Abstract
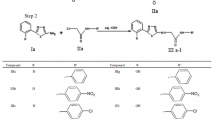
A novel series of thiazolo[3,2-a]benzimidazole derivatives containing benzofuran nucleus (5a–l) have been synthesized. The key intermediate, substituted benzimidazol-sulfanyl benzofuran ethanone (3a–d) was prepared by refluxing the mixture of substituted 2-acetyl benzofuran and substituted 2-mercaptobenzimidazole in acetic acid. The cyclisation of compounds (3a–d) using polyphosphoric acid furnished the corresponding 6-substituted benzofuran thiazolo[3,2-a]benzimidazoles (4a–d). Further, the cyclized compounds (4a–d) were subjected for Mannich reaction to give corresponding Mannich bases (5a–l). All newly synthesized compounds were screened for antifungal and anthelmintic activity. Amongst the tested compounds, 4b and 4d exhibited potential antifungal activity. From the anthelmintic activity data, it was found that the compounds 3a, 3b and 5i were found to be more effective against the tested earthworm Pheretima posthuma. In correlation to anthelmintic activity, the selected compounds were subjected for molecular docking studies and the compounds 3a and 5i have emerged as active anthelmintic agents with maximum binding affinity (−3.7 and −5.4 kcal/mol).



Similar content being viewed by others
References
Gil C, Brase S (2009) Solid-phase synthesis of biologically active benzoannelated nitrogen heterocycles-an update. J Comb Chem 11:174–197
Butler MS (2004) The role of natural product chemistry in drug discovery. J Nat Prod 67:2141–2145
Ravindernath A, Reddy MS (2013) Synthesis and evaluation of anti-inflammatory, antioxidant and antimicrobial activities of densely functionalized novel benzo [d] imidazolyl Tetrahydropyridine carboxylates. doi:10.1016/j.arabjc.2013.02.011
Premakumari C, Muralikrishna A, Padmaja A, Padmavathi V, Park SJ, Kim TJ, Reddy GD (2014) Synthesis, antimicrobial and anticancer activities of amido sulfonamido methane linked bis heterocycles. Ara J Chem 7:385–395
Gellis A, Kovacic H, Boufatah N, Vanelle P (2008) Synthesis and cytotoxicity evaluation of some benzimidazole-4,7-diones as bioreductive anticancer agents. Eur J Med Chem 43:1858–1864
Borgers M, De Nollin S (1975) Ultra structural changes in Ascarissuum intestine after mebendazole treatment in vivo. J Parasitol 61:110–122
Lacey E (1990) Mode of action of benzimidazoles. Parasitol Today 6:112–115
Katiyar SK, Gordon VR, McLaughlin GL, Edlind TD (1994) Antiprotozoal activities of benzimidazoles and correlations with beta-tubulin sequence. Antimicrob Agents Chemother 38(9):2086–2090
Dustin P (1984) Microtubules. Springer, Heidelberg Germany, p. 482
Mavrova ATS, Anichina KK, Vuchev DI, Denkova JATS, Kondeva MS, Micheva MK (2006) Antihelminthic activity of some newly synthesized5 (6)-(un)substituted-1Hbenzimidazol-2-ylthioacetylpiperazine derivatives. E J Med Chem 41:1412–1420
Aryapour H, Riazi GH, Foroumadi A, Ahmadian S, Shafiee A, Karima O, Mahdavi M, Emami S, Sorkhi M, Khodadady S (2011) Biological evaluation of synthetic analogues of curcumin: chloro-substituted-20-hydroxychalcones as potential inhibitors of tubulin polymerization and cell proliferation. Med Chem Res 20:503–510
Satyendra RV, Vishnumurthy KA, Vagdevi HM, Rajesh KP, Manjunatha H, Shruthi A (2011) Synthesis, in vitro antioxidant, anthelmintic and molecular docking studies of novel dichloro substituted benzoxazole-triazolo-thione derivatives. Eur J Med Chem 46:3078–3084
Townsend LB, Wise DS (1990) The synthesis and chemistry of certain anthelmintic benzimidazoles. Parasit Today 6(4):107–112
Chua T, Moore CL, Perri MB, Donabedian SM, Masch W, Vager D, Davis SL, Lulek K, Zimnicki B, Zervos MJ (2008) Molecular epidemiology of methicillin resistant Staphylococcus aureus bloodstream isolates in urban Detroit. J Clin Microbiol 46:2345–2352
Chu DTW, Plattner JJ, Katz L (1996) New directions in antibacterial research. J Med Chem:3853–3874
Hollomon DW, Butters JA, Barker Z, Hall L (1998) Fungal β-tubulin, expressed as a fusion protein, binds benzimidazole and phenyl carbamate fungicides. Antimicrob Agents Chemother:2171–2173
Samia MR, Soad AME, Hesham TYF, Hazzaa AA, EI-Meligy MMM (2006) Synthesis of novel benzofuran and related benzimidazole derivatives for evaluation of in vitro anti-HIV-1, anticancer and antimicrobial activities. Arch Pharm Res 29(10):826–833
Mahadevan KM, Vaidya VP (2003) Synthesis and pharmacological evaluation of some potent naphtho [2, 1-b] furo-pyrazolyl, oxadiazolyl and coumaryl derivatives. Indian J Pharm Sci 65:128–134
Simunovic M, Perkovic I, Zorc B, Ester K, Kralj M, Hadjipavlou-Litina D, Pontiki E (2009) Urea and carbamate derivatives of primaquine: synthesis, cytostatic and antioxidant activities. Bioorg Med Chem 17:5605–5613
Rangaswamy J, Kumar HV, Harini ST, Nagaraja N (2013) Functionalized 3-(benzofuran-2-yl)-5-(4-methoxyphenyl)-4,5-dihydro-1H-pyrazole scaffolds: A new class of antimicrobials and antioxidants. doi:10.1016/j.arabjc.2013.10.012
Kapche GDWF, Christian DF, Jean HD, Ghislain WF, Dawe A, Angèle NT, Merhatibeb B, Paul FM, Bonaventure TN, Berhanu MA (2009) Prenylated arylbenzofuran derivatives from Morus mesozygia with antioxidant activity. Phytochem 70:216–221
Jadhav VB, Kulkarni MV, Rasal VP, Biradar SS, Vinay MD (2008) Synthesis and anti-inflammatory evaluation of methylene bridged benzofuranyl imidazo[2,1- b] [1,3,4]thiadiazoles. E J Med Chem 43:1721–1729
Rekha R, Makrandi JK (2009) Microwave assisted and antimicrobial activity of some 2-(1-benzofuran-2-yl)-7-(substituted) imidazo, [2, 1-b] benzothiazole. Ind J Chem 48B:1614–1617
Sheelavanth S, Bodke YD, Santosh Kumar S (2012) Synthesis and antimicrobial activity of some imidazo thiazole derivatives of benzofuran. doi:10.1016/j.arabjc.2012.10.018
Kenchappa R, Bodke YD, Peethambar SK, Telkar S, Venkatesh KB (2013) Synthesis of ß-amino carbonyl derivatives of coumarin and benzofuran and evaluation of their biological activity. Med Chem Res 22:4787–4797
Kenchappa R, Bodke YD, Asha B, Telkar S, Aruna Sindhe M (2014) Synthesis, antimicrobial, and antioxidant activity of benzofuran barbitone and benzofuran thiobarbitone derivatives. Med Chem Res 23:3065–3081
Aruna SM, Yadav DB, Kenchappa R, Sandeep T, Chandrashekar A (2016) Synthesis of a series of novel 2,5-disubstituted-1,3,4-oxadiazolederivatives as potential antioxidant and antibacterial agents. J Chem Biol. doi:10.1007/s12154-016-0153-9
Sheelavanth S, Bodke YD, Sundar SM (2013) Synthesis, antioxidant, and antibacterial studies of phenolic esters and amides of 2-(1-benzofuran-2-yl) quinoline-4-carboxylic acid. Med Chem Res 22:1163–1171
Nasser Khalil SAM (2010) Efficient synthesis of novel 1, 2, 4-triazole fused acyclic and 21–28 membered macrocyclic and/or lariat macrocyclic oxaazathia crown compounds with potential antimicrobial activity. Eur J Med Chem 45:5265–5277
Sander T, Freyss J, von Korff M (2009) OSIRIS, an entirely in-house developed drug discovery informatics system. J Chem Inf Model 49:232–246
Trott O, Olson AJ (2010) Auto Dock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31:455–461
Robinson MW, McFerran N, Trudgett A, Hoey L, Fairweather I (2004) A possible model of benzimidazole binding to beta-tubulin disclosed by invoking an inter-domain movement. J Mol Graph Model 23(3):275–284
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, Olson AJ (1998) Automated docking using a Lamarckian genetic algorithm and empirical binding free energy function. J Comput Chem 19:1639–1662
Laskowski RA, Swindells MB (2011) LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. J Chem Inf Model 51:2778–2786
Delano WL (2002) The PyMOL Molecular Graphics System, Version 1.5.0.4 Schro¨dinger, LLC. (1998)
Çetinkaya E, Denizci A, Özdemir I, Ozturk HT, Karaboz I, Çetinkaya B (2002) Remarkable substituent effects on antimicrobial activities of 1, 3-diorganylimidazolidinium salts. J Chemother 14:241–245
Hayati T, Ceyhan N, Yavasoglu NU, Güven O, Çetinkaya B (2011) Synthesis and antimicrobial activities of hexahydro imidazo [1, 5-a] pyridinium bromides with varying benzyl substituents. Eur J Med Chem 46:2895–2900
Dash GK, Suresh P, Kar DM, Ganpaty S, Panda SB (2002) Evaluation of Evolvulus alsinoides Linn. For anthelmintic and antimicrobial activities J Nat Rem 2:182–185
Shivkumar YM, Kumar VL (2003) Anthelmintic activity of latex of Calotropis procera Pharma. Biol 41:263–265
Sharma P, Rane N, Gurram VK (2004) Synthesis and QSAR studies of pyrimido [4, 5-d] pyrimidine- 2, 5-dione derivatives as potential antimicrobial agents. Bioorg Med Chem Lett 14:4185–4190
Dhorajiya BD, Bhakhar BS, Dholakiya BZ (2013) Synthesis, characterization, solvate chromic properties and antimicrobial evaluation of 5-acetyl-2-thioxo-dihydropyrimidine-4,6-dione-based chalcones. Med Chem Res 22:4075–4086
Acknowledgments
The authors are thankful to the Chairman, Department of Industrial Chemistry, Kuvempu University, Shankaraghatta, for providing the laboratory facilities. One of the authors (Kenchappa. R) is thankful to the UGC for the award of Post-Doctoral Fellowship.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 3559 kb)
Rights and permissions
About this article
Cite this article
Kenchappa, R., Bodke, Y.D., Telkar, S. et al. Antifungal and anthelmintic activity of novel benzofuran derivatives containing thiazolo benzimidazole nucleus: an in vitro evaluation. J Chem Biol 10, 11–23 (2017). https://doi.org/10.1007/s12154-016-0160-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12154-016-0160-x