Abstract
Objective
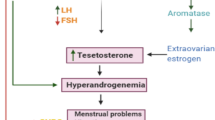
Polycystic ovary syndrome (PCOS) is one of the most common reproductive, endocrine, and metabolic disorder in premenopausal women. Even though the pathophysiology of PCOS is complex and obscure, the disorder is prominently considered as the syndrome of hyperandrogenism. C-Terminal binding protein 1 antisense (CTBP1-AS) acts as a novel androgen receptor regulating long noncoding RNA (lncRNA). Therefore, the present study was aimed to establish the possible association of androgen receptor regulating long noncoding RNA CTBP1-AS with PCOS.
Methods
A total of 178 subjects including 105 PCOS cases and 73 age-matched healthy controls were recruited for the study. The anthropometric, hormonal, and biochemical parameters of all subjects were analyzed. Total RNA was isolated from peripheral venous blood and expression analysis was done by quantitative real-time PCR. The correlation analysis was performed to evaluate the association between and various clinical parameters and lncRNA CTBP1-AS expression.
Results and conclusion
The mean expression level of CTBP1-AS was found to be significantly higher in the PCOS women than in the healthy controls (-lnCTBP1-AS, 4.23 ± 1.68 versus 1.24 ± 0.29, P < 0.001). Furthermore, subjects with higher expression level of CTBP1-AS had significantly higher risk of PCOS compared to subjects with low levels of CTBP1-AS expression (actual OR = 11.36, 95% CI = 5.59–23.08, P < 0.001). The area under receiver operator characteristic (ROC) curve was 0.987 (SE 0.006, 95% CI 0.976–0.99). However, lncRNA CTBP1-AS was found to have no association with different clinical characteristics of PCOS. In conclusion, androgen receptor coregulating lncRNA CTBP1-AS is associated with PCOS women and high expression of CTBP1-AS is a risk factor for PCOS in Kashmiri women


Similar content being viewed by others
Data availability
The data and materials will be provided by authors on reasonable request.
References
H.F. Escobar-Morreale, Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nat. Rev. Endocrinol. 14(5), 270–284 (2018). https://doi.org/10.1038/nrendo.2018.24
R. Azziz, E. Carmina, D. Dewailly et al. Position statement: criteria for defining polycystic ovary syndrome as a predominantly hyperandrogenic syndrome: an androgen excess society guideline. J. Clin. Endocrinol. Metab. 91(11), 4237–4245 (2006). https://doi.org/10.1210/jc.2006-0178
S. Liu, S. Kumari, Q. Hu et al. A comprehensive analysis of coregulator recruitment, androgen receptor function and gene expression in prostate cancer. eLife 6, e28482 (2017). https://doi.org/10.7554/eLife.28482
S. Djebali, C.A. Davis, A. Merkel et al. Landscape of transcription in human cells. Nature 489(7414), 101–108 (2012). https://doi.org/10.1038/nature11233
Q. Liao, H. Xiao, D. Bu et al. NcFANs: a web server for functional annotation of long non-coding RNAs. Nucleic Acids Res. 39(web server issue), w1118–w1124 (2011). https://doi.org/10.1093/nar/gkr432
A. Fatica, I. Bozzoni, Long non-coding RNAs: new players in cell differentiation and development. Nat. Rev. Genet. 15(1), 7–21 (2014). https://doi.org/10.1038/nrg3606
M. Sun, W.L. Kraus, From discovery to function: the expanding roles of long noncoding RNAs in physiology and disease. Endocr. Rev. 36(1), 25–64 (2015). https://doi.org/10.1210/er.2014-1034
X. Yang, L. Gao, X. Guo et al. A network based method for analysis of lncRNA-disease associations and prediction of lncRNAs implicated in diseases. PLoS One 9(1), e87797 (2014). https://doi.org/10.1371/journal.pone.0087797
K.I. Takayama, K. Horie-Inoue, S. Katayama et al. Androgen-responsive long noncoding RNA CTBP1-AS promotes prostate cancer. EMBO J. 32(12), 1665–1680 (2013). https://doi.org/10.1038/emboj.2013.99
B.C.J.M. Fauser, Tarlatzis, Fauser et al. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Hum. Reprod. 19(1), 41–47 (2004). https://doi.org/10.1093/humrep/deh098
D.C. Rio, M. Ares, G.J. Hannon, T.W. Nilsen, Purification of RNA using TRIzol (TRI Reagent). Cold Spring Harb. Protoc. 2010(6), pdb.prot5439 (2010). https://doi.org/10.1101/pdb.prot5439
D.T. Schmittgen, K.J. Livak, Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 3(6), 1101–1108 (2008)
P. Anagnostis, B.C. Tarlatzis, R.P. Kauffman, Polycystic ovarian syndrome (PCOS): long-term metabolic consequences. Metab. Clin. Exp. 86, 33–43 (2018). https://doi.org/10.1016/j.metabol.2017.09.016
Z. Behboodi Moghadam, B. Fereidooni, M. Saffari, A. Montazeri, Measures of health-related quality of life in pcos women: a systematic review. Int. J. Womens Health 10, 397–408 (2018). https://doi.org/10.2147/IJWH.S165794
S.U.A. Rasool, S. Ashraf, M. Nabi, F. Rashid, K.M. Fazili, S. Amin, Elevated fasting insulin is associated with cardiovascular and metabolic risk in women with polycystic ovary syndrome. Diabetes Metab. Syndr.: Clin. Res. Rev. 13(0), 2098–2105 (2019). https://doi.org/10.1016/j.dsx.2019.05.003
S. Ashraf, S.U.A. Rasool, M. Nabi et al. CYP17 gene polymorphic sequence variation is associated with hyperandrogenism in Kashmiri women with polycystic ovarian syndrome. Gynecol. Endocrinol. 37(3), 230–234 (2020). https://doi.org/10.1080/09513590.2020.1770724
S.U.A. Rasool, S. Ashraf, M. Nabi et al. Insulin gene VNTR class III allele is a risk factor for insulin resistance in Kashmiri women with polycystic ovary syndrome. Meta Gene 21, 100597 (2019). https://doi.org/10.1016/j.mgene.2019.100597
S. Thathapudi, V. Kodati, J. Erukkambattu, A. Katragadda, U. Addepally, Q. Hasan, Anthropometric and biochemical characteristics of polycystic ovarian syndrome in south indian women using AES-2006 criteria. Int. J. Endocrinol. Metab. 21(1), e12470 (2014). https://doi.org/10.5812/ijem.12470
C.N. Wijeyaratne, R.D.A. Seneviratne, S. Dahanayake et al. Phenotype and metabolic profile of South Asian women with polycystic ovary syndrome (PCOS): results of a large database from a specialist Endocrine Clinic. Hum. Reprod. 26(1), 202–213 (2011). https://doi.org/10.1093/humrep/deq310
E.T. Wang, C.N. Kao, K. Shinkai, L. Pasch, M.I. Cedars, H.G. Huddleston, Phenotypic comparison of Caucasian and Asian women with polycystic ovary syndrome: a cross-sectional study. Ferti. Steril. 100(1), 214–218 (2013). https://doi.org/10.1016/j.fertnstert.2013.03.010
S. Tan, L.P. Bechmann, S. Benson et al. Apoptotic markers indicate nonalcoholic steatohepatitis in polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 95(1), 343–348 (2010). https://doi.org/10.1210/jc.2009-1834
S. Minato, N. Sakane, K. Kotani et al. Prevalence and risk factors of elevated liver enzymes in Japanese women with polycystic ovary syndrome. J. Clin. Med. Res. 10(12), 904–910 (2018). https://doi.org/10.14740/jocmr3639
M.J. Cox, M.C. Edwards, V. Rodriguez Paris et al. Androgen action in adipose tissue and the brain are key mediators in the development of PCOS traits in a mouse model. Endocrinology 161(7), bqaa061 (2020). https://doi.org/10.1210/endocr/bqaa061
K.A. Walters, D.J. Handelsman, Role of androgens in the ovary. Mol. Cell. Endocrinol 465(0), 36–47 (2018). https://doi.org/10.1016/j.mce.2017.06.026
A.S.L. Caldwell, M.C. Edwards, R. Desai et al. Neuroendocrine androgen action is a key extraovarian mediator in the development of polycystic ovary syndrome. Proc. Natl Acad. Sci. USA. 114(16), E3334–E3343 (2017). https://doi.org/10.1073/pnas.1616467114
K.A. Walters, M.C. Edwards, D. Tesic et al. The role of central androgen receptor actions in regulating the hypothalamic-pituitary-ovarian axis. Neuroendocrinology 106(4), 389–400 (2018). https://doi.org/10.1159/000487762
J.C.R. Fernandes, S.M. Acuña, J.I. Aoki, L.M. Floeter-Winter, S.M. Muxel, Long non-coding RNAs in the regulation of gene expression: physiology and disease. Noncoding RNA 5(1), 17 (2019). https://doi.org/10.3390/ncrna5010017
M.K. Iyer, Y.S. Niknafs, R. Malik et al. The landscape of long noncoding RNAs in the human transcriptome. Nat. Genet. 47(3), 199–208 (2015). https://doi.org/10.1038/ng.3192
M. Knoll, H.F. Lodish, L. Sun, Long non-coding RNAs as regulators of the endocrine system. Nat. Rev. Endocrinol. 11, 151–160 (2015). https://doi.org/10.1038/nrendo.2014.229
M. Zhao, Y. Qiu, B. Yang et al. Long non-coding RNAs involved in gynecological cancer. Int. J. Gynecol. Cancer 24(7), 1140–1145 (2014). https://doi.org/10.1097/IGC.0000000000000212
Z. Liu, C. Hao, D. Song, N. Zhang, H. Bao, Q. Qu, Androgen receptor coregulator CTBP1-AS is associated with polycystic ovary syndrome in Chinese women: a preliminary study. Reprod. Sci. 22(7), 829–837 (2015). https://doi.org/10.1177/1933719114565037
H.F. Escobar-Morreale, Reproductive endocrinology: menstrual dysfunction–a proxy for insulin resistance in PCOS? Nat. Rev. Endocrinol. 10(1), 10–11 (2014)
B.C.J.M. Fauser, B.C. Tarlatzis, R.W. Rebar et al. Consensus on womens health aspects of polycystic ovary syndrome (PCOS). Hum. Reprod. 27(1), 14–24 (2012). https://doi.org/10.1093/humrep/der396
H. Teede, A. Deeks, L. Moran, Polycystic ovary syndrome: a complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 8, 41 (2010). https://doi.org/10.1186/1741-7015-8-41
C. Kshetrimayum, A. Sharma, V.V. Mishra, S. Kumar, Polycystic ovarian syndrome: environmental/occupational, lifestyle factors; an overview. J. Turk. Ger. Gynecol. Assoc. 20(4), 255–263 (2019). https://doi.org/10.4274/jtgga.galenos.2019.2018.0142
J.S. Archer, R.J. Chang, Hirsutism and acne in polycystic ovary syndrome. Best Pract. Res. Clin. Obstet. Gynaecol. 18(5), 737–754 (2004). https://doi.org/10.1016/j.bpobgyn.2004.05.007
L. Fatsetti, A. Gambera, S. Andrico, E. Sartori, Acne and hirsutism in polycystic ovary syndrome: clinical, endocrine-metabolic and ultrasonographic differences. Gynecol. Endocrinol. 16(4), 275–284 (2002). https://doi.org/10.1080/gye.16.4.275.284
Acknowledgements
We thank all the study subjects. We are also thankful to Suhail Murtaza, technical assistant, who assisted in sampling of PCOS cases and control women. We are thankful to co-scholars for their help during the course of experimental studies.
Funding
This work was supported by the Indian Council of Medical Research under grant No. RBMH/FW/2019/3.
Author information
Authors and Affiliations
Contributions
M.N.: major work done, manuscript writing, project development, recruitment of participants, sample collection, data collection. S.M.A: primer designing and qPCR validation. S.U.A.R.: data collection, Data analysis, manuscript revision. S.A.: recruitment of participants, manuscript revision, data collection. I.M.: diagnosis and recruitment of cases. S.A.: conception and design, manuscript writing, project development and management. All authors read and approved the final draft.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent of participation
All participants were recruited after written informed consent was obtained from them.
Consent for publication
All authors have approved the manuscript for submission.
Ethics approval
This study was approved by the ethical committee Government Medical College Srinagar under ethical approval no. 94/ETH/GMC/ICMR.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nabi, M., Andrabi, S.M., Rasool, S.U.A. et al. Androgen receptor coregulator long noncoding RNA CTBP1-AS is associated with polycystic ovary syndrome in Kashmiri women. Endocrine 75, 614–622 (2022). https://doi.org/10.1007/s12020-021-02894-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02894-9



