Abstract
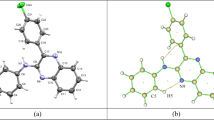
X-ray diffraction studies were carried out on single crystals of two flavonoids, viz. 5-hydroxy-6,7,4′-trimethoxyflavone, C18H16O6, (I) and 5-hydroxy-3,7,4′-trimethoxyflavone, C18H16O6, (II). Crystal structures of both the flavonoids were solved by direct methods and refined by full-matrix least-squares procedures. In both the molecules, the benzopyran moiety is planar. The dihedral angle between the phenyl ring and the benzopyran portion is 5.50(4)° in (I) and 29.11(5)° in (II). In (I), the crystal packing is influenced by O-H…O hydrogen bonds, and weak C-H…O and π…π interactions whereas in (II) the crystal structure is stabilized by the presence of four intermolecular short contacts of the type C-H…O. There is also one C-H…π hydrogen bond with H… centroid distance of <2.7 Å. The molecules are further stabilized by π-π interactions.
Similar content being viewed by others
References
Allen F H 2002 Acta Crystallogr. B58 380
Allen F H, Kennard O, Watson D G, Brammer L, Orpen A G and Taylor R 1987 J. Chem. Soc. Perkin Trans-II S1–S19
Amabilino D B and Stoddart J F 1995 Chem. Rev. 95 2725
Asolkar L V, Kakkar K K and Chakre O J 1992 Second supplement to glossary of Indian medicinal plants with active principles (New Delhi: CSIR) Part 1, p. 195
Brahmachari G 2005 Private communication, Visva Bharati University, Santiniketan
Brahmachari G and Gorai D 2006 Curr. Org. Chem. 10 873
Brahmachari G and Gorai D 2007 Recent developments in the research of naturally occurring flavonoids: An overview, in Chemistry of natural products: Recent trends and developments (ed.) G Brahmachari (Trivandrum: Research Signpost) Ch. 4
Brown S P, Schnell I, Brand J D, Mullen K and Spiess H 1999 J. Am. Chem. Soc. 121 6712
Chipot C, Jaffe R, Maigret B, Pearlman D A and Kollman P A 1996 J. Am. Chem. Soc. 118 11217
Claessens C G and Stoddart J F 1997 J. Phys. Org. Chem. 10 254
Desiraju G R 1992 Mol. Cryst. Liq. Cryst. 211 63
Desiraju G R 1996 Acc. Chem. Res. 29 441
Desiraju G R and Steiner T 1999 The weak hydrogen bond in structural chemistry and biology (Oxford: Oxford University Press)
Farrugia L J 1997 J. Appl. Cryst. 30 565
Farrugia L J 1999 J. Appl. Cryst. 32 837
Hannon M J, Painting C L and Alcock N W 1999 Chem. Commun. 2023
Hirsch K A, Wilson S R and Moore J S 1997 Chem. Eur. J. 3 765
Hunter C A 1993 Angew. Chem.: Int. Ed. Engl. 32 1653
Hunter C A 1994 Chem. Soc. Rev. 101
Janiak C 2000 J. Chem Soc. Dalton Trans. 3885
Janiak C, Temizdemir S and Dechert S 2000 Inorg. Chem. Commun. 3 271
Kapil V, Sinha A K and Sinha G K 1983 Bull. Med. Ethnobot. Res. 4 124
Lightfoot M P, Mair F S, Pritchard R G and Warren J W 1999 Chem. Commun. 1945
Malecka M, Massa W and Budzisz E 2004 Acta Crystallogr. C60 762
Marek J, Vesela D, Liskova M and Zemlicka M 2003 Acta Crystallogr. C59 127
Nardelli M 1995 J. Appl. Cryst. 28 659
Ning G L, Wu L P, Sugimoto K, Munakata M, Kuroda-Sowa T and Maekawa M 1999 J. Chem. Soc. Dalton Trans. 2529
Nishio M and Hirota M 1989 Tetrahedron 45 7201
Nishio M, Umezawa Y, Hirota M and Takeuchi Y 1995 Tetrahedron 51 8665
Nishio M, Hirota M and Umezawa Y 1998 The C-H … π interactions (Evidence, nature and consequences) (New York: Wiley-VCH)
North A C T, Phillips D C and Mathews F S 1968 Acta Crystallogr. A24 351
Ryparczyl-Pirek A J and Nawrot-Modranka J 2004 Acta Crystallogr. E60 988
Seetharaman J and Rajan S S 1992 Acta Crystallogr. C48 1714
Sheldrick G M 1997 SHELXS97 and SHELXL97 (Germany: University of Gottingen)
Shoja M 1989 Acta Crystallogr. C45 828
Steiner T 1996 Cryst. Rev. 6 1
Takahashi H, Tsuboyama S, Umezawa Y, Honda K and Nishio M 2000 Tetrahedron 56 6185
Thinagar S, Velmurugan D, Shanmuga Sundara Raj S, Fun H-K, Gupta S C, Merazig H and Bouacida S 2003 Acta Crystallogr. C59 181
Ting H and Watson W H 1972 Acta Crystallogr. B28 1046
Wahl M C and Sundaralingam M 1997 Biochem. Sci. 22 97
Wallet J C, Gaydou E M, Tinant B, Declercq J-P, Baldy A and Bonifassi P 1990 Acta Crystallogr. C46 1131
Wallet J C, Gaydou E M, Espinosa E, Osorno O, Molins E and Miravitlles C 1992 Acta Crystallogr. C48 86
Wallet J C, Wojtczak A, Cody V, Galitsky N and Gaydou E M 1993 Acta Crystallogr. C49 357
Wealth of India 1962 Raw materials (New Delhi: CSIR) p. 116
Author information
Authors and Affiliations
Corresponding author
Additional information
CCDC-602176 and CCDC-610487 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Rights and permissions
About this article
Cite this article
Sharma, D., Gupta, V.K., Brahmachari, G. et al. X-ray study of weak interactions in two flavonoids. Bull Mater Sci 30, 469–475 (2007). https://doi.org/10.1007/s12034-007-0074-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-007-0074-x