Abstract
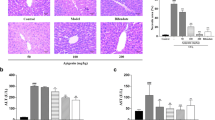
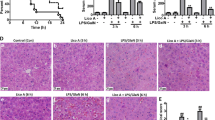
Systemic or hepatic inflammation is caused by intraperitoneal application of lipopolysaccharide (LPS). In this study, we investigated anti-inflammatory and antioxidant properties of combination of ginsenoside-Rg2 (G-Rg2) and -Rh1 (G-Rh1) on liver function under LPS challenging. We first confirmed that G-Rg2 and -Rh1 at 100 μg/ml did not show cytotoxicity in HepG2 cells. G-Rg2 and -Rh1 treatment significantly inhibited activation of STAT3 and TAK1, and inflammatory factors including iNOS, TNF-α, and IL-1β in peritoneal macrophages. In HepG2 cells, G-Rg2 and -Rh1 treatment inhibited activation of STAT3 and TAK1/c-Jun N-terminal kinase, and down-regulated nuclear translocation of NF-κB transcription factor. In addition, LPS-induced mitochondrial dysfunction was restored by treatment with G-Rg2 and -Rh1. Interestingly, pretreatment with G-Rg2 and -Rh1 effectively inhibited mitochondrial damage-mediated ROS production induced by LPS stimulation, and alterations of Nrf2 nuclear translocation and ARE promotor activity were involved in G-Rg2 and -Rh1 effects on balancing ROS levels. In liver tissues of LPS-treated mice, G-Rg2 and -Rh1 treatment protected liver damages and increased Nrf2 expression while reducing CD45 expression. Taken together, G-Rg2 and -Rh1 exerts a protective effect on liver function by increasing antioxidant through Nrf2 and anti-inflammatory activities through STAT3/TAK1 and NF-κB signaling pathways in liver cells and macrophages.







Similar content being viewed by others
References
Ahmed SM, Luo L, Namani A, Wang XJ, Tang X (2017) Nrf2 signaling pathway: pivotal roles in inflammation. Biochim Biophys Acta Mol Basis Dis 1863:585–597. https://doi.org/10.1016/j.bbadis.2016.11.005
Baatar D, Siddiqi MZ, Im WT, Ul Khaliq N, Hwang SG (2018) Anti-inflammatory effect of ginsenoside Rh2-Mix on lipopolysaccharide-stimulated RAW 264.7 murine macrophage cells. J Med Food 21:951–960. https://doi.org/10.1089/jmf.2018.4180
Bak MJ, Jun M, Jeong WS (2012) Antioxidant and hepatoprotective effects of the red ginseng essential oil in H(2)O(2)-treated hepG2 cells and CCl(4)-treated mice. Int J Mol Sci 13:2314–2330. https://doi.org/10.3390/ijms13022314
Cecconi M, Evans L, Levy M, Rhodes A (2018) Sepsis and septic shock. Lancet 392:75–87. https://doi.org/10.1016/S0140-6736(18)30696-2
Hai Y, Wang X, Song P, Li JY, Zhao LH, Xie F, Tan XM, Xie QJ, Yu L, Li Y, Wu ZR, Li HY (2019) Realgar transforming solution-induced differentiation of NB4 cell by the degradation of PML/RARalpha partially through the ubiquitin-proteasome pathway. Arch Pharm Res 42:684–694. https://doi.org/10.1007/s12272-019-01170-9
Hassanein T, Frederick T (2004) Mitochondrial dysfunction in liver disease and organ transplantation. Mitochondrion 4:609–620. https://doi.org/10.1016/j.mito.2004.07.015
Huynh DTN, Baek N, Sim S, Myung CS, Heo KS (2020a) Minor ginsenoside Rg2 and Rh1 attenuates LPS- via downregulating induced acute liver and kidney damages activation of TLR4-STAT1 and inflammatory cytokine production in macrophages. Int J Mol Sci 21:6656. https://doi.org/10.3390/ijms21186656
Huynh DTN, Jin Y, Myung CS, Heo KS (2020b) Inhibition of p90RSK is critical to abolish Angiotensin II-induced rat aortic smooth muscle cell proliferation and migration. Biochem Biophys Res Commun 523:267–273. https://doi.org/10.1016/j.bbrc.2019.12.053
Irie T, Muta T, Takeshige K (2000) TAK1 mediates an activation signal from toll-like receptor(s) to nuclear factor-kappaB in lipopolysaccharide-stimulated macrophages. FEBS Lett 467:160–164. https://doi.org/10.1016/s0014-5793(00)01146-7
Jaeschke H (2000) Reactive oxygen and mechanisms of inflammatory liver injury. J Gastroenterol Hepatol 15:718–724. https://doi.org/10.1046/j.1440-1746.2000.02207.x
Jiang Z, Meng Y, Bo L, Wang C, Bian J, Deng X (2018) Sophocarpine attenuates LPS-induced liver injury and improves survival of mice through suppressing oxidative stress, inflammation, and apoptosis. Mediat Inflamm 2018:5871431. https://doi.org/10.1155/2018/5871431
Jin Y, Huynh DTN, Nguyen TLL, Jeon H, Heo KS (2020) Therapeutic effects of ginsenosides on breast cancer growth and metastasis. Arch Pharm Res 43:773–787. https://doi.org/10.1007/s12272-020-01265-8
Kim DH (2018) Gut microbiota-mediated pharmacokinetics of ginseng saponins. J Ginseng Res 42:255–263. https://doi.org/10.1016/j.jgr.2017.04.011
Lee J, Park JS, Roh YS (2019) Molecular insights into the role of mitochondria in non-alcoholic fatty liver disease. Arch Pharm Res 42:935–946. https://doi.org/10.1007/s12272-019-01178-1
Lee SB, Lee WS, Shin JS, Jang DS, Lee KT (2017) Xanthotoxin suppresses LPS-induced expression of iNOS, COX-2, TNF-alpha, and IL-6 via AP-1, NF-kappaB, and JAK-STAT inactivation in RAW 264.7 macrophages. Int Immunopharmacol 49:21–29. https://doi.org/10.1016/j.intimp.2017.05.021
Li S, Hong M, Tan HY, Wang N, Feng Y (2016) Insights into the role and interdependence of oxidative stress and inflammation in liver diseases. Oxid Med Cell Longev 2016:4234061. https://doi.org/10.1155/2016/4234061
Liu H, Liu M, Jin Z, Yaqoob S, Zheng M, Cai D, Liu J, Guo S (2019) Ginsenoside Rg2 inhibits adipogenesis in 3T3-L1 preadipocytes and suppresses obesity in high-fat-diet-induced obese mice through the AMPK pathway. Food Funct 10:3603–3614. https://doi.org/10.1039/c9fo00027e
Liu X, Yin S, Chen Y, Wu Y, Zheng W, Dong H, Bai Y, Qin Y, Li J, Feng S, Zhao P (2018) LPSinduced proinflammatory cytokine expression in human airway epithelial cells and macrophages via NFkappaB, STAT3 or AP1 activation. Mol Med Rep 17:5484–5491. https://doi.org/10.3892/mmr.2018.8542
Lugrin J, Rosenblatt-Velin N, Parapanov R, Liaudet L (2014) The role of oxidative stress during inflammatory processes. Biol Chem 395:203–230. https://doi.org/10.1515/hsz-2013-0241
Mihaly SR, Ninomiya-Tsuji J, Morioka S (2014) TAK1 control of cell death. Cell Death Differ 21:1667–1676. https://doi.org/10.1038/cdd.2014.123
Nesseler N, Launey Y, Aninat C, Morel F, Malledant Y, Seguin P (2012) Clinical review: the liver in sepsis. Crit Care 16:235. https://doi.org/10.1186/cc11381
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284:13291–13295. https://doi.org/10.1074/jbc.R900010200
Qi GY, Mi YS, Fan R, Li RN, Wang YW, Li XY, Huang SX, Liu XB (2017) Tea polyphenols ameliorate hydrogen peroxide-and constant darkness-triggered oxidative stress via modulating the Keap1/Nrf2 transcriptional signaling pathway in HepG2 cells and mice liver. RSC Adv 7:32198–32208. https://doi.org/10.1039/c7ra05000c
Quan LH, Min JW, Sathiyamoorthy S, Yang DU, Kim YJ, Yang DC (2012) Biotransformation of ginsenosides Re and Rg1 into ginsenosides Rg2 and Rh1 by recombinant beta-glucosidase. Biotechnol Lett 34:913–917. https://doi.org/10.1007/s10529-012-0849-z
Ren Y, Wang JL, Zhang X, Wang H, Ye Y, Song L, Wang YJ, Tu MJ, Wang WW, Yang L, Jiang B (2017) Antidepressant-like effects of ginsenoside Rg2 in a chronic mild stress model of depression. Brain Res Bull 134:211–219. https://doi.org/10.1016/j.brainresbull.2017.08.009
Scholzen T, Gerdes J (2000) The Ki-67 protein: from the known and the unknown. J Cell Physiol 182:311–322. https://doi.org/10.1002/(sici)1097-4652(200003)182:3%3c311::Aid-jcp1%3e3.0.Co;2-9
Taguchi K, Kensler TW (2020) Nrf2 in liver toxicology. Arch Pharm Res 43:337–349. https://doi.org/10.1007/s12272-019-01192-3
Villarino AV, Kanno Y, Ferdinand JR, O’Shea JJ (2015) Mechanisms of Jak/STAT signaling in immunity and disease. J Immunol 194:21–27. https://doi.org/10.4049/jimmunol.1401867
Walker SR, Frank DA (2012) Screening approaches to generating STAT inhibitors: allowing the hits to identify the targets. JAKSTAT 1:292–299. https://doi.org/10.4161/jkst.22662
Yang YQ, Yan XT, Wang K, Tian RM, Lu ZY, Wu LL, Xu HT, Wu YS, Liu XS, Mao W, Xu P, Liu B (2018) Triptriolide alleviates lipopolysaccharide-induced liver injury by Nrf2 and NF-kappaB signaling pathways. Front Pharmacol 9:999. https://doi.org/10.3389/fphar.2018.00999
Yuan HD, Kim DY, Quan HY, Kim SJ, Jung MS, Chung SH (2012) Ginsenoside Rg2 induces orphan nuclear receptor SHP gene expression and inactivates GSK3beta via AMP-activated protein kinase to inhibit hepatic glucose production in HepG2 cells. Chem Biol Interact 195:35–42. https://doi.org/10.1016/j.cbi.2011.10.006
Zapelini PH, Rezin GT, Cardoso MR, Ritter C, Klamt F, Moreira JC, Streck EL, Dal-Pizzol F (2008) Antioxidant treatment reverses mitochondrial dysfunction in a sepsis animal model. Mitochondrion 8:211–218. https://doi.org/10.1016/j.mito.2008.03.002
Acknowledgements
This research was supported by National Research Foundation of Korea (2019R1C1C100733112).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declared that they have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nguyen, T.L.L., Huynh, D.T.N., Jin, Y. et al. Protective effects of ginsenoside-Rg2 and -Rh1 on liver function through inhibiting TAK1 and STAT3-mediated inflammatory activity and Nrf2/ARE-mediated antioxidant signaling pathway. Arch. Pharm. Res. 44, 241–252 (2021). https://doi.org/10.1007/s12272-020-01304-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-020-01304-4