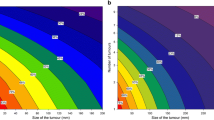
Abstract
Background
Evidence-based decision-making is critical to optimize the benefits and mitigate futility associated with surgery for patients with malignancies. Untreated hepatocellular carcinoma (HCC) has a median survival of only 6 months. The objective was to develop and validate an individualized patient-specific tool to predict preoperatively the benefit of surgery to provide a survival benefit of at least 6 months following resection.
Methods
Using an international multicenter database, patients who underwent curative-intent liver resection for HCC from 2008 to 2017 were identified. Using random assignment, two-thirds of patients were assigned to a training cohort with the remaining one-third assigned to the validation cohort. Independent predictors of postoperative death within 6 months after surgery for HCC were identified and used to construct a nomogram model with a corresponding online calculator. The predictive accuracy of the calculator was assessed using C-index and calibration curves.
Results
Independent factors associated with death within 6 months of surgery included age, Child–Pugh grading, portal hypertension, alpha-fetoprotein level, tumor rupture, tumor size, tumor number and gross vascular invasion. A nomogram that incorporated these factors demonstrated excellent calibration and good performance in both the training and validation cohorts (C-indexes: 0.802 and 0.798). The nomogram also performed better than four other commonly-used HCC staging systems (C-indexes: 0.800 vs. 0.542–0.748).
Conclusions
An easy-to-use online prediction calculator was able to identify patients at highest risk of death within 6 months of surgery for HCC. The proposed online calculator may help guide surgical decision-making to avoid futile surgery for patients with HCC.




Similar content being viewed by others
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- PVTT:
-
Portal vein tumor thrombus
- HVTT:
-
Hepatic vein tumor thrombus
- PVE:
-
Portal vein embolization
- ALPPS:
-
Associating liver partition and portal vein ligation for staged hepatectomy
- TACE:
-
Transcatheter arterial chemoembolization
- BSC:
-
Best supportive care
- RCT:
-
Randomized controlled trial
- ECOG:
-
Eastern Cooperative Oncology Group
- ASA:
-
American society of anesthesiologists
- HBV:
-
Hepatitis B virus
- HCV:
-
Hepatitis C virus
- INR:
-
International normalized ratio
- TB:
-
Total bilirubin
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate transaminase
- AFP:
-
Alpha-fetoprotein
- CT:
-
Computerized tomography
- MRI:
-
Magnetic resonance imaging
- ROC:
-
Receiver operating characteristic
- TNM:
-
Tumor-node-metastasis
- CLIP:
-
Cancer of the liver Italian program
- BCLC:
-
Barcelona clinic liver cancer
- DCA:
-
Decision curve analysis
References
Wilson A, Ronnekleiv-Kelly SM, Pawlik TM. Regret in surgical decision making: a systematic review of patient and physician perspectives. World J Surg 2017;41:1454–1465
Robson N, Rew D. Collective wisdom and decision making in surgical oncology. Eur J Surg Oncol 2010;36:230–236
Lamb B, Green JS, Vincent C, et al. Decision making in surgical oncology. Surg Oncol 2011;20:163–8
Yang JD, Roberts LR. Hepatocellular carcinoma: a global view. Nat Rev Gastroenterol Hepatol 2010;7:448–458
Nagasue N, Yukaya H, Hamada T, et al. The natural history of hepatocellular carcinoma. A study of 100 untreated cases. Cancer 1984;54:1461–1465
Okuda K, Ohtsuki T, Obata H, et al. Natural history of hepatocellular carcinoma and prognosis in relation to treatment. Study of 850 patients. Cancer 1985;56:918–928
Cheng AL, Kang YK, Chen Z, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25–34
Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008;359:378–390
Knox JJ, Cleary SP, Dawson LA. Localized and systemic approaches to treating hepatocellular carcinoma. J Clin Oncol 2015;33:1835–1844
Kulik L, El-Serag HB. Epidemiology and management of hepatocellular carcinoma. Gastroenterology 2019;156(477–491):e1
Rahbari NN, Mehrabi A, Mollberg NM, et al. Hepatocellular carcinoma: current management and perspectives for the future. Ann Surg 2011;253:453–469
Song TJ, Ip EW, Fong Y. Hepatocellular carcinoma: current surgical management. Gastroenterology 2004;127:S248-S260
Liu J, Zhang H, Xia Y, et al. Impact of clinically significant portal hypertension on outcomes after partial hepatectomy for hepatocellular carcinoma: a systematic review and meta-analysis. HPB (Oxford) 2019;21:1–13
Berardi G, Morise Z, Sposito C, et al. Development of a nomogram to predict outcome after liver resection for hepatocellular carcinoma in Child-Pugh B cirrhosis. J Hepatol 2020;72:75–84
Vitale A, Burra P, Frigo AC, et al. Survival benefit of liver resection for patients with hepatocellular carcinoma across different Barcelona Clinic Liver Cancer stages: a multicentre study. J Hepatol 2015;62:617–624
Kokudo T, Hasegawa K, Yamamoto S, et al. Surgical treatment of hepatocellular carcinoma associated with hepatic vein tumor thrombosis. J Hepatol. 2014;61:583–588
Pawlik TM, Poon RT, Abdalla EK, et al. Hepatectomy for hepatocellular carcinoma with major portal or hepatic vein invasion: results of a multicenter study. Surgery 2005;137:403–10
Kokudo T, Hasegawa K, Matsuyama Y, et al. Survival benefit of liver resection for hepatocellular carcinoma associated with portal vein invasion. J Hepatol 2016;65:938–943
Dhir M, Melin AA, Douaiher J, et al. A review and update of treatment options and controversies in the management of hepatocellular carcinoma. Ann Surg 2016;263:1112–1125
Yang T, Lu JH, Lau WY, et al. Perioperative blood transfusion does not influence recurrence-free and overall survivals after curative resection for hepatocellular carcinoma: a propensity score matching analysis. J Hepatol 2016;64:583–593
Xu XF, Xing H, Han J, et al. Risk factors, patterns, and outcomes of late recurrence after liver resection for hepatocellular carcinoma: a multicenter study from China. JAMA Surg 2019;154:209–217
Yang T, Liu K, Liu CF, et al. Impact of postoperative infective complications on long-term survival after liver resection for hepatocellular carcinoma. Br J Surg. 2019;106:1228–1236
Wang MD, Li C, Li J, et al. Long-term survival outcomes after liver resection for binodular hepatocellular carcinoma: a multicenter cohort study. Oncologist 2019;24:e730–e739
Youden WJ. Index for rating diagnostic tests. Cancer. 1950;3:32–35
Tellapuri S, Sutphin PD, Beg MS, et al. Staging systems of hepatocellular carcinoma: a review. Indian J Gastroenterol 2018;37:481–491
Fitzgerald M, Saville BR, Lewis RJ. Decision curve analysis. JAMA 2015;313:409–410
Kerr KF, Brown MD, Zhu K, et al. Assessing the clinical impact of risk prediction models with decision curves: guidance for correct interpretation and appropriate use. J Clin Oncol 2016;34:2534–40
Dasari B, Hodson J, Sutcliffe RP, et al. Developing and validating a preoperative risk score to predict 90-day mortality after liver resection. J Surg Oncol 2019;119:472–478
Shim JH, Jun MJ, Han S, et al. Prognostic nomograms for prediction of recurrence and survival after curative liver resection for hepatocellular carcinoma. Ann Surg 2015;261:939–946
Yang P, Qiu J, Li J, et al. Nomograms for Pre- and Postoperative Prediction of Long-term Survival for Patients Who Underwent Hepatectomy for Multiple Hepatocellular Carcinomas. Ann Surg 2016;263:778–786
Rebouissou S, Nault JC. Advances in molecular classification and precision oncology in hepatocellular carcinoma. J Hepatol 2020;72:215–229
Qin S, Ren Z, Meng Z, et al. Camrelizumab in patients with previously treated advanced hepatocellular carcinoma: a multicentre, open-label, parallel-group, randomised, phase 2 trial. Lancet Oncol 2020
Lai EC, Lau WY. Spontaneous rupture of hepatocellular carcinoma: a systematic review. Arch Surg 2006;141:191–8
Yang T, Sun YF, Zhang J, et al. Partial hepatectomy for ruptured hepatocellular carcinoma. Br J Surg 2013;100:1071–1079
Moris D, Chakedis J, Sun SH, et al. Management, outcomes, and prognostic factors of ruptured hepatocellular carcinoma: a systematic review. J Surg Oncol 2018;117:341–353
Glantzounis GK, Tokidis E, Basourakos SP, et al. The role of portal vein embolization in the surgical management of primary hepatobiliary cancers. A systematic review. Eur J Surg Oncol 2017;43:32–41
Song T. Recent advances in surgical treatment of hepatocellular carcinoma. Drug Discov Ther. 2015;9:319–330
Raptis DA, Linecker M, Kambakamba P, et al. Defining benchmark outcomes for ALPPS. Ann Surg 2019;270:835–841
Olthof PB, Schnitzbauer AA, Schadde E. The HPB controversy of the decade: 2007–2017 - ten years of ALPPS. Eur J Surg Oncol 2018;44:1624–1627
Kokudo T, Hasegawa K, Matsuyama Y, et al. Liver resection for hepatocellular carcinoma associated with hepatic vein invasion: a Japanese nationwide survey. Hepatology. 2017;66:510–517
Liang L, Chen TH, Li C, et al. A systematic review comparing outcomes of surgical resection and non-surgical treatments for patients with hepatocellular carcinoma and portal vein tumor thrombus. HPB (Oxford) 2018;20:1119–1129
Grant SB, Modi PK, Singer EA. Futility and the care of surgical patients: ethical dilemmas. World J Surg. 2014;38:1631–1637
Morris AM. Rational vs. intuitive judgment in surgical decision making. Ann Surg 2016;264:887–888
Funding
This work was supported by the National Natural Science Foundation of China (No. 81672699 and 81972726). The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
Drs L. Liang, Quan, Wu, Diao and Li contributed equally to this work. Drs Huang, Shen and Yang had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: L. Liang, Quan, Lau, Huang, Shen, Yang. Administrative support: Shen, Zhang, Huang, Yang. Provision of study materials or patients: Lau, Pawlik, C.W. Zhang, Huang, Shen, Yang. Collection and assembly of data: L. Liang, Wu, Diao, Jia, Li, Chen, Zhang, Zhou, Zhang, Wang, Serenari, Cescon, Schwartz, Zeng, Liang, Xing, Li, Wang, Chen. Data analysis and interpretation: L. Liang, Quan, Wu, Diao, Jia, Xing, Yang. Manuscript writing: All authors. Final approval of manuscript: All authors. Obtained funding: Yang.
Corresponding authors
Ethics declarations
Conflict of interest
None of the authors report any conflicts of interest.
Ethical approval
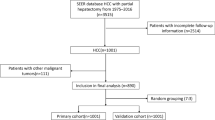
Informed consent was obtained from all patients before Flow chart of patient inclusion and the study was approved by the Institutional Review Board or Clinical Analyses at each of these hospitals. The study was performed in accordance with the Declaration of Helsinki and the Ethical Guidelines for Clinical Studies in all participating hospitals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liang, L., Quan, B., Wu, H. et al. Development and validation of an individualized prediction calculator of postoperative mortality within 6 months after surgical resection for hepatocellular carcinoma: an international multicenter study. Hepatol Int 15, 459–471 (2021). https://doi.org/10.1007/s12072-021-10140-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-021-10140-7