Abstract
Purpose
Perioperative intravenous ketamine may be a useful addition in pain management regimens. Previous systematic reviews have included all methods of ketamine administration, and heterogeneity between studies has been substantial. This study addresses this issue by narrowing the inclusion criteria, using a random effects model, and performing subgroup analysis to determine the specific types of patients, surgery, and clinical indications which may benefit from perioperative ketamine administration.
Source
We included published studies from 1966 to 2010 which were randomized, double-blinded, and placebo-controlled using intravenous ketamine (bolus or infusion) to decrease postoperative pain. Studies using any form of regional anesthesia were excluded. No limitation was placed on the ketamine dose, patient age, or language of publication.
Principal findings
Ninety-one comparisons in seventy studies involving 4,701 patients met the inclusion criteria (2,652 in ketamine groups and 2,049 in placebo groups). Forty-seven of these studies were appropriate for evaluation in the core meta-analysis, and the remaining 23 studies were used to corroborate the results. A reduction in total opioid consumption and an increase in the time to first analgesic were observed across all studies (P < 0.001). The greatest efficacy was found for thoracic, upper abdominal, and major orthopedic surgical subgroups. Despite using less opioid, 25 out of 32 treatment groups (78%) experienced less pain than the placebo groups at some point postoperatively when ketamine was efficacious. This finding implies an improved quality of pain control in addition to decreased opioid consumption. Hallucinations and nightmares were more common with ketamine but sedation was not. When ketamine was efficacious for pain, postoperative nausea and vomiting was less frequent in the ketamine group. The dose-dependent role of ketamine analgesia could not be determined.
Conclusion
Intravenous ketamine is an effective adjunct for postoperative analgesia. Particular benefit was observed in painful procedures, including upper abdominal, thoracic, and major orthopedic surgeries. The analgesic effect of ketamine was independent of the type of intraoperative opioid administered, timing of ketamine administration, and ketamine dose.
Résumé
Objectif
La kétamine intraveineuse périopératoire peut constituer un ajout utile à l’arsenal thérapeutique de prise en charge de la douleur. Les revues méthodiques précédentes ont examiné toutes les méthodes d’administration de la kétamine, et l’hétérogénéité entre les études est considérable. Cette étude aborde la question en limitant les critères d’inclusion, en utilisant un modèle à effets aléatoires et en réalisant une analyse de sous-groupe afin de déterminer les types spécifiques de patients et de chirurgie ainsi que les indications cliniques qui pourraient bénéficier de l’administration périopératoire de kétamine.
Source
Nous avons inclus les études publiées entre 1966 et 2010 qui étaient randomisées, à double insu et contrôlées par placebo et qui ont utilisé la kétamine intraveineuse (bolus ou perfusion) afin de réduire la douleur postopératoire. Les études ayant recours à une forme d’anesthésie régionale, quelle qu’elle soit, ont été exclues. Aucune limite n’a été placée au niveau de la dose de kétamine, de l’âge des patients ou de la langue de publication.
Constatations principales
Quatre-vingt-onze comparaisons dans soixante-dix études portant sur 4701 patients respectaient nos critères d’inclusion (2652 dans les groupes kétamine et 2049 dans les groupes placebo). Quarante-sept de ces études convenaient à une évaluation dans la méta-analyse centrale, et les autres 23 études ont été utilisées pour corroborer les résultats. Une réduction de la consommation totale d’opioïdes et une augmentation du temps jusqu’à administration du premier agent analgésique ont été observées dans toutes les études (P < 0,001). La plus grande efficacité a été observée dans les sous-groupes de chirurgie thoracique, de chirurgie abdominale supérieure et de chirurgie orthopédique majeure. Malgré l’utilisation de moins d’opioïdes, 25 des 32 groupes de traitement (78 %) ont ressenti moins de douleur que les groupes placebo à un certain moment en postopératoire lorsque la kétamine était efficace. Ces résultats indiquent une meilleure qualité du contrôle de la douleur outre une consommation réduite d’opioïdes. Les hallucinations et les cauchemars étaient plus fréquents avec la kétamine, mais pas la sédation. Lorsque la kétamine a permis un contrôle efficace de la douleur, les nausées et vomissements postopératoires étaient moins fréquents dans le groupe kétamine. Le rôle dose-dépendant de l’analgésie à base de kétamine n’a pas pu être déterminé.
Conclusion
La kétamine intraveineuse est un ajout efficace à l’analgésie postopératoire. Des bienfaits particuliers ont été observés dans les interventions douloureuses, notamment les chirurgies abdominales supérieures, thoraciques et orthopédiques majeures. L’effet analgésique de la kétamine était indépendant du type d’opioïde peropératoire administré, du moment d’administration de la kétamine, et de la dose de kétamine.
Similar content being viewed by others
Acute pain management is an important aspect of perioperative anesthetic care. Inadequate postoperative analgesia has been shown to contribute to adverse outcomes, including, but not limited to, immunosuppression, hyperglycemia, poor rehabilitation, and progression to chronic pain. Opioids are the mainstay for the management of acute pain, with adjuvant therapies, such as non-steroidal anti-inflammatory drugs, acetaminophen, gabapentin, and regional techniques, reducing opioid consumption. However, these therapeutic options are not desirable in all patients due to side effects, opioid tolerance, organ failure, and various drug interactions.
Ketamine, an N-methyl-D-aspartate (NMDA) antagonist, has undergone a recent resurgence of interest amongst acute care providers as an adjunct in acute pain management. Blocking the NMDA receptor may improve the efficacy of opioids and reduce the development of chronic pain syndromes. Ketamine used in higher doses is less desirable due to adverse effects, such as hallucinations, nightmares, nausea, dizziness, and blurred vision, and when used in low doses for the management of acute postoperative pain, evidence of efficacy has been inconclusive. Also, the definition of “low-dose” ketamine has been inconsistent and inconclusive.1,2 Randomized controlled trials (RCTs) and subsequent systematic reviews evaluating the efficacy of ketamine for postoperative analgesia have had mixed results, with some results being very positive and others showing no difference from placebo. In previous systematic reviews, ketamine has been evaluated over a wide range of doses as well as via various routes of administration.3-9 Results have been relatively inconclusive, due at least in part to the significant heterogeneity amongst the studies.
We therefore undertook a new systematic review using a random effects model with a focus on RCTs reporting the perioperative use of intravenous ketamine. The primary outcome was total postoperative opioid consumption. Secondary outcomes included time to first analgesic, pain scores, and side effects. Further subgroup analyses provided insights into the subsets of patients which might benefit most from the use of perioperative ketamine and the factors which might influence the efficacy of ketamine in postoperative pain management.
Methods
The systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.10 Since ketamine was developed as a drug in 1962, only studies after this date were searched. A literature search was conducted using Medline (1966–2010), EMBASE (1980–2010), The Cochrane Library, and hand searching relevant reference lists, including those of the existing systematic reviews.3-9 An attempt was made to contact the manufacturers of ketamine (Pfizer and Erfa) and individual authors for missing data as necessary. Searches were conducted using an extensive search strategy as previously described by Bell et al.3 (see the Appendix).
Studies meeting inclusion criteria were identified by title and abstract. These criteria include the following: 1) double-blinded randomized placebo-controlled trials; 2) administration of perioperative ketamine; 3) administration of intravenous ketamine; and 4) measurement of postoperative analgesic use or pain. Studies using any form of regional anesthesia were excluded. There were no limitations on ketamine dose, patient age, or language of the study.
Two independent reviewers identified all abstracts for possible inclusion and exclusion according to the above-mentioned criteria, with removal of duplicates. Full text articles of this subset were further reviewed for inclusion and exclusion. Discrepancies between independent reviewers were resolved by discussion with an independent third party.
Two reviewers then jointly performed the assessment of risk of bias and data extraction. Only high-quality double-blinded randomized placebo-controlled studies were included. Assessment of risk of bias included evaluation of the adequacy of randomization, concealment of allocation, appropriate blinding, and the extent of loss to follow-up as suggested in the Cochrane Collaboration’s tool for assessing risk of bias.11 Where these details were not explicitly reported, a consensus judgement was made by the reviewers to estimate risk of bias, and studies with a high risk of bias were excluded.
Data extraction was performed collecting the following data points on a predesigned form: 1) year of publication, 2) number of patients (treatment vs placebo), 3) patient age (adult vs child), 4) dose of ketamine used (total dose, bolus, and infusion rates), 5) timing (preincision, intraoperative, postoperative, concurrent with patient-controlled analgesia), 6) type of surgery, 7) intraoperative narcotic used, 8) pain scores (<24 hr, >24 hr, and maximum postoperative), 9) total narcotic used, 10) time to first narcotic, 11) side effects reported (hallucinations, nausea, sedation, other), 12) use of nitrous oxide, and 13) use of benzodiazepines. Attempts were made to complete missing data fields as necessary by contacting authors directly.
Data was analyzed using a random effects model within a comprehensive meta-analysis database program.12 A random effects model was chosen due to the highly divergent outcomes found in previous meta-analyses. This model is statistically more robust where study population and sample size are heterogeneous among studies as it takes account of variance both within and between studies.13 Outcome measures examined quantitatively included total opioid consumption and time to first analgesic, which were measured by standard difference in means (SDM), also known as Cohen’s effect size or Cohen’s d. When the confidence interval (CI) was reported, we calculated standard deviation (SD) using the following formula: 95% CI = x ± (1.96)(SEM) where SEM = SD/√(n). When mean and SD or CI were not available, the studies were excluded from the initial meta-analysis. These datasets were converted to mean and SD equivalents and used to corroborate the results of the meta-analysis. When available, P values and sample sizes were used. When only median and interquartile range (IQR) were available, normal distribution was assumed, and SD was calculated using SD = IQR/1.349 as done in previous meta-analyses.14
Standard difference in means was used as the effect size (ES) measurement to allow combining different opioids that have very different appropriate doses. There is reasonably good agreement among statisticians about ES interpretation for SDM. Cohen, who described the measurement, stated that a small ES < 0.2, a medium ES = 0.5, and a large ES > 0.8.15 Lipsey and Wilson16 studied the issue formally in 302 meta-analyses, and they found the bottom quartile effect size ≤ 0.3, the median = 0.5, and the upper quartile ≥ 0.67. Based on this, we consider that SDM > 0.6 is evidence of a difference which is likely to be clinically significant.
Predetermined subgroup analyses included patient age, ketamine dose, timing and duration of administration, type of intraoperative opioid, site of surgery, worst reported postoperative pain, and use of nitrous oxide. Use of nitrous oxide was considered important as it is also known to have NMDA antagonist activity.17 Bonferroni correction of the significance level α for multiple comparisons was used by family, with each subgroup considered a family.18 The effects of maximum pain score and total dose of ketamine on total opioid use were analyzed as fixed effects using metaregression. The maximum pain score was converted to a fraction of the scale’s maximum to allow comparisons between scales.19
Meta-analysis of the primary outcome, total postoperative opioid use, was examined for publication bias using classic fail-safe as well as Duval and Tweedie’s trim and fill analysis.20 Heterogeneity was determined using I-squared (I2).21
Due to the various methods of reporting pain scores, including scales, timing, and numerous data points, it was difficult to merge these data into a meaningful quantitative analysis. Thus, pain scores and side effects were analyzed qualitatively. Only studies explicitly reporting the side effect were included in the analysis of that side effect. Nausea and vomiting were combined as “postoperative nausea and vomiting” (PONV), with either or both counting as one event. A Chi square test was used to calculate P values for each side effect as well as for subgroups when ketamine was efficacious (as defined by SDM > 0.6 for total narcotic use) and when it was not. Bonferroni corrections were also applied to these P values.19
Results
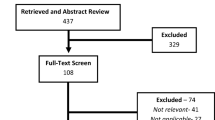
A total of 70 studies met inclusion criteria (see Fig. 1). Literature searches and review of reference lists as of December 31, 2010 revealed 2,257 titles and abstracts which were screened by two independent reviewers. Once duplicated studies were removed, the remaining 1,746 abstracts were screened. A total of 129 full text studies were assessed for eligibility, and 59 of these were excluded. Of these studies, 11 were excluded due to the use of regional anesthesia, 23 had an inadequate placebo group, ten were not explicit about their blinding procedures, five had inadequate randomization, six had different outcomes than our analysis, three were not RCTs, and one study was unavailable as it was in the British Lending Library and the section of the library holding the paper was closed for asbestos removal.22
Of the 70 studies meeting inclusion criteria, 64 were suitable for inclusion in the quantitative analysis, and the remaining six were added to the qualitative analysis. These six studies reported data which were not useable in the quantitative analysis, such as median and range (two), mean only (one), mean and range (one), graphical data and SEM (one), and number of events of non-opioid rescue analgesia (one). The 70 studies included a total of 4,701 patients (2,049 in placebo groups and 2,652 in treatment groups) with 92 treatment arms and 71 placebo arms. Of the studies included, 14 single treatment arms were excluded (13 studies: one due to route of administration, four due to lack of control, and nine because a different drug was used for comparison). Within the 64 studies remaining for the quantitative analysis, 17 reported data other than mean and standard deviations or confidence intervals. Specifically, seven studies reported median and interquartile ranges, five studies reported integer data or events, and five studies reported a sample size and P value. Thus, 47 studies23-69 were initially analyzed quantitatively, an additional 17 studies70-86 were used to corroborate the results within the quantitative analysis, and the remaining six studies87-92 were added to the qualitative analysis.
All 70 studies had either low or unclear risks of bias in all six categories of the Cochrane Collaboration’s tool for assessing risk of bias (see Table 1). Publication bias was shown to be highly unlikely. Classic fail-safe analysis required finding 6,709 additional missing studies to bring the P value to >0.05. Duval and Tweedie’s trim and fill test for effects of publication or other bias applied to the primary outcome, postoperative opioid use, showed no change in the SDM or in the 95% CI (Q-value = 399; P < 0.0001). As expected, there was considerable heterogeneity (I2 = 82.98). Therefore, a random effects model with subgroup analysis was undertaken as proposed.
The analysis of the 47 core studies using the random effects model resulted in a SDM for total opioid consumed of −0.631 (95%CI = −0.802 to −0.459; P < 0.001), demonstrating an opioid sparing effect with treatment (see Fig. 2). The SDM for time to first analgesic was 0.905 (95%CI = 0.551 to 1.259; P < 0.001), demonstrating a significant increase in the time to first postoperative analgesic (see Fig. 3). Heterogeneity within the 47 studies remained high (I2 = 83.44), as can also be seen visually on the forest plot graph (see Fig. 2). In comparison, when adding the 17 studies requiring assumptions and/or conversions to the analysis for a total of 64 studies, the SDM for total opioid consumed remained significant (SDM = −0.646; 95%CI = −0.797 to −0.495; P < 0.001), and the SDM for time to first analgesic also remained significant (SDM = 0.848; 95%CI = 0.542 to 1.155; P < 0.001).
Subgroups were created as described previously, and the 47 core studies were analyzed initially using total opioid consumed as the measured endpoint (see Table 2). No differences were identified based on age, ketamine dose, timing and duration of administration, type of intraoperative opioid, or presence of nitrous oxide. The most significant differences were found based on the site of surgery and the maximum visual analogue scale (VAS)-equivalents reported immediately postoperatively. The least opioid reduction was found in those patients undergoing head and neck surgery, dental surgery, or tonsillectomy (SDM = 0.065; 95%CI = −0.201 to 0.331; P = 0.631). This was also the group with the least heterogeneity (I2 = 13.2). Upper abdominal and thoracic procedures showed the greatest decrease in opioid used (SDM = −1.741; 95%CI = −2.625 to −0.857; P < 0.001). Orthopedic (limb and spine), intra-abdominal and lower abdominal surgery also showed decreased opioid use with the use of ketamine. Maximum VAS-equivalents greater than seven out of ten showed the greatest opioid reduction with ketamine use (SDM = −1.063; 95%CI = −1.521 to −0.605; P < 0.001). In contrast, no decrease in opioid dose was found with maximum VAS-equivalents less than four out of ten (SDM = −0.437; 95%CI = −1.209 to 0.335; P = 0.268). Metaregression of maximum VAS-equivalents showed a significant correlation (slope = −1.22; 95%CI = −1.701 to −0.746; P < 0.001) (see Fig. 4). Metaregression of the total dose of ketamine received showed a much less significant degree of correlation (slope = −0.04; 95%CI = −0.069 to −0.001; P = 0.009). Addition of the 17 studies requiring assumptions to each subgroup analysis resulted in no significant differences from the core analysis.
Pain scores were reported using various scales in all 70 studies. These scales included VAS, verbal rating scale, verbal pain scale, numerical rating scale, Children’s Hospital of Eastern Ontario pain scale, Hanallah pain score, objective pain scale, McGill pain questionnaire, and other pain scores or measurements. Overall, 37.5% of studies showed a significant decrease in early pain scores (30 min-4 hr), and 25% showed a significant decrease in late pain scores (24-72 hr). Despite using more opioid, 78% of the placebo groups experienced significantly more pain than the ketamine treatment groups when ketamine was efficacious. This occurred in only 16% of the studies where ketamine was not efficacious.
No serious side effects (death, loss of limb, or organ) were reported. Although reporting was variable, most studies reported neuropsychiatric effects (hallucinations, psychiatric disturbances, unpleasant dreams, diplopia, blurred vision, nystagmus, or dysphoria), nausea and/or vomiting, and sedation. Most studies reported event counts rather than severity, so we analysed only counts. Overall, the treatment groups had increased neuropsychiatric effects (P = 0.018), decreased PONV (P = 0.018), and no difference in sedation (P = 0.99) when compared with placebo. However, when efficacious studies were analyzed alone, the neuropsychiatric effects became significant (P < 0.001), and PONV was significantly decreased in the treatment groups (P < 0.001) (see Table 3). There was no difference in the incidence of pruritis, urinary retention, or other reported side effects.
Discussion
Heterogeneity among studies was significant and remains a limitation of systemically reviewing this topic. This study uses two approaches to search more deeply than previous reviews in dealing with the heterogeneity of RCTs. First, we attempted to decrease heterogeneity by limiting the review to RCTs using intravenous ketamine without regional anesthesia. Since regional techniques are so successful at decreasing postoperative pain, we presumed that regional anesthesia would act as a confounder that would increase heterogeneity and mask efficacy. In our view, the main role of ketamine would be to treat patients where regional anesthesia is not used. Second, we extracted the most severe pain recorded in each study to determine whether severity of pain is a factor in ketamine efficacy.
Several of the studies meeting inclusion criteria did not report data as mean (SD), and they may not have tested for normality. The meta-analysis program deals with these issues by making the assumption of normal distribution of data, which may not always be the case. We tested the importance of this issue by analyzing a subset of 47 studies which expressed their results as mean (SD) or 95%CI. In general, including all 64 studies rather than limiting them to those 47 did not change the conclusions but only strengthened them.
The results of this analysis suggest that intravenous ketamine has an opioid sparing effect. This effect is seen as a reduction in the quantity of opioid administered and prolonged time to first analgesic across all studies. The choice of subgroup categories is necessarily somewhat arbitrary, with some overlap of categories. Nonetheless, particular benefit, as defined by decreased opioid use, was observed in painful procedures (VAS-equivalent > 70% of the maximum score on any pain scale). Ketamine was not beneficial for surgery associated with mild pain (VAS-equivalent < 40% of the maximum score on any pain scale). By surgical site, ketamine was not efficacious for tonsillectomy, dental or head and neck surgery, but it had significant analgesic benefit for those procedures involving the upper abdomen and thorax. Timing of administration (pre- or post-incision) and size of dose had no influence, and there was no pre-emptive effect identified.
Within subgroup comparisons, the pediatric subgroup appears to be poorly represented (only four studies). However, there were a large number of pediatric studies present in the additional 17 studies. When these data are included (total 11 treatment arms), the lack of benefit persists in the pediatric populations studied. This is explained, at least partially, by the relatively high representation of pediatric tonsillectomy studies. The lack of efficacy in children may be a product of surgical site rather than age.
Despite the clear opioid-reducing effect of ketamine in certain clinical scenarios, what is the actual clinical benefit? In essence, what is wrong with using higher doses of opioid? This question can be answered, at least partially, by looking more closely at the pain scores in these studies. Despite using more opioid, 78% of the placebo groups experienced significantly more pain than the ketamine treatment groups when ketamine was efficacious. This implies an improved quality of pain control in addition to the decrease in opioid consumption. One weakness of all analgesic studies is the lack of an objective measure of pain; however, the pain scores used are validated measures. Another potential clinical benefit is the reduction of opioid-induced side effects. Overall, for example, ketamine demonstrated a significant reduction in the percentage of PONV (P = 0.018). This becomes much more significant when ketamine is efficacious (P < 0.001) and becomes insignificant when it is not.
The perioperative use of ketamine as an adjuvant to pain management was shown to be relatively safe with no serious side effects. However, ketamine is known for its neuropsychiatric side effects, and this is a drawback for the routine use of this drug. Overall, there was an increase in such side effects with the treatment of ketamine (P = 0.018), which becomes more prevalent with treatment efficacy (P < 0.001). This is despite most individual articles finding these effects to be not statistically significant. Many papers commented that the psychological side effects were well tolerated.
These results suggest that perioperative low-dose ketamine may be useful in a variety of clinical settings. First, it may be used when the anticipated postoperative VAS score is greater than 7/10, and it may be given at any point (preemptively, intraoperatively, postoperatively) and in any method (bolus, infusion, patient-controlled analgesia co-administration). Second, the site of surgery (and possibly the extent of the incision) has an impact on the efficacy of ketamine as a perioperative adjuvant drug. However, we postulate that pain severity is more important than surgical site. So, for example, it would be wrong to deny all lower abdominal surgery patients ketamine on the basis of this review, since there is a large difference in postoperative pain between minor and major operations. There may be factors other than the severity of surgical pain that contribute to success or failure of intravenous ketamine, but that remains unexplored. The reduction in opioid dose may particularly benefit those patients for whom opioid sparing is important, such as patients who are intolerant to opioid side effects and those with a history of PONV. Since neuropsychiatric disturbances remain an issue with the use of ketamine, one must weigh the risks and benefits of this intervention. Also, these disturbances may be potentially decreased by the use of a benzodiazepine (although most studies included did this) and by limiting the dose. The optimal dose of ketamine remains unclear as there appeared to be no correlation between dose and efficacy.
There is little to be gained from more RCTs evaluating ketamine’s role in surgery known to produce mild pain. Further study should be directed towards unanswered questions; the focus should be on studying patients at risk for severe postoperative pain and respiratory depression and on investigating the rescue of patients who continue to suffer severe postoperative pain despite routine treatment. The orthopedic studies included mildly painful arthroscopic surgery which showed no benefit from ketamine. More painful surgery should be studied, such as total joint replacement or fracture fixation under general anesthesia. Finally, the prevention and treatment of chronic pain is relatively unexplored.
In conclusion, intravenous ketamine is effective in reducing total opioid requirements and delaying the time to first analgesic dose for many patients with postoperative pain. Subgroup analyses show the greatest opioid sparing effect occurring when high maximum postoperative pain scores are encountered. When the drug spares opioid requirements, it decreases PONV at the expense of an increase in neuropsychiatric disturbances. Thus, ketamine has a clinical benefit when used perioperatively in certain clinical circumstances while potentially causing harm in others.
References
Himmelseher S, Durieux M. Ketamine for perioperative pain management. Anesthesiology 2005; 102: 211-20.
Berti M, Baciarello M, Troglio R, Fanelli G. Clinical uses of low-dose ketamine in patients undergoing surgery. Curr Drug Targets 2009; 10: 707-15.
Bell RF, Dahl JB, Moore RA, Kalso E. Perioperative ketamine for acute postoperative pain. Cochrane Database Syst Rev 2006; 1: CD004603.
Elia N, Tramer MR. Ketamine and postoperative pain – a quantitative systematic review of randomized trials. Pain 2005; 113: 61-70.
Schmid RL, Sandler AN, Katz J. Use and efficacy of low-dose ketamine in the management of acute postoperative pain: a review of current techniques and outcomes. Pain 1999; 82: 111-25.
McCartney CJ, Sinha A, Katz J. A qualitative systematic review of the role of N-methyl-D-aspartate receptor antagonists in preventive analgesia. Anesth Analg 2004; 98: 1385-400.
Ong CK, Lirk P, Seymour RA, Jenkins BJ. The efficacy of preemptive analgesia for acute postoperative pain management: a meta-analysis. Anesth Analg 2005; 100: 757-73.
Subramaniam K, Subramaniam B, Steinbrook RA. Ketamine as adjuvant analgesic to opioids: a quantitative and qualitative systematic review. Anesth Analg 2004; 99: 482-95.
Carstensen M, Moller AM. Adding ketamine to morphine for intravenous patient-controlled analgesia for acute postoperative pain: a qualitative review of randomized trials. Br J Anaesth 2010; 104: 401-6.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009; 6: e1000097.
Higgins JP, Green S. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from URL: www.cochrane-handbook.org (accessed June 2011).
Borenstein M, Hedges L, Higgins J, Rothstein H. Comprehensive Meta-analysis; Version 2.2.046. Englewood, NJ: BioStat Inc; 2005.
Borenstein M, Hedges LV, Higgins JP, Rothstein HR. Introduction to Meta-Analysis. Hoboken, NJ: John Wiley & Sons, Ltd; 2009.
Seib RK, Paul JE. Preoperative gabapentin for postoperative analgesia: a meta-analysis. Can J Anesth 2006; 53: 461-9.
Lipsey MW, Wilson DB. Practical meta-analysis. Applied social research methods series 2001; vol 49.
Lipsey MW, Wilson DB. The efficacy of psychological, educational, and behavioral treatment. Confirmation from meta-analysis. Am Psychol 1993; 48: 1181-209.
Jevtovic-Todorovic V, Todorovic SM, Mennerick S, et al. Nitrous oxide (laughing gas) is an NMDA antagonist, neuroprotectant and neurotoxin. Nat Med 1998; 4: 460-3.
Proschan MA, Waclawiw MA. Practical guidelines for multiplicity adjustment in clinical trials. Control Clin Trials 2000; 21: 527-39.
Williamson A, Hoggart B. Pain: a review of three commonly used pain rating scales. J Clin Nurs 2005; 14: 798-804.
Duval SJ, Tweedie RL. A non-parametric “trim and fill” method of accounting for publication bias in meta-analysis. Journal of the American Statistical Association 2000; 95: 89-98.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med 2001; 21: 1539-58.
Kaizu M. Study of ketamine injection technique under general anesthesia: comparison between micro-mini drip and bolus injection. Journal of the Japanese Dental Society of Anesthesiology 2001; 29: 24-33.
Adriaenssens G, Vermeyen KM, Hoffmann VL, Mertens E, Adriaensen HF. Postoperative analgesia with i.v. patient-controlled morphine: effect of adding ketamine. Br J Anaesth 1999; 83: 393-6.
Aubrun F, Gaillat C, Rosenthal D, et al. Effect of a low-dose ketamine regimen on pain, mood, cognitive function and memory after major gynaecological surgery: a randomized, double-blind, placebo-controlled trial. Eur J Anaesthesiol 2008; 25: 97-105.
Aveline C, Gautier JF, Vautier P, et al. Postoperative analgesia and early rehabilitation after total knee replacement: a comparison of continuous low-dose intravenous ketamine versus nefopam. Eur J Pain 2009; 13: 613-9.
Batra YK, Shamsah M, Al-Khasti MJ, Rawdhan HJ, Al-Qattan AR, Belani KG. Intraoperative small-dose ketamine does not reduce pain or analgesic consumption during perioperative opioid analgesia in children after tonsillectomy. Int J Clin Pharm Ther 2007; 45: 155-60.
Chazan S, Buda I, Nesher N, Paz J, Weinbroum AA. Low-dose ketamine via intravenous patient-controlled analgesia device after various transthoracic procedures improves analgesia and patient satisfaction. Pain Manag Nurs 2010; 11: 169-76.
Dahl V, Ernoe PE, Steen T, Raeder JC, White PF. Does ketamine have preemptive effects in women undergoing abdominal hysterectomy procedures? Anesth Analg 2000; 90: 1419-22.
Dal D, Kose A, Honca M, Akinci SB, Basgul E, Aypar U. Efficacy of prophylactic ketamine in preventing postoperative shivering. Br J Anaesth 2005; 95: 189-92.
Deng GF, Zheng JP, Wang S, Tian B, Zhang SG. Remifentanil combined with low-dose ketamine for post-operative analgesia of lower limb fracture: a double-blind, controlled study. Chin J Traumatol 2009; 12: 223-7.
Dullenkopf A, Muller R, Dillmann F, Wiedemeier P, Hegis TR, Gautschi S. An intraoperative pre-incision single dose of intravenous ketamine does not have an effect on postoperative analgesic requirements under clinical conditions. Anaesth Intensive Care 2009; 37: 753-7.
Engelhardt T, Zaarour C, Naser B, et al. Intraoperative low-dose ketamine does not prevent a remifentanil-induced increase in morphine requirement after pediatric scoliosis surgery. Anesth Analg 2008; 107: 1170-5.
Ganne O, Abisseror M, Menault P, et al. Low-dose ketamine failed to spare morphine after a remifentanil-based anaesthesia for ear, nose and throat surgery. Eur J Anaesthesiol 2005; 22: 426-30.
Gilabert Morell A, Sanchez Perez C. Effect of low-dose intravenous ketamine in postoperative analgesia for hysterectomy and adnexectomy (Spanish). Rev Esp Anestesiol Reanim 2002; 49: 247-53.
Gillies A, Lindholm D, Angliss M, Orr A. The use of ketamine as rescue analgesia in the recovery room following morphine administration – a double-blind randomised controlled trial in postoperative patients. Anaesth Intensive Care 2007; 35: 199-203.
Guillou N, Tanguy M, Seguin P, Branger B, Campion JP, Malledant Y. The effects of small-dose ketamine on morphine consumption in surgical intensive care unit patients after major abdominal surgery. Anesth Analg 2003; 97: 843-7.
Hadi BA, Al Ramadani R, Daas R, Naylor I, Zelko R. Remifentanil in combination with ketamine versus remifentanil in spinal fusion surgery – a double blind study. Int J Clin Pharmacol Ther 2010; 48: 542-8.
Heinke W, Grimm D. Preemptive effects caused by co-analgesia with ketamine in gynecological laparotomies? (German). Anaesthesiol Reanim 1999; 24: 60-4.
Hercock T, Gillham MJ, Sleigh J, Jones SF. The addition of ketamine to patient controlled morphine analgesia does not improve quality of analgesia after total abdominal hysterectomy. Acute Pain 1999; 2: 68-72.
Jaksch W, Lang S, Reichhalter R, Raab G, Dann K, Fitzal S. Perioperative small-dose S(+)-ketamine has no incremental beneficial effects on postoperative pain when standard-practice opioid infusions are used. Anesth Analg 2002; 94: 981-6.
Javery KB, Ussery TW, Steger HG, Colclough GW. Comparison of morphine and morphine with ketamine for postoperative analgesia. Can J Anesth 1996; 43: 212-5.
Jensen LL, Handberg G, Helbo-Hansen HS, et al. No morphine sparing effect of ketamine added to morphine for patient-controlled intravenous analgesia after uterine artery embolization. Acta Anesthesiol Scand 2008; 52: 479-86.
Kafali H, Aldemir B, Kaygusuz K, Gursoy S, Kunt N. Small-dose ketamine decreases postoperative morphine requirements. Eur J Anaesthesiol 2004; 21: 916-7.
Kapfer B, Alfonsi P, Guignard B, Sessler DI, Chauvin M. Nefopam and ketamine comparably enhance postoperative analgesia. Anesth Analg 2005; 100: 169-74.
Karaman S, Kocabas S, Zincircioglu C, Firat V. Has ketamine preemptive analgesic effect in patients undergoing abdominal hysterectomy? (Turkish). Agri 2006; 18: 36-44.
Katz J, Schmid R, Snijdelaar DG, Coderre TJ, McCartney CJ, Wowk A. Pre-emptive analgesia using intravenous fentanyl plus low-dose ketamine for radical prostatectomy under general anesthesia does not produce short-term or long-term reductions in pain or analgesic use. Pain 2004; 110: 707-18.
Kwok RF, Lim J, Chan MT, Gin T, Chiu WK. Preoperative ketamine improves postoperative analgesia after gynecologic laparoscopic surgery. Anesth Analg 2004; 98: 1044-9.
Lahtinen P, Kokki H, Hakala T, Hynynen M. S(+)-ketamine as an analgesic adjunct reduces opioid consumption after cardiac surgery. Anesth Analg 2004; 99: 1295-301.
Lak M, Foroozanmehr MJ, Ramazani MA, Araghizadeh H, Zahedi-Shoolami L. Assessment of ketamine effect as adjuvant to morphine in post-operative pain reduction in donor kidney transplanted. Iranian Red Crescent Medical Journal 2010; 12: 38-44.
Lebrun T, Van Elstraete AC, Sandefo I, Polin B, Pierre-Louis L. Lack of a pre-emptive effect of low-dose ketamine on postoperative pain following oral surgery. Can J Anesth 2006; 53: 146-52.
Lehmann KA, Klaschik M. Lack of pre-emptive analgesic effect of low-dose ketamine in postoperative patients. A prospective, randomised double-blind study (German). Schmerz 2001; 15: 248-53.
Loftus RW, Yeager MP, Clark JA, et al. Intraoperative ketamine reduces perioperative opiate consumption in opiate-dependent patients with chronic back pain undergoing back surgery. Anesthesiology 2010; 113: 639-46.
McKay WP, Donais P. Bowel function after bowel surgery: morphine with ketamine or placebo; a randomized controlled trial pilot study. Acta Anaesthesiol Scand 2007; 51: 1166-71.
Menigaux C, Fletcher D, Dupont X, Guignard B, Guirimand F, Chauvin M. The benefits of intraoperative small-dose ketamine on postoperative pain after anterior cruciate ligament repair. Anesth Analg 2000; 90: 129-35.
Murdoch CJ, Crooks BA, Miller CD. Effect of the addition of ketamine to morphine in patient-controlled analgesia. Anaesthesia 2002; 57: 484-8.
Ogun CO, Duman A, Okesli S. The comparison of postoperative analgesic effects of preemptive ketamine and fentanyl use in mastectomy operations (Turkish). Agri 2001; 13: 31-40.
Pirim A, Karaman S, Uyar M, Certug A. Addition of ketamine infusion to patient controlled analgesia with intravenous morphine after abdominal hysterectomy (Turkish). Agri 2006; 18: 52-8.
Reeves M, Lindholm DE, Myles PS, Fletcher H, Hunt JO. Adding ketamine to morphine for patient-controlled analgesia after major abdominal surgery: a double-blinded, randomized controlled trial. Anesth Analg 2001; 93: 116-20.
Remerand F, Le Tendre C, Baud A, et al. The early and delayed analgesic effects of ketamine after total hip arthroplasty: a prospective, randomized, controlled, double-blind study. Anesth Analg 2009; 109: 1963-71.
Reza FM, Zahra F, Esmaeel F, Hossein A. Preemptive analgesic effect of ketamine in patients undergoing elective cesarean section. Clin J Pain 2010; 26: 223-6.
Roytblat L, Korotkoruchko A, Katz J, Glazer M, Greemberg L, Fisher A. Postoperative pain: the effect of low-dose ketamine in addition to general anesthesia. Anesth Analg 1993; 77: 1161-5.
Sahin A, Canbay O, Cuhadar A, Celebi N, Aypar U. Bolus ketamine does not decrease hyperalgesia after remifentanil infusion. Pain Clinic 2004; 16: 407-11.
Sen H, Sizlan A, Yanarates O, et al. A comparison of gabapentin and ketamine in acute and chronic pain after hysterectomy. Anesth Analg 2009; 109: 1645-50.
Snijdelaar DG, Cornelisse HB, Schmid RL, Katz J. A randomised, controlled study of peri-operative low dose s(+)-ketamine in combination with postoperative patient-controlled s(+)-ketamine and morphine after radical prostatectomy. Anaesthesia 2004; 59: 222-8.
Suzuki M, Tsueda K, Lansing PS, et al. Small-dose ketamine enhances morphine-induced analgesia after outpatient surgery. Anesth Analg 1999; 89: 98-103.
Unlugenc H, Gunduz M, Ozalevli M, Akman H. A comparative study on the analgesic effect of tramadol, tramadol plus magnesium, and tramadol plus ketamine for postoperative pain management after major abdominal surgery. Acta Anaesthesiol Scand 2002; 46: 1025-30.
Van Elstraete AC, Lebrun T, Sandefo I, Polin B. Ketamine does not decrease postoperative pain after remifentanil-based anaesthesia for tonsillectomy in adults. Acta Anaesthesiol Scand 2004; 48: 756-60.
Yamauchi M, Asano M, Watanabe M, Iwasaki S, Furuse S, Namiki A. Continuous low-dose ketamine improves the analgesic effects of fentanyl patient-controlled analgesia after cervical spine surgery. Anesth Analg 2008; 107: 1041-4.
Yentur EA, Topcu I, Keles GT, Tasyuz T, Sakarya M. Subanalgesic dose of ketamine added to tramadol does not reduce analgesic demand (Turkish). Turk Anesi Rean Der Dergisi 2004; 32: 106-12.
Abu-Shahwan I. Ketamine does not reduce postoperative morphine consumption after tonsillectomy in children. Clin J Pain 2008; 24: 395-8.
Aveline C, Hetet HL, Vautier P, Gautier JF, Bonnet F. Peroperative ketamine and morphine for postoperative pain control after lumbar disk surgery. Eur J Pain 2006; 10: 653-8.
Becke K, Albrecht S, Schmitz B, et al. Intraoperative low-dose S-ketamine has no preventive effects on postoperative pain and morphine consumption after major urological surgery in children. Pediatr Anesth 2005; 15: 484-90.
Burstal R, Danjoux G, Hayes C, Lantry G. PCA ketamine and morphine after abdominal hysterectomy. Anaesth Intensive Care 2001; 29: 246-51.
Butkovic D, Kralik S, Matolic M, Jakobovic J, Zganjer M, Radesic L. Comparison of a preincisional and postincisional small dose of ketamine for postoperative analgesia in children. Bratisl Lek Listy 2007; 108: 184-8.
Colombani S, Kabbani Y, Mathoulin-Pelissier S, et al. Administration of ketamine during induction and maintenance of anaesthesia in postoperative pain prevention (French). Ann Fr Anesth Reanim 2008; 27: 202-7.
Dal D, Celebi N, Elvan EG, Celiker V, Aypar U. The efficacy of intravenous or peritonsillar infiltration of ketamine for postoperative pain relief in children following adenotonsillectomy. Pediatr Anesth 2007; 17: 263-9.
Darwish HM, Marzouk S, El Kholy G, El-Din WS. Low dose ketamine prevents acute opioid tolerance induced by remifentanil infusion. Eg J Anaesth 2005; 21: 259-66.
Guignard B, Coste C, Costes H, et al. Supplementing desflurane-remifentanil anesthesia with small-dose ketamine reduces perioperative opioid analgesic requirements. Anesth Analg 2002; 95: 103-8.
Hayes C, Armstrong-Brown A, Burstal R. Perioperative intravenous ketamine infusion for the prevention of persistent post-amputation pain: a randomized, controlled trial. Anaesth Intensive Care 2004; 32: 330-8.
Joly V, Richebe P, Guignard B, et al. Remifentanil-induced postoperative hyperalgesia and its prevention with small-dose ketamine. Anesthesiology 2005; 103: 147-55.
Michelet P, Guervilly C, Helaine A, et al. Adding ketamine to morphine for patient-controlled analgesia after thoracic surgery: influence on morphine consumption, respiratory function, and nocturnal desaturation. Br J Anaesth 2007; 99: 396-403.
Murray WB, Yankelowitz SM, le Roux M, Bester HF. Prevention of post-tonsillectomy pain with analgesic doses of ketamine. S Afr Med J 1987; 72: 839-42.
O’Flaherty JE, Lin CX. Does ketamine or magnesium affect posttonsillectomy pain in children? Pediatr Anesth 2003; 13: 413-21.
Ozgun S, Ugur B, Aydin ON, Eyigor H, Erpek G. The effect of preemptive ketamine on analgesia and analgesic consuption after tonsillectomy. Turk Anest Rean Der Dergisi 2003; 31: 247-52.
Webb AR, Skinner BS, Leong S, et al. The addition of a small-dose ketamine infusion to tramadol for postoperative analgesia: a double-blinded, placebo-controlled, randomized trial after abdominal surgery. Anesth Analg 2007; 104: 912-7.
Zakine J, Samarcq D, Lorne E, et al. Postoperative ketamine administration decreases morphine consumption in major abdominal surgery: a prospective, randomized, double-blind, controlled study. Anesth Analg 2008; 106: 1856-61.
Ayoglu H, Karadeniz U, Kunduracilar Z, Ayoglu FN, Erdemli O. The analgesic effect of magnesium sulfate and ketamine in patients undergoing laparoscopic cholecystectomy. Pain Clinic 2005; 17: 45-53.
Darabi ME, Mireskandari SM, Sadeghi M, Salamati P, Rahimi E. Ketamine has no pre-emptive analgesic effect in children undergoing inguinal hernia repair. Acta Medica Iranica 2008; 46: 451-6.
Edwards ND, Fletcher A, Cole JR, Peacock JE. Combined infusions of morphine and ketamine for postoperative pain in elderly patients. Anaesthesia 1993; 48: 124-7.
Mebazaa MS, Mestiri T, Kaabi B, Ben Ammar MS. Clinical benefits related to the combination of ketamine with morphine for patient controlled analgesia after major abdominal surgery. Tunis Med 2008; 86: 435-40.
Unlugenc H, Ozalevli M, Guler T, Isik G. Postoperative pain management with intravenous patient-controlled morphine: comparison of the effect of adding magnesium or ketamine. Eur J Anaesthesiol 2003; 20: 416-21.
Wu YD, Li H, Xiong JC, et al. Effects of patient-controlled analgesia with small dose ketamine combined with morphine and the influence thereof on plasma beta-endorphin level in patients after radical operation for esophageal carcinoma (Chinese). Zhonghua Yi Xie Za Zhi 2009; 89: 314-7.
Acknowledgements
We gratefully acknowledge Dr. Raymond Yip for his contribution as third reviewer. We also thank the following individuals for their invaluable assistance with translation: Dr. Kotoo Meguro, Xiuli Gao, Dr. Fatih Al-Katib, Andrea Dwyer, Dr. Shefali Thakore, Dr. Guoming Xie, Dr. Richard Schaan, Dr. Suzan Akin, Dr. Zurab Ivanishvili, and Misil Jeong. Finally we thank Hitesh Bhatt for his statistical assistance with the analysis.
Disclosure
Study funded by research fund within the Department of Anesthesiology, Perioperative Medicine and Pain Management at the University of Saskatchewan. No affiliations, associations or consultancies.
Competing interests
None declared.
Author information
Authors and Affiliations
Corresponding author
Appendix: Search strategy (EMBASE, Medline)
Appendix: Search strategy (EMBASE, Medline)
-
1.
KETAMINE
-
2.
ketamine OR ketalar
-
3.
(1 or 2)
-
4.
PAIN POSTOPERATIVE
-
5.
(postoperat* near pain*)
-
6.
(pain* next following next surg*)
-
7.
(pain* next following next treat*)
-
8.
(pain* next following next operat*)
-
9.
postoperat* pain
-
10.
((post near surg*) or postsurg* OR post-surg*)
-
11.
((post near operat*) or postoperat* OR post-operat*)
-
12.
pain*
-
13.
(10 and 11)
-
14.
(12 and 13)
-
15.
(post-operat* near analgesi*)
-
16.
(postoperat* near analgesi*)
-
17.
(post-surg* near analgesi*)
-
18.
(postsurgi* near analgesi*)
-
19.
(analgesi* next following next surg*)
-
20.
(analgesi* next following next operat*)
-
21.
(4 or 5 or 6 or 7 or 8 or 9 or 14 or 15 or 16 or 17 or 18 or 19 or 20)
-
22.
(3 and 21)
Rights and permissions
About this article
Cite this article
Laskowski, K., Stirling, A., McKay, W.P. et al. A systematic review of intravenous ketamine for postoperative analgesia. Can J Anesth/J Can Anesth 58, 911–923 (2011). https://doi.org/10.1007/s12630-011-9560-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-011-9560-0