Abstract
Purpose
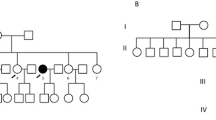
Germline mutations in DNA repair-related genes have been recently reported in cases with familial non-medullary thyroid carcinoma (FNMTC). A Portuguese family from the Roma ethnic group with four members affected with papillary thyroid carcinoma (PTC), and three members with multinodular goiter (MNG) was identified. The aim of this study was to investigate the involvement of DNA repair-related genes in the etiology of FNMTC in this family and in the Roma ethnic group.
Methods
Ninety-four hereditary cancer predisposition genes were analyzed through next-generation sequencing. Sanger sequencing was used for variant confirmation and screening. Twelve polymorphic markers were genotyped for haplotype analysis in the CHEK2 locus.
Results
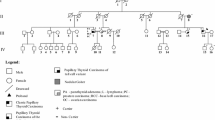
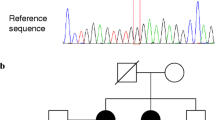
A germline pathogenic frameshift variant in the CHEK2 gene [c.596dupA, p.(Tyr199Ter)] was detected in homozygosity in the proband (PTC) and in his brother (MNG), being heterozygous in his mother (PTC), two sisters (PTC), and one nephew (MNG). This variant was absent in 100 general population controls. The screening of the CHEK2 variant was extended to other Roma individuals, being detected in 2/33 Roma patients with thyroid cancer, and in 1/15 Roma controls. Haplotype segregation analysis identified a common ancestral core haplotype (Hcac), covering 10 Mb in the CHEK2 locus, shared by affected CHEK2 variant carriers. Analysis of 62 individuals CHEK2 wild-type indicated that none presented the Hcac haplotype. The estimated age for this variant suggested that it was transmitted by a relatively recent common ancestor.
Conclusions
We identified a founder CHEK2 pathogenic variant, which is likely to underlie thyroid cancer and other cancer manifestations in the Roma population.



Similar content being viewed by others
References
Q.T. Nguyen, E.J. Lee, M.G. Huang, Y.I. Parkn, A. Khullar, R.A. Plodkowski,Diagnosis and treatment of patients with thyroid cancer. Am. Health Drug Benefits. 8(1), 30–40 (2015)
R.L. Siegel, K.D. Mille, A. Jemal, Cancer Statistics, 2017. CA Cancer J. Clin. 67(1), 7–30 (2017)
C. Sturgeon, O.H. Clark, Familial nonmedullary thyroid cancer. Thyroid 15(6), 588–593 (2005)
A.E. Pinto, G.L. Silva, R. Henrique, F.D. Menezes, M.R. Teixeira, V. Leite, B.M. Cavaco, Familial vs sporadic papillary thyroid carcinoma: a matched-case comparative study showing similar clinical/prognostic behaviour. Eur. J. Endocrinol. 170, 321–327 (2014)
S.P. Yang, J. Ngeo, Familial non-medullary thyroid cancer: unraveling the genetic maze. Endocr. Relat. Cancer 23(12), R577–R595 (2016)
J. Klubo-Gwiezdzinska, Y. Kushchayeva, S.K. Gara, E. Kebebew. Familial non-medullary thyroid cancer. In Practical Management of Thyroid Cancer, ed. by U.K. Mallick, C. Harmer (Springer, Cham, 2018), pp. 241–270
J.A. Carney, C. Lyssikatos, R.R. Seethala, P. Lakatos, A. Perez-Atayde, H. Lahner et al. The spectrum of thyroid gland pathology in carney complex: the importance of Follicular Carcinoma. Am. J. Surg. Pathol. 42(5), 587–594 (2018)
J. Dotto, V. Nosé, Familial thyroid carcinoma: a diagnostic algorithm. Adv. Anat. Pathol. 15(6), 332–349 (2008)
J.E. Hall, D.J. Abdollahian, R.J. Sinard, Thyroid disease associated with Cowden syndrome: a meta-analysis. Head Neck 35(8), 1189–1194 (2013)
C.D. Malchoff, D.M. Malchoff, The genetics of hereditary nonmedullary thyroid carcinoma. J. Clin. Endocrinol. Metab. 87(6), 2455–2459 (2002)
C.D. Malchoff, M. Sarfarazi, B. Tendler, F. Forouhar, G. Whalen, V. Joshi et al. Papillary thyroid carcinoma associated with papillary renal neoplasia: genetic linkage analysis of a distinct heritable tumor syndrome. J. Clin. Endocrinol. Metab. 85(5), 1758–1764 (2000)
M. Rohaizak, A.Y. Jasmi, M.A.M. Ismail, M.J.J. Munchar, F.A. Meah, Thyroid carcinoma in patients with familial adenomatous polyposis. Asian J. Surg. 26(3), 183–185 (2003).
G.R. Bignell, F. Canzian, M. Shayeghi, M. Stark, Y.Y. Shugart, P. Biggs, J. Mangion, R. Hamoudi, J. Rosenblatt, P. Buu et al. Familial nontoxic multinodular thyroid goiter locus maps to chromosome 14q but does not account for familial nonmedullary thyroid cancer. Am. J. Hum. Genet. 61(5), 1123–1130 (1997)
F. Canzian, P. Amati, H.R. Harach, J.L. Kraimps, F. Lesueur, J. Barbier et al. A gene predisposing to familial thyroid tumors with cell oxyphilia maps to chromosome 19p13.2. Am. J. Hum. Genet. 63(6), 1743–1748 (1998)
J.D. McKay, F. Lesueur, L. Jonard, A. Pastore, J. Williamson, L. Hoffman et al. Localization of a susceptibility gene for familial nonmedullary thyroid carcinoma to chromosome 2q21. Am. J. Hum. Genet. 69(2), 440–446 (2001)
B.M. Cavaco, P.F. Batista, L.G. Sobrinho, V. Leite, Mapping a new familial thyroid epithelial neoplasia susceptibility locus to chromosome 8p23.1-p22 by high-density single-nucleotide polymorphism genome-wide linkage analysis. J. Clin. Endocrinol. Metab. 93(11), 4426–4430 (2008).
I. Suh, S. Filetti, M.R. Vriens, M.A. Guerrero, S. Tumino, M. Wong et al. Distinct loci on chromosome 1q21 and 6q22 predispose to familial nonmedullary thyroid cancer: a SNP array-based linkage analysis of 38 families. Surgery. 146(6), 1073–1080 (2009)
H. He, R. Nagy, S. Liyanarachchi, H. Jiao, W. Li, S. Suster et al. A susceptibility locus for papillary thyroid carcinoma on chromosome 8q24. Cancer Res. 69(2), 625–631 (2009)
E.S.W. Ngan, B.H.H. Lang, T. Liu, C.K.Y. Shum, M.T. So, D.K.C. Lau et al. A germline mutation (A339V) in thyroid transcription factor-1 (TITF-1/NKX2.1) in patients with multinodular goiter and papillary thyroid carcinoma. J. Natl Cancer Inst. 101(3), 162–175 (2009)
T. Rio Frio, A. Bahubeshi, C. Kanellopoulou, N. Hamel, M. Niedziela, N. Sabbaghian et al. DICER1 mutations in familial multinodular goiter with and without ovarian Sertoli-Leydig cell tumors. JAMA 305(1), 68–77 (2011)
H. He, A. Bronisz, S. Liyanarachchi, R. Nagy, W. Li, Y. Huang et al. SRGAP1 is a candidate gene for papillary thyroid carcinoma susceptibility. J. Clin. Endocrinol. Metab. 98(5), 973–980 (2013)
J.S. Pereira, J.G. da Silva, R.A. Tomaz, A.E. Pinto, M.J. Bugalho, V. Leite et al. Identification of a novel germline FOXE1 variant in patients with familial non-medullary thyroid carcinoma (FNMTC). Endocrine 49(1), 204–214 (2015)
S.K. Gara, L. Jia, M.J. Merino, S.K. Agarwal, L. Zhang, M. Cam et al. Germline HABP2 mutation causing familial nonmedullary thyroid cancer. N. Engl. J. Med. 373(5), 448–455 (2015)
J. Tomsic, H. He, K. Akagi, S. Liyanarachchi, Q. Pan, B. Bertani et al. A germline mutation in SRRM2, a splicing factor gene, is implicated in papillary thyroid carcinoma predisposition. Sci. Rep. 5(1), 1–13 (2015)
C. Liu, Y. Yu, G. Yin, J. Zhang, W. Wen, X. Ruan et al. C14orf93 (RTFC) is identified as a novel susceptibility gene for familial nonmedullary thyroid cancer. Biochem. Biophys. Res. Commun. 482(4), 590–596 (2017)
F. Ye, H. Gao, L. Xiao, Z. Zuo, Y. Liu, Q. Zhao et al. Whole exome and target sequencing identifies MAP2K5 as novel susceptibility gene for familial non-medullary thyroid carcinoma. Int. J. Cancer 144(6), 1321–1330 (2018)
C. Diquigiovanni, C. Bergamini, C. Evangelisti, F. Isidori, A. Vettori, N. Tiso et al. Mutant MYO1F alters the mitochondrial network and induces tumor proliferation in thyroid cancer. Int. J. Cancer. 143(7), 1706–1719 (2018)
S. Fahiminiya, L. de Kock, W.D. Foulkes, Biologic and clinical perspectives on thyroid cancer. N. Engl. J. Med. 375(11), 1054–1067 (2016)
A. Srivastava, A. Kumar, S. Giangiobbe, E. Bonora, K. Hemminki, A. Försti et al. Whole genome sequencing of familial non-medullary thyroid cancer identifies germline alterations in MAPK/ERK and PI3K/AKT signaling pathways. Biomolecules 9(10), 605 (2019)
Y. Yu, L. Dong, D. Li, S. Chuai, Z. Wu, X. Zheng et al. Targeted DNA sequencing detects mutations related to susceptibility among familial non-medullary thyroid cancer. Sci. Rep. 5, 16129 (2015)
Y. Wang, S. Liyanarachchi, K.E. Miller, T.T. Nieminen, D.F. Comiskey Jr, W. Li, H. He, A. de la Chapelle et al. Identification of rare variants predisposing to thyroid cancer. Thyroid 29(7), 946–955 (2019)
Y. Zhao, T. Yu, L. Chen, D. Xie, F. Wang, L. Fu, C. Cheng, Y. Li, X. Zhu, G. Miao, A germline CHEK2 mutation in a family with papillary thyroid cancer. Thyroid. 30(6), 924–930 (2020)
S. Martins, F. Calafell, C. Gaspar, V.C. Wong, I. Silveira, G.A. Nicholson et al. Asian origin for the worldwide-spread mutational event in Machado-Joseph disease. Arch. Neurol. 64(10), 1502–1508 (2007)
J.H. Choi, R. Balasubramanian, P.H. Lee, N.D. Shaw, J.E. Hall, L. Plummer, R. Quinton, Expanding the spectrum of founder mutations causing isolated gonadotropin-releasing hormone deficiency. J. Clin. Endocrinol. Metab. 100(10), E1378–E1385 (2015)
T. Strachan, A. Read. Human Molecular Genetics, 4th edn (Garland Science, Taylor & Francis Group, Madison Avenue, New York, 2010)
P.M. Machado, R.D. Brandão, B.M. Cavaco, J. Eugénio, S. Bento, M. Nave, F. Vaz, Screening for a BRCA2 rearrangement in high-risk breast/ovarian cancer families: evidence for a founder effect and analysis of the associated phenotypes. J. Clin. Oncol. 25(15), 2027–2034 (2007)
M. Pinheiro, I. Francisco, C. Pinto, A. Peixoto, I. Veiga, B. Filipe, R. Santos, The nonsense mutation MSH2 c. 2152C> T shows a founder effect in Portuguese Lynch syndrome families. Genes, Chromosomes Cancer. 58(9), 657–664 (2019)
S. Matsuoka, M. Huang, S.J. Elledge, Linkage of ATM to cell cycle regulation by the Chk2 protein kinase. Science. 282(5395), 1893–1897 (1998)
B.M. Cavaco, P.F. Batista, C. Martins, A. Banito, F. do Rosário, E. Limbert, L.G. Sobrinho, V. Leite, Familial non-medullary thyroid carcinoma (FNMTC): analysis of fPTC/PRN, NMTC1, MNG1 and TCO susceptibility loci and identification of somatic BRAF and RAS mutations. Endocr. Relat. Cancer. 15(1), 207–215 (2008)
I.J. Marques, M.M. Moura, R. Cabrera, A.E. Pinto, J. Simões-Pereira, C. Santos, F.D. Menezes, D. Montezuma, R. Henrique, M. Rodrigues Teixeira, V. Leite, B.M. Cavaco, Identification of somatic TERT promoter mutations in familial nonmedullary thyroid carcinomas. Clin. Endocrinol. 87(4), 394–399 (2017)
M. Fiore, X. Pillois, P. Nurden, A.T. Nurden, F. Austerlitz, Founder effect and estimation of the age of the French Gypsy mutation associated with Glanzmann thrombasthenia in Manouche families. Eur. J. Hum. Genet. 19(9), 981–987 (2011)
M. Cabral, A.V. Neves. Caracterização das condições de habitação das Comunidades Ciganas residentes em Portugal (Instituto da Habitação e da Reabilitação Urbana, I.P, Lisboa, 2014) pp. 1–28
C. Cybulski, B. Górski, T. Huzarski, B. Masojć, M. Mierzejewski, T. Dębniak et al. CHEK2 is a multiorgan cancer susceptibility gene. Am. J. Hum. Genet. 75(6), 1131–1135 (2004)
C. Stanislaw, Y. Xue, W.R. Wilcox, Genetic evaluation and testing for hereditary forms of cancer in the era of next-generation sequencing. Cancer Biol. Med. 13(1), 55–67 (2016)
R. Vassilopoulou-Sellin, L. Palmer, S. Taylor, C.S. Cooksley, Incidence of breast carcinoma in women with thyroid carcinoma. Cancer. 85(3), 696–705 (1999)
A.Y. Chen, L. Levy, H. Goepfert, B.W. Brown, M.R. Spitz, R. Vassilopoulou-Sellin, The development of breast carcinoma in women with thyroid carcinoma. Cancer. 92(2), 225–231 (2001)
C.I. Li, M.A. Rossing, L.F. Voigt, J.R. Daling, Multiple primary breast and thyroid cancers: role of age at diagnosis and cancer treatments (United States). Cancer Causes Control. 11(9), 805–811 (2000)
M. Siołek, C. Cybulski, D. Gąsior-Perczak, A. Kowalik, B. Kozak-Klonowska, A. Kowalska et al. CHEK2 mutations and the risk of papillary thyroid cancer. Int. J. Cancer. 137(3), 548–552 (2015)
M.A. Adank, M.A. Jonker, I. Kluijt, S.E. van Mil, R.A. Oldenburg, W.J. Mooi, A.W. van der Vaart, CHEK2* 1100delC homozygosity is associated with a high breast cancer risk in women. J. Med. Genet. 48(12), 860–863 (2011)
M. Kaczmarek-Ryś, K. Ziemnicka, S.T. Hryhorowicz, K. Górczak, J. Hoppe-Gołębiewska, M. Skrzypczak-Zielińska, I. Siatkowski, The c. 470T>C CHEK2 missense variant increases the risk of differentiated thyroid carcinoma in the Great Poland population. Hered Cancer Clin. Pract. 13(1), 8 (2015)
P. Apostolou, F. Fostira, V. Mollaki, A. Delimitsou, M. Vlassi, G. Pentheroudakis, I. Konstantopoulou, Characterization and prevalence of two novel CHEK2 large deletions in Greek breast cancer patients. J. Hum. Genet. 63(8), 877–886 (2018)
E. Złowocka, C. Cybulski, B. Górski, T. Dębniak, M. Słojewski, D. Wokołorczyk, R.J. Scott, Germline mutations in the CHEK2 kinase gene are associated with an increased risk of bladder cancer. Int. J. Cancer. 122(3), 583–586 (2008)
Acknowledgements
The authors are grateful to the patients and their families for their cooperation.
Funding
This work was funded by Liga Portuguesa Contra o Cancro — Núcleo Regional do Sul (LPCC-NRS), Televisão Independente (TVI), Instituto Português de Oncologia de Lisboa Francisco Gentil (IPOLFG), iNOVA4Health — UIDB/04462/2020 (a program financially supported by Fundação para a Ciência e Tecnologia/Ministério da Educação e Ciência), and Associação de Endocrinologia Oncológica.
Author information
Authors and Affiliations
Contributions
C.P.: investigation, methodology, formal analysis, data analysis, writing—original draft, writing—review and editing; I.J.M.: investigation, methodology, formal analysis, data analysis, writing—review and editing; D.D.: investigation, clinical data analysis, writing—review and editing; A.S.: methodology, data analysis, writing—review and editing; V.L.: investigation, clinical data analysis, writing—review and editing; B.M.C.: investigation, conceptualization, supervision, formal analysis, data analysis, funding acquisition, writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This study was approved by the Ethical Committee of Instituto Português de Oncologia de Lisboa Francisco Gentil, and is in accordance with the ethical standards from the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
The collection of biological samples from all subjects involved in this study was performed after written informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Pires, C., Marques, I.J., Dias, D. et al. A pathogenic variant in CHEK2 shows a founder effect in Portuguese Roma patients with thyroid cancer. Endocrine 73, 588–597 (2021). https://doi.org/10.1007/s12020-021-02660-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02660-x