Abstract
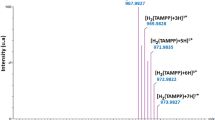
A new mixed-ligand binuclear copper(II) complex [Cu(MS)(bpy)]2.(ClO4)2, built of 5-methylsalicylaldehyde and 2,2′-bipyridyl has been synthesized and characterized by using elemental analysis, IR and UV-Vis spectroscopy. Crystal structure of the complex shows that copper(II) ion lies in a square pyramidal coordination environment. The structure consists of two symmetrical half units in which the copper(II) ion of one half unit connected with the phenolate oxygen atom of other half unit along with one perchlorate anion in the crystal lattice as free molecule. Presence of uncoordinated perchlorate anion was also confirmed by IR spectroscopy. Absorption spectroscopy exhibits d-d transition at 628 nm, which further supports the square pyramidal geometry around the copper(II) ions. EPR spectrum of the copper(II) complex at room temperature shows a broad signal without any splitting pattern at g || = 2.26,g ⊥ = 2.03 and the magnetic moment (μ eff= 1.31 BM) obtained at room temperature indicate an antiferromagnetic interaction between the two copper(II) ions through phenoxide-bridge. Binding studies reveal that the complex possesses good binding propensity (K b = 5.2 ± 1.7 × 104 M−1) and bind to nitrogenous bases of DNA through intercalation. Nuclease activity of the complex with pBR322 DNA shows that the effect of hydrolytic cleavage is dose-dependent and the oxidative cleavage indicates the involvement of hydroxyl radical and singlet-oxygen as reactive oxygen species.

Mixed-ligand binuclear copper(II) complex was isolated and characterized. The binding studies of the complex suggest the intercalation mode of binding with CTDNA. The nuclease activity with pBR322 DNA shows that the hydrolytic cleavage is dose-dependent and the oxidative mechanism implies the involvement of hydroxyl radical and singlet-oxygen as reactive oxygen species.






Similar content being viewed by others
References
Barve A, Kumbhar A, Bhat M, Joshi B, Butcher R, Sonawane U and Joshi R 2009 Inorg. Chem. 48 9120
Hui J K H, Yu Z and MacLachlan M J 2007 Angew. Chem. Int. Ed. 119 8126
Loganathan R, Ramakrishnan S, Suresh E, Riyasdeen A, Akbarsha M A and Palaniandavar M 2012 Inorg. Chem. 51 5512
Ramakrishnan S and Palaniandavar M 2005 J. Chem. Sci. 117 179
Roy M, Pathak B, Patra A K, Jemmis E D, Nethaji M and Chakravarty A R 2007 Inorg. Chem. 46 11122
Kumar A, Mitra A, Ajay A K, Bhat M K and Rao C P 2012 J. Chem. Sci. 124 1217
Ng C H, Kong K C, Von S T, Balraj P, Jensen P, Thirthagiri E, Hamada H and Chikira M 2008 Dalton Trans. 447
Ramakrishnan S, Rajendiran V, Palaniandavar M, Periasamy V S, Srinag B S, Krishnamurthy H and Akbarsha M A 2009 Inorg. Chem. 48 1309
Sammes P G and Yahioglu G 1994 Chem. Soc. Rev. 23 327
Chen G J, Qiao X, Qiao P Q, Xu G J, Xu J Y, Tian J L, Gu W, Liu X and Yan S P 2011 J. Inorg. Biochem. 105 119
Bruker-Nonius 2004APEX-II and SAINT-plus (Version 7.06a), Bruker AXS Inc., Madison, Wisconsin, USA
Altomare A, Cascarano G, Giacovazzo C and Guagliardi A 1993 J. Appl. Crystallogr. 26 343
Sheldrick G M 2008 Acta Crystallogr. A 64 112
Farrugia L J 1997 J. Appl. Crystallogr. 30 565
Macrae C F, Edgington P R, McCabe P, Pidcock E, Shields G P, Taylor R, Towler M and van de Streek J 2006 J. Appl. Crystallogr. 39 453
Marmur J 1961 J. Mol. Biol. 3 208
Reichmann M E, Rice S A, Thomas C A and Doty P 1954 J. Am. Chem. Soc. 76 3047
Carter M T, Rodriguez M and Bard A J 1989 J. Am. Chem. Soc. 111 8901
Smith S R, Neyhart G A, Kalsbeck W A and Thorp H H 1994 New J. Chem. 18 397
Friedman A E, Chambron J C, Sauvage J P, Turro N J and Barton J K 1990 J. Am. Chem. Soc. 112 4960
Cohen G and Eisenberg H 1969 Biopolymers 8 45
Satyanarayana S, Dabrowiak J C and Chaires J B 1992 Biochemistry 31 9319
Addison A W, Rao T N, Reedijk J, van Rijn J and Verschoor G C 1984 J. Chem. Soc. Dalton Trans. 1349
Dhara P K, Pramanik S, Lu T H, Drew M G B and Chattopadhyay P 2004 Polyhedron 23 2457
Weyhermüller T, Wagner R and Chaudhuri P 2011 Eur. J. Inorg. Chem. 2011 2547
Fei B L, Li W, Xu W S, Li Y G, Long J Y, Liu Q B, Shao K Z, Su Z M and Sun W Y 2013 J. Photochem. Photobiol. B 125 32
Desiraju G R and Steiner T 1999 The weak hydrogen bond in structural chemistry and biology. Oxford University PressNew York
Bharathi K S, Sreedaran S, Rahiman A K and Narayanan V 2013 Spectrochim. Acta A 105 245
Geary W J 1971 Coord. Chem. Rev. 7 81
Barefield E K, Freeman G M and Derveer D G V 1986 Inorg. Chem. 25 552
Connick P A and Macor K A 1991 Inorg. Chem. 30 4654
Benzekri A, Dubourdeaux P, Latour J M, Rey P and Laugier J 1991J. Chem. Soc. Dalton. Trans. 3359
Tolman W B, Rardin R L and Lippard S J 1989 J. Am. Chem. Soc. 111 4532
Guzar S H and Qin-han J I N 2008 J. Appl. Sci. 8 2480
Dougherty G and Pigram W J 1982 Crit. Rev. Biochem. 12 103
Bejune S A, Shelton A H and McMillin D R 2003 Inorg. Chem. 42 8465
Raja D S, Bhuvanesh N S P and Natarajan K 2011 Eur. J. Med. Chem. 46 4584
Cory M, Mckee D D, Kagan J, Henry D W and Miller J A 1985 J. Am. Chem. Soc. 107 2528
Fin L and Yang P 1997 J. Inorg. Biochem. 68 79
Gabbay E J, Scofield R E and Baxter C S 1973 J. Am. Chem. Soc. 95 7850
Ramakrishnan S, Shakthipriya D, Suresh E, Periasamy V S, Akbarsha M A and Palaniandavar M 2011 Inorg. Chem. 50 6458
Anbu S, Kandaswamy M, Kamalraj S, Muthumarry J and Varghese B 2011 Dalton Trans. 40 7310
Acknowledgements
The authors thank Sophisticated Analytical Instruments Facility (SAIF), Indian Institute Technology Madras (IIT-M), Chennai 600 025, for solving the crystal structure of the complex.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information
CCDC-924217 contains supplementary crystallographic data of this article. These data files can be obtained free of charge from The Cambridge Crystallographic Data Centre via http://www.ccdc.cam.ac.uk/data_request/cif). Figures , and show the packing diagram, cyclic voltammogram and T4 ligation experiment of the complex, respectively. For details, see www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
GURUMOORTHY, P., RAVICHANDRAN, J. & RAHIMAN, A.K. Mixed-ligand binuclear copper(II) complex of 5-methylsalicylaldehyde and 2,2′-bipyridyl: Synthesis, crystal structure, DNA binding and nuclease activity. J Chem Sci 126, 783–792 (2014). https://doi.org/10.1007/s12039-014-0607-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-014-0607-y