcoran, K. E., Trzaska, K. A., Fernandes, H., et al. (2008). Mesenchymal stem cells in early entry of breast cancer. PloS One, 3, 1–10.
Patel, S. A., Meyer, J. R., Greco, S. J., Corcoran, K. E., Bryan, M., & Rameshwar, P. (2010). Mesenchymal stem cells protect breast cancer cells through regulatory T cells: Role of mesenchymal stem cell-derived TGF-β. Journal of Immunology, 184, 5885–5894.
Son, B. R., Marquez-Curtis, L. A., Kucia, M., et al. (2006). Migration of bone marrow and cord blood mesenchymal stem cells in vitro is regulated by stromal-derived factor-1-CXCR4 and hepatocyte growth factor-c-met axes and involves matrix metalloproteinases. Stem Cells, 24, 1254–1264.
Hao, N.-B., Li, C.-Z., Lü, M.-H., et al. (2015). SDF-1/CXCR4 Axis promotes MSCs to repair liver injury partially through trans-differentiation and fusion with hepatocytes. Stem Cells International, 2015, 960387.
Hu, C., Yong, X., Li, C., et al. (2013). CXCL12/CXCR4 axis promotes mesenchymal stem cell mobilization to burn wounds and contributes to wound repair. The Journal of Surgical Research, 183, 427–434.
Won, Y.-W., Patel, A. N., & Bull, D. A. (2014). Cell surface engineering to enhance mesenchymal stem cell migration toward an SDF-1 gradient. Biomaterials, 35, 5627–5635.
De Becker, A., & Van Riet, I. (2016). Homing and migration of mesenchymal stromal cells: How to improve the efficacy of cell therapy? World Journal of Stem Cells, 8, 73.
Zhang, S., Lv, C., Yang, X., et al. (2014). Corticosterone mediates the inhibitory effect of restraint stress on the migration of mesenchymal stem cell to carbon tetrachloride-induced fibrotic liver by downregulating CXCR4/7 expression. Stem Cells and Development, 24, 587–596.
Würth, R., Bajetto, A., Harrison, J. K., Barbieri, F., & Florio, T. (2014). CXCL12 modulation of CXCR4 and CXCR7 activity in human glioblastoma stem-like cells and regulation of the tumor microenvironment. Frontiers in Cellular Neuroscience, 8, 144.
Chan, J. L., Tang, K. C., Patel, A. P., et al. (2006). Antigen-presenting property of mesenchymal stem cells occurs during a narrow window at low levels of interferon-γ. Blood, 107, 4817–4824.
Marquez-Curtis, L. A., & Janowska-Wieczorek, A. (2013). Enhancing the migration ability of mesenchymal stromal cells by targeting the SDF-1/CXCR4 axis. BioMed Research International, 2013, 561098.
Greco, S. J., Patel, S. A., Bryan, M., Pliner, L. F., Banerjee, D., & Rameshwar, P. (2011). AMD3100-mediated production of interleukin-1 from mesenchymal stem cells is key to chemosensitivity of breast cancer cells. American Journal of Cancer Research, 1, 701.
Soeder, Y., Loss, M., Johnson, C. L., et al. (2015). First-in-human case study: Multipotent adult progenitor cells for immunomodulation after liver transplantation. Stem Cells Translational Medicine, 4, 899–904.
Shah, K. (2012). Mesenchymal stem cells engineered for cancer therapy. Advanced Drug Delivery Reviews, 64, 739–748.
Jones, B. J., & McTaggart, S. J. (2008). Immunosuppression by mesenchymal stromal cells: From culture to clinic. Experimental Hematology, 36, 733–741.
Hammond, A., Ramersdorfer, C., Palitzsch, K., Schölmerich, J., & Lock, G. (1999). Fatal liver failure after corticosteroid treatment of a hepatitis B virus carrier. Deutsche Medizinische Wochenschrift, 124, 687–690.
Steinberg, H., Webb, W. M., & Rafsky, H. A. (1952). Hepatomegaly with fatty infiltration secondary to cortisone therapy: Case report. Gastroenterology, 21, 304–309.
Tacke, F. (2012). Functional role of intrahepatic monocyte subsets for the progression of liver inflammation and liver fibrosis in vivo. Fibrogenesis & Tissue Repair, 5, 1.
Yin, C., Evason, K. J., Asahina, K., & Stainier, D. Y. (2013). Hepatic stellate cells in liver development, regeneration, and cancer. The Journal of Clinical Investigation, 123, 1902–1910.
Kovacsovics-Bankowski, M., Mauch, K., Raber, A., et al. (2008). Pre-clinical safety testing supporting clinical use of allogeneic multipotent adult progenitor cells. Cytotherapy, 10, 730–742.
Acknowledgements
This work was funded by a grant from the FM Kirby Foundation. This work was performed in partial fullfilment of a doctoral thesis for NDW
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of Interest
The authors have no conflict to declare.
Additional information
The work is in partial fullfilment for the doctoral thesis of Nykia D. Walker.
Electronic supplementary material
ESM 1
(DOCX 25 kb)
Rights and permissions
About this article
Cite this article
Walker, N.D., Mourad, Y., Liu, K. et al. Steroid-Mediated Decrease in Blood Mesenchymal Stem Cells in Liver Transplant could Impact Long-Term Recovery. Stem Cell Rev and Rep 13, 644–658 (2017). https://doi.org/10.1007/s12015-017-9751-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-017-9751-3