Abstract
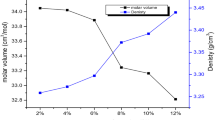
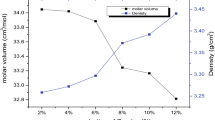
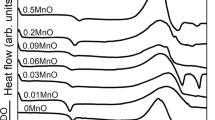
A glass system of 40P2O5–(21 − x) Na2O–38ZnO–x Co3O4–1CuO where (x = 1, 2, 3, 4 and 5) has been prepared by melt quenching technique. The density of the glasses was measured, and molar volume was calculated. The structure has been investigated using IR spectroscopy. The spectral analyses showed that the addition of Co3O4 to P2O5 glass is unequivocally influencing the short-range order of the glassy matrix. The spectra also denote the formation of P–O–Co bonds in the glasses that replace P–O−–Na+ bonds. The results are related to the dual role of cobalt ions as a glass former and modifier.


Similar content being viewed by others
References
F. Lichtenberg, K. Kleinsorgen, J. Power Sources 62, 2 (1996)
H.K. Lin, H.C. Chiu, H.C. Tsai, S.H. Chien, C.B. Wang, Catal. Lett. 88, 3–4 (2003)
S.A. Makhlouf, J. Magn. Magn. Mater. 246, 1–2 (2002)
H. Yamaura, K. Moriya, N. Miura, N. Yamazoe, Sens. Actuators B Chem. 65, 1–3 (2000)
R.J. Van Zee, Y.M. Hamrick, S. Li, W. Weltner Jr., J. Phys. Chem. 96, 18 (1992)
A.F. Trotman-Dickenson (ed.), Comprehensive Inorganic Chemistry, vol. 3 (Pergamon Press, Oxford, 1973), p. 1107
M.M. Elsemongy, M.M.A. Gouda, Y.A. Elewady, J. Electroanal. Chem. Interfacial Electrochem. 76, 3 (1977)
Y.-W.D. Chen, R.N. Noufi, Electrodeposition of nickel and cobalt oxides onto platinum and graphite electrodes for alkaline water electrolysis. J. Electrochem. Soc. 131(4), 731–735 (1984). https://doi.org/10.1149/1.2115689
C.B. Wang, H.K. Lin, C.W. Tang, Catal. Lett. 94, 1–2 (2004)
G.A. El-Shobaky, I.F. Hewaidy, Th. El-Nabarawy, Surf. Technol. (Online) 10, 3 (1980)
G.A. El-Shobaky, I.F. Hewaidy, Th. El-Nabarawy, Surf. Technol. (Online) 10, 4 (1980)
M. Figlarz, J. Guenot, F. Fievent-Vincent, J. Mater. Sci. 11, 2267–2270 (1976)
Y.H. Elbashar, M.A. Mohamed, A.M. Badr, H.A. Elshaikh, D.A. Rayan, X-ray spectroscopic analysis of nanocrystal phase growth in cobalt oxide-doped copper zinc sodium phosphate glass matrix. J. Opt. 50, 253–256 (2021). https://doi.org/10.1007/s12596-021-00679-x
D. Möncke, D. Ehrt, Radiation-induced defects in CoO- and NiO-doped fluoride, phosphate, silicate and borosilicate glasses. Glass Sci. Technol 75, 243 (2002)
N.N. Greenwood, A. Earnshaw, Chemistry of the Elements, 2nd edn. (Butterworth-Heinemann, Oxford, 1997)
S.Y. Marzouk, Ultrasonic and infrared measurements of copper-doped sodium phosphate glasses. Mater. Chem. Phys. 114, 188–193 (2009)
Y.H. Elbashar, M.A. Mohamed, D. Rayan, A.M. Badr, H.A. Elshaikh, J. Opt. 49, 2 (2020)
D.A. Rayan, Y.H. Elbashar, A.B. El Basaty, M.M. Rashad, Infrared spectroscopy of cupric oxide doped barium phosphate glass. Res. J. Pharm., Biol. Chem. Sci. 6(3), 1026 (2015)
A.H. Verhoef, H.W. den Hartog, J. Non Cryst. Solids 182, 235–247 (1995)
F.H. ElBatal, Y.M. Hamdy, S.Y. Marzouk, Mater. Chem. Phys. 112, 991–1000 (2008)
A. Abdel-Kader, A.A. Higazy, M.M. ElKholy, Compositional dependence of infrared absorption spectra studies for TeO2–P2O5 and TeO2–P2O5–Bi2O3 glasses. J. Mater. Sci. Mater. Electron. 2, 157–163 (1991)
G.J. Exarhos, in Structure and Bonding in Non-Crystalline Solids. ed. by G.E. Walrafen, A.G. Revez (Plenum Press, New York, 1986), p. 203
N.M. Rasdi, Y.W. Fen, N.A.S. Omar, R.S. Azis, M.H.M. Zaid, Effects of cobalt doping on structural, morphological, and optical properties of Zn2SiO4 nanophosphors prepared by sol–gel method. Results Phys. (2017). https://doi.org/10.1016/j.rinp.2017.09.05
C.W. Tang, C.B. Wang, S.H. Chien, Characterization of cobalt oxides studied by FT-IR, Raman, TPR and TG-MS. Thermochim. Acta 473, 68–73 (2008)
M. Kharroubi, H. Assad, S. Balme, L. Gacem, C. Maghni, Influence of Zn/Co ratio on dielectric behavior of Na2Zn1−xCoxP2O7 glasses. Ionics 22, 2355–2361 (2016). https://doi.org/10.1007/s11581-016-1770-6
K.J. Rao, H. Benqlilou-Moudden, B. Desbat, P. Vinatier, A. Levasseur, Infrared spectroscopic study of LiCoO2 thin films. J. Solid State Chem. 165, 42–47 (2002). https://doi.org/10.1006/jssc.2001.9487
P. Naresh, G. Naga Raju, C. Srinivasa Rao, S.V.G.V.A. Prasad, V. Ravikumar, N. Veeraiah, Influence of ligand coordination of cobalt ions on structural properties of ZnO–ZnF2–B2O3 glass system by means of spectroscopic studies. Phys. B 407, 712–718 (2012). https://doi.org/10.1016/j.physb.2011.12.007
S.G. Christoskovaa, M. Stoyanova, M. Georgieva, D. Mehandjiev, Preparation and characterization of a higher cobalt oxide. Mater. Chem. Phys. 60, 39–43 (1999)
T. Satyanarayana, I.V. Kityk, M. Piasecki, P. Bragiel, M.G. Brik, Y. Gandhi, N. Veeraiah, Structural investigations on PbO–Sb2O3–B2O3:CoO glass ceramics by means of spectroscopic and dielectric studies. J. Phys. Condens. Matter 21, 245104 (2009)
P. Singh, S.S. Das, S.A. Agnihotry, Structure-property correlation in (50–x) Na2O–50P2O5"–xCoCl2 glassy systems. J. Non-Cryst. Solids 351, 3730–3737 (2005). https://doi.org/10.1016/j.jnoncrysol.2005.09.034
D.E.C. Corbridge, E.J. Lowe, The infrared spectra of inorganic phosphorus compounds. Part II. Some salts of phosphorus oxy-acids. J. Chem. Soc. 493, 4555–4564 (1954). https://doi.org/10.1039/jr9540004555
P.P. Tsai, M. Greenblatt, Lithium ion conducting glasses in the system LiCI–Li2O–P2O5–SiO2. J. Non-Cryst. Solids 103, 101 (1988)
Y.M. Moustafa, K. El-Egili, Infrared spectra of sodium phosphate glasses. J. Non-Cryst. Solids 240, 144–153 (1998). https://doi.org/10.1016/S0022-3093(98)00711-X
M.J. Cleare, W.P. Griffith, Infrared spectra of isotopically substituted Nitro-, spectra of isotopically substituted Nitro-, Nitrito-, and Nitrosyl Corn pl exes. J. Chem. SOC. (A), pp. 1144–1147 (1967)
C. Garrigou-Lagrange, M. Ouchetto, B. Elouadi, Can. J. Chem. 63, 1436–1446 (1985)
P.K. Jha, O.P. Pandey, K. Singh, FTIR spectral analysis and mechanical properties of sodium phosphate glass-ceramics. J. Mol. Struct. (2014). https://doi.org/10.1016/j.molstruc.2014.11.027
Y.B. Saddeek, S.M. Abo-naf, Arch. Acoust. 37, 3 (2012)
V.N. Rai, B.N. Raja Sekhar, D.M. Phase, S.K. Deb, Effect Of gamma irradiation on the structure and valence state of Nd in phosphate glass (2014). arXiv:1406.4686
H.S. Liu, T.S. Chin, S.W. Yung, Mater. Chem. Phys. 50, 1 (1997)
W. Ahmina, M. El Moudane, M. Zriouil, M. Taibi, J. Mater. Environ. Sci. 7, 2 (2016)
E. Nabhan, A. Nabhan, N. Abd El Aal, Am. J. Phys. Appl. (Online) 4, 6 (2016)
B. Tiwari, A. Dixit, G.P. Kothiyal, M. Pandey, S.K. Deb, BARC Newsl. (Online) 285, 167–173 (2007)
A. Shaim, M. Et-tabirou, Mater. Chem. Phys. 80, 63–67 (2003)
A.M. Nassar, M.M. El Oker, Sh.N. Radwan, E. Nabhan, Curr. Sci. Int. 2, 2 (2013)
Y.M. Lai, X.F. Liang, S.Y. Yang, J.X. Wanga, L.H. Cao, B. Dai, Raman and FTIR spectra of iron phosphate glasses containing cerium. J. Mol. Struct. 992, 84–88 (2011)
A.A. Higazy, B. Bridge, J. Mater. Sci. 20, 2345–2358 (1985)
D.A. Magdas, O. Cozar, V. Chis, I. Ardelean, N. Vedeanu, The structural dual role of Fe2O3 in some lead-phosphate glasses. Vib. Spectrosc. 48, 251–254 (2008)
H. Doweidar, Y.M. Moustafa, K. El-Egili, I. Abbas, Infrared spectra of Fe2O3–PbO–P2O5 glasses. Vib. Spectrosc. 37, 91–96 (2005)
Y.H. Elbashar, A.E. Omran, S.M. Hussien, M.A. Mohamed, R.A. Ibrahem, W.A. Rashidy, A.S. AbdelRahaman, H.H. Hassan, Molecular and spectroscopic analysis of zinc oxide doped sodium phosphate glass. Nonlinear Opt. Quantum Opt. 52(3–4), 337–347 (2020)
Y. Elbashar, D. Rayan, S. ElGabaly, A. Mohamed, Egypt J. Chem. 63, 6 (2020)
E.M. Elfeky, Y.H. Elbashar, M.H. Barakat, M.R. Shehata, S.S. Hassan, Structural investigation and applications of glassy sodium phosphate including the kinetics of dissolution rates and spectral analysis of the prepared samples with a focus on their effects on water treatment. Opt. Quantum Electron. 51, 102 (2019). https://doi.org/10.1007/s11082-019-1822-z
Y.H. Elbashar, S.S. Moslem, D.A. Rayan, H.H. Hassan, J. Microw. Power Electromagn. Energy 53, 3 (2019)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elbashar, Y.H., Mohamed, M.A., Badr, A.M. et al. Molecular spectroscopic analysis of cobalt oxide-doped copper zinc sodium phosphate glass matrix. J Opt 51, 795–800 (2022). https://doi.org/10.1007/s12596-021-00752-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12596-021-00752-5