Abstract
Background
Advanced non-small cell lung cancer (NSCLC) has remained challenging to treat effectively. This study aimed to investigate the clinical effects and safety of immunotherapy with dendritic cells and cytokine-induced killer cells (DC-CIK) administered with chemotherapy (CT) in this malignancy.
Methods
We have developed a new clinical trial design termed as the prospective patient’s preference-based study (PPPS). Consecutive patients (n = 135) with advanced NSCLC were treated with DC-CIK administered with CT or mono-therapy (CT or DC-CIK alone).
Results
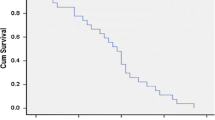
For all the patients, the median PFS was 5.7 months and the median OS was 17.5 months. The 1-year PFS and OS rates were 29.4% and 58.2%, respectively. The 1-year PFS and OS rates for DC-CIK plus CT were significantly higher than that in the group of patients who received DC-CIK alone and CT alone (P < 0.05). The number of adoptively infused DC-CIK cells was associated with clinical efficacy. After adjusting for competing risk factors, DC-CIK combined with CT and infused number of CIKs remained independent predictors of PFS and OS. Phenotypic analysis of peripheral blood mononuclear cells showed that CD8+CD28+, and CD8+CD28− T cells, changed significantly in all groups (P < 0.01). The CD3+ T cells increased in the chemotherapy plus immunotherapy and the immunotherapy alone group (P < 0.01), while CD3−CD16+CD56 T cells decreased in the chemotherapy plus immunotherapy and the immunotherapy alone group (P < 0.01).
Conclusions
DC-CIK combined with chemotherapy administration resulted in numerically superior PFS and OS compared with monotherapy in advanced NSCLC.


Similar content being viewed by others
References
Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Juergens R, Brahmer J. Targeting the epidermal growth factor receptor in non-small-cell lung cancer: who, which, when, and how? Curr Oncol Rep. 2007;9:255–64.
Choudhury A, Palma M, Mellstedt H. The future of cancer vaccines for non-small-cell lung cancer: ongoing trials. Clin Lung Cancer. 2008;9(Suppl 1):S37–44.
Till BG, Jensen MC, Wang J, et al. Adoptive immunotherapy for indolent non-Hodgkin lymphoma and mantle cell lymphoma using genetically modified autologous CD20-specific T cells. Blood. 2008;112:2261–71.
Rizvi NA, Hellmann MD, Snyder A, et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 2015;348:124–8.
Li W, Xu LP, Wang L, et al. Cytokine-induced killer cell therapy for advanced pancreatic adenocarcinoma: a case report and review of the literature. Oncol Lett. 2013;5:1427–9.
Chung MJ, Park JY, Bang S, et al. Phase II clinical trial of ex vivo-expanded cytokine-induced killer cells therapy in advanced pancreatic cancer. Cancer Immunol Immunother. 2014;63:939–46.
Chen R, Deng X, Wu H, et al. Combined immunotherapy with dendritic cells and cytokine-induced killer cells for malignant tumors: a systematic review and meta-analysis. Int Immunopharmacol. 2014;22:451–64.
Hontscha C, Borck Y, Zhou H, et al. Clinical trials on CIK cells: first report of the international registry on CIK cells (IRCC). J Cancer Res Clin Oncol. 2011;137:305–10.
Schmidt-Wolf IG, Negrin RS, Kiem HP, et al. Use of a SCID mouse/human lymphoma model to evaluate cytokine-induced killer cells with potent antitumor cell activity. J Exp Med. 1991;174:139–49.
Zhou C, Liu D, Li J, et al. Chemotherapy plus dendritic cells co-cultured with cytokine-induced killer cells versus chemotherapy alone to treat advanced non-small-cell lung cancer: a meta-analysis. Oncotarget. 2016;7:86500–10.
Cui J, Li L, Wang C, et al. Combined cellular immunotherapy and chemotherapy improves clinical outcome in patients with gastric carcinoma. Cytotherapy. 2015;17:979–88.
Zhu Y, Zhang H, Li Y, et al. Efficacy of postoperative adjuvant transfusion of cytokine-induced killer cells combined with chemotherapy in patients with colorectal cancer. Cancer Immunol Immunother. 2013;62:1629–35.
Ren J, Di L, Song G, et al. Selections of appropriate regimen of high-dose chemotherapy combined with adoptive cellular therapy with dendritic and cytokine-induced killer cells improved progression-free and overall survival in patients with metastatic breast cancer: reargument of such contentious therapeutic preferences. Clin Transl Oncol. 2013;15:780–8.
Kane HL, Halpern MT, Squiers LB, et al. Implementing and evaluating shared decision making in oncology practice. CA Cancer J Clin. 2014;64:377–88.
Stiggelbout AM, Pieterse AH, De Haes JC. Shared decision making: concepts, evidence, and practice. Patient Educ Couns. 2015;98:1172–9.
Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–55.
Kaplan EL, Meier P. Nonparametric estimations from incomplete observations. J Am Stat Assoc. 1958;53:457–81.
Dr C. Regression models and life-tables. J R Stat Soc B. 1972;34:187–220.
Bayman NA, Blackhall F, Jain P, et al. Management of unresectable stage III non-small-cell lung cancer with combined-modality therapy: a review of the current literature and recommendations for treatment. Clin Lung Cancer. 2008;9:92–101.
Wu C, Jiang J, Shi L, Xu N. Prospective study of chemotherapy in combination with cytokine-induced killer cells in patients suffering from advanced non-small cell lung cancer. Anticancer Res. 2008;28:3997–4002.
Kong DS, Nam DH, Kang SH, et al. Phase III randomized trial of autologous cytokine-induced killer cell immunotherapy for newly diagnosed glioblastoma in Korea. Oncotarget. 2017;8:7003–13.
Wang M, Cao JX, Pan JH, et al. Adoptive immunotherapy of cytokine-induced killer cell therapy in the treatment of non-small cell lung cancer. PLoS One. 2014;9:e112662.
Tiwari M. From tumor immunology to cancer immunotherapy: miles to go. J Cancer Res Ther. 2010;6:427–31.
Giannoni P, Cutrona G, de Totero D. Survival and immunosuppression induced by hepatocyte growth factor in chronic lymphocytic leukemia. Curr Mol Med. 2017;17:24–33.
Kim R, Emi M, Tanabe K, Arihiro K. Tumor-driven evolution of immunosuppressive networks during malignant progression. Cancer Res. 2006;66:5527–36.
Sangiolo D. Cytokine induced killer cells as promising immunotherapy for solid tumors. J Cancer. 2011;2:363–8.
Zou W. Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat Rev Cancer. 2005;5:263–74.
Kosmaczewska A, Bocko D, Ciszak L, et al. Dysregulated expression of both the costimulatory CD28 and inhibitory CTLA-4 molecules in PB T cells of advanced cervical cancer patients suggests systemic immunosuppression related to disease progression. Pathol Oncol Res. 2012;18:479–89.
Song G, Wang X, Jia J, et al. Elevated level of peripheral CD8(+)CD28(−) T lymphocytes are an independent predictor of progression-free survival in patients with metastatic breast cancer during the course of chemotherapy. Cancer Immunol Immunother. 2013;62:1123–30.
Zhao YJ, Jiang N, Song QK, et al. Continuous DC-CIK infusions restore CD8+ cellular immunity, physical activity and improve clinical efficacy in advanced cancer patients unresponsive to conventional treatments. Asian Pac J Cancer Prev. 2015;16:2419–23.
Okita R, Saeki T, Takashima S, et al. CD4+CD25+ regulatory T cells in the peripheral blood of patients with breast cancer and non-small cell lung cancer. Oncol Rep. 2005;14:1269–73.
Caserta S, Alessi P, Guarnerio J, et al. Synthetic CD4+ T cell-targeted antigen-presenting cells elicit protective antitumor responses. Cancer Res. 2008;68:3010–8.
Wang X, Ren J, Zhang J, et al. Prospective study of cyclophosphamide, thiotepa, carboplatin combined with adoptive DC-CIK followed by metronomic cyclophosphamide therapy as salvage treatment for triple negative metastatic breast cancers patients (aged < 45). Clin Transl Oncol. 2016;18:82–7.
Jiang N, Qiao G, Wang X, et al. Dendritic cell/cytokine-induced killer cell immunotherapy combined with S-1 in patients with advanced pancreatic cancer: a prospective study. Clin Cancer Res. 2017;23:5066–73.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors who have taken part in this study declared that they have nothing to disclose regarding funding or conflict of interest with respect to this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the ethics committee of Capital Medical University.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Zhao, Y., Qiao, G., Wang, X. et al. Combination of DC/CIK adoptive T cell immunotherapy with chemotherapy in advanced non-small-cell lung cancer (NSCLC) patients: a prospective patients’ preference-based study (PPPS). Clin Transl Oncol 21, 721–728 (2019). https://doi.org/10.1007/s12094-018-1968-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-018-1968-3