Abstract
Purpose
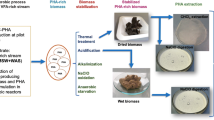
This paper presents an assessment of the production of polyhydroxyalkanoates (PHA) in biomass sourced from wastewater treatment plants (WWTPs). The purpose was to examine both the potential for more generic utilization of waste activated sludge as biomass for PHA production and the quality of the polymers produced.
Methods
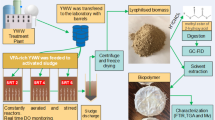
Biomass was sourced from two full-scale WWTPs. A refined acetic and propionic acid feedstock was used to produce a P(3HB-co-3HV) (polyhydroxy-butyrate-valerate or PHBV) copolymer in a single-stage from this biomass as-received.
Results
The mean overall storage yield was 0.25 ± 0.03 gCOD of polymer per gCOD feedstock consumed. The average PHA cell content was 0.26 ± 0.08 gPHA gVSS−1. Microstructure and thermal properties analysis showed that the polymers produced were most likely a mixture of random copolymers, with polymer composition being mainly a function of substrate composition. The PHA recovered was of high purity and exhibited thermal characteristics similar to published values.
Conclusions
Based on published data, the overall yield for a conventional PHA production system, utilizing volatile fatty acid (VFA) feedstocks for biomass and biopolymer production, is 0.19 gPHA per gVFA(COD), which is similar to that obtained in this work. In other words, efficiency of VFA feedstock utilization is similar. Considering activated sludge biomass is abundantly available around the world, this work suggests that industrial scale PHA production could be achieved more generally in conjunction with sludge management rather than using purpose specific biorefineries where biomass is grown on high value feedstocks.



Similar content being viewed by others

References
Doi, Y.: Biotechnology: unnatural biopolymers. Nat. Mater. 1, 207–208 (2002)
Lee, S.Y.: Deciphering bioplastic production. Nat. Biotech. 24, 1227–1229 (2006)
Bengtsson, S., Pisco, A.R., Reis, M.A.M., Lemos, P.C.: Production of polyhydroxyalkanoates from fermented sugar cane molasses by a mixed culture enriched in glycogen accumulating organisms. J. Biotechnol. 145, 253–263 (2010)
Dias, J.M.L., Lemos, P.C., Serafim, L.S., Oliveira, C., Eiroa, M., Albuquerque, M.G.E., et al.: Recent advances in polyhydroxyalkanoate production by mixed aerobic cultures: from the substrate to the final product. Macromol. Biosci. 6, 885–906 (2006)
Serafim, L., Lemos, P., Albuquerque, M., Reis, M.: Strategies for PHA production by mixed cultures and renewable waste materials. Appl. Microbiol. Biotechnol. 81, 615–628 (2008)
Albuquerque, M.G.E., Martino, V., Pollet, E., Avérous, L., Reis, M.A.M.: Mixed culture polyhydroxyalkanoate (PHA) production from volatile fatty acid (VFA)-rich streams: effect of substrate composition and feeding regime on PHA productivity, composition and properties. J. Biotechnol. 151, 66–76 (2011)
Albuquerque, M.G.E., Torres, C.A.V., Reis, M.A.M.: Polyhydroxyalkanoate (PHA) production by a mixed microbial culture using sugar molasses: effect of the influent substrate concentration on culture selection. Water Res. 44, 3419–3433 (2010)
Bengtsson, S., Werker, A., Christensson, M., Welander, T.: Production of polyhydroxyalkanoates by activated sludge treating a paper mill wastewater. Bioresour. Technol. 99, 509–516 (2008)
Johnson, K., Jiang, Y., Kleerebezem, R., Muyzer, G., van Loosdrecht, M.C.: Enrichment of a mixed bacterial culture with a high polyhydroxyalkanoate storage capacity. Biomacromolecules 10, 670–676 (2009)
Johnson, K., Kleerebezem, R., van Loosdrecht, M.C.M.: Influence of ammonium on the accumulation of polyhydroxybutyrate (PHB) in aerobic open mixed cultures. J. Biotechnol. 147, 73–79 (2010)
Chua, H., Yu, P., Ma, C.: Accumulation of biopolymers in activated sludge biomass. Appl. Biochem. Biotechnol. 78, 389–399 (1999)
Mengmeng, C., Hong, C., Qingliang, Z., Shirley, S.N., Jie, R.: Optimal production of polyhydroxyalkanoates (PHA) in activated sludge fed by volatile fatty acids (VFAs) generated from alkaline excess sludge fermentation. Bioresour. Technol. 100, 1399–1405 (2009)
Laycock, B., Halley, P., Pratt, S., Werker, A., Lant, P.: The chemomechanical properties of microbial polyhydroxyalkanoates. Prog. Polym. Sci. (2012). http://dx.doi.org/10.1016/j.progpolymsci.2012.06.003
Gurieff, N.: Production of biodegradable Polyhydroxyalkanoate polymers using advanced biological wastewater treatment process technology. University of Queensland, Brisbane (2007)
Serafim, L.S., Lemos, P.C., Oliveira, R., Reis, M.A.M.: Optimization of polyhydroxybutyrate production by mixed cultures submitted to aerobic dynamic feeding conditions. Biotechnol. Bioeng. 87, 145–160 (2004)
Arcos-Hernandez, M.V., Gurieff, N., Pratt, S., Magnusson, P., Werker, A., Vargas, A., et al.: Rapid quantification of intracellular PHA using infrared spectroscopy: an application in mixed cultures. J. Biotechnol. 150, 372–379 (2010)
Oehmen, A., Keller-Lehmann, B., Zeng, R.J., Yuan, Z., Keller, J.: Optimisation of poly-β-hydroxyalkanoate analysis using gas chromatography for enhanced biological phosphorus removal systems. J. Chromatogr. A 1070, 131–136 (2005)
Werker, A., Lind, P., Bengtsson, S., Nordström, F.: Chlorinated-solvent-free gas chromatographic analysis of biomass containing polyhydroxyalkanoates. Water Res. 42, 2517–2526 (2008)
APHA: Standard methods for the examination of water and wastewater. American Public Health Association, Washington (1995)
Dong, Z., Sun, X.: A new method of recovering polyhydroxyalkanoate from Azotobacter chroococcum. Chin. Sci. Bull. 45, 252–256 (2000)
Kim, M., Cho, K.S., Ryu, H.W., Lee, E.G., Chang, Y.K.: Recovery of poly(3-hydroxybutyrate) from high cell density culture of Ralstonia eutropha by direct addition of sodium dodecyl sulfate. Biotechnol. Lett. 25, 55–59 (2003)
Hahn, S.K., Chang, Y.K., KimBeom, S., Chang, H.N.: Optimization of microbial poly(3-hydroxybutyrate) recover using dispersions of sodium hypochlorite solution and chloroform. Biotechnol. Bioeng. 44, 256–261 (1994)
Systat Software: PeakFit. 4.00 ed (1991)
Dai, Y., Lambert, L., Yuan, Z., Keller, J.: Characterisation of polyhydroxyalkanoate copolymers with controllable four-monomer composition. J. Biotechnol. 134, 137–145 (2008)
Beun, J.J., Dircks, K., Van Loosdrecht, M.C.M., Heijnen, J.J.: Poly-β-hydroxybutyrate metabolism in dynamically fed mixed microbial cultures. Water Res. 36, 1167–1180 (2002)
Dionisi, D., Majone, M., Papa, V., Beccari, M.: biodegradable polymers from organic acids by using activated sludge enriched by aerobic periodic feeding. Biotechnol. Bioeng. 85, 569–579 (2004)
Dionisi, D., Carucci, G., Papini, M.P., Riccardi, C., Majone, M., Carrasco, F.: Olive oil mill effluents as a feedstock for production of biodegradable polymers. Water Res. 39, 2076–2084 (2005)
Bosco, F., Chiampo, F.: Production of polyhydroxyalconates (PHAs) using milk whey and dairy wastewater activated sludge: production of bioplastics using dairy residues. J. Biosci. Bioeng. 109, 418–421 (2010)
Lemos, P.C., Serafim, L.S., Reis, M.A.M.: Synthesis of polyhydroxyalkanoates from different short-chain fatty acids by mixed cultures submitted to aerobic dynamic feeding. J. Biotechnol. 122, 226–238 (2006)
Serafim, L.S., Lemos, P.C., Torres, C., Reis, M.A.M., Ramos, A.M.: The influence of process parameters on the characteristics of polyhydroxyalkanoates produced by mixed cultures. Macromol. Biosci. 8, 355–366 (2008)
Reis, M.A.M., Serafim, L.S., Lemos, P.C., Ramos, A.M., Aguiar, F.R., Van Loosdrecht, M.C.M.: Production of polyhydroxyalkanoates by mixed microbial cultures. Bioprocess Biosyst. Eng. 25, 377–385 (2003)
Bengtsson, S., Pisco, A.R., Johansson, P., Lemos, P.C., Reis, M.A.M.: Molecular weight and thermal properties of polyhydroxyalkanoates produced from fermented sugar molasses by open mixed cultures. J. Biotechnol. 147, 172–179 (2010)
Morgan-Sagastume, F., Karlsson, A., Johansson, P., Pratt, S., Boon, N., Lant, P., et al.: Production of polyhydroxyalkanoates in open, mixed cultures from a waste sludge stream containing high levels of soluble organics, nitrogen and phosphorus. Water Res. 44, 5196–5211 (2010)
Patel, M., Gapes, D., Newman, R., Dare, P.: Physico-chemical properties of polyhydroxyalkanoate produced by mixed-culture nitrogen-fixing bacteria. Appl. Microbiol. Biotechnol. 82, 545–555 (2009)
Pederson, E.N., McChalicher, C.W.J., Srienc, F.: Bacterial synthesis of PHA block copolymers. Biomacromolecules 7, 1904–1911 (2006)
Yoshie, N., Inoue, Y.: Chemical composition distribution of bacterial copolyesters. Int. J. Biol. Macromol. 25, 193–200 (1999)
Liu, T., Petermann, J.: Multiple melting behavior in isothermally cold-crystallized isotactic polystyrene. Polymer 42, 6453–6461 (2001)
Kamiya, N., Yamamoto, Y., Inoue, Y., Chujo, R., Doi, Y.: Microstructure of bacterially synthesized poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Macromolecules 22, 1676–1682 (1989)
Kamiya, N., Inoue, Y., Yamamoto, Y., Chujo, R., Doi, Y.: Conformational analysis of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) in solution by proton NMR spectroscopy. Macromolecules 23, 1313–1317 (1990)
Doi, Y., Kunioka, M., Nakamura, Y., Soga, K.: Nuclear magnetic resonance studies on poly(β-hydroxybutyrate) and a copolyester of β-hydroxybutyrate andβ-hydroxyvalerate isolated from Alcaligenes eutrophus H16. Macromolecules 19, 2860–2864 (1986)
Žagar, E., Kržan, A., Adamus, G., Kowalczuk, M.: Sequence distribution in microbial poly(3-hydroxybutyrate-co-3-hydroxyvalerate) co-polyesters determined by NMR and MS. Biomacromolecules 7, 2210–2216 (2006)
Gurieff, N., Lant, P.: Comparative life cycle assessment and financial analysis of mixed culture polyhydroxyalkanoate production. Bioresour. Technol. 98, 3393–3403 (2007)
Acknowledgments
The School of Chemical Engineering (UQ) for the tuition fee waiver scholarship and top-up scholarship and Consejo Nacional de Ciencia y Tecnologia (CONACYT) of Mexico for the scholarship granted for postgraduate studies (303754). Australian Research Council for funding through grant ARC LP0990917. From UQ: L. Lambert, B. Keller-Lehman, K. M. Leung and S. Cooke. From Queensland University of Technology (QUT): M. Nikolić, B. Radi and E. Martinez. From Anoxkaldnes: L. Karabegovic, F. Morgan-Sagástume, S. Bengtsson, A. Karlsson and P. Magnusson. From Veolia Environment: D. Cirne and E. Blanchet.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Extraction Method with Sodium Dodecyl Sulphate of PHA from Mixed Cultures
Immediately after accumulation, biomass was concentrated by centrifugation (3,500 rpm, 30 min) to obtain a cake. Non-PHA mass content was estimated as the difference between the weights of the estimated content of TSS in the concentrated cake and the estimated PHA mass content from IR spectra of biomass at the end of accumulation [16].
One gram of sodium dodecyl sulphate (SDS) was used for each 0.72 g of non-PHA material [21] and dissolved in distilled water to 10 g L−1 concentration [20]. The concentrated biomass was then dissolved in this solution, and reacted for 40 min at 55 °C with powerful magnetic stirring. The solid phase was separated from solution by centrifugation (3,500 rpm, 30 min), and then washed with distilled water, resuspended and centrifuged again. After this treatment a thin layer of polymer over the non-PHA material was observed. Further treatment with chloroform was used to achieve better recovery [22].
The washed pellet was re-suspended in chloroform; 50 mL of CHCl3 per 1 g of estimated TSS content was used. The dispersion was incubated at 37 °C for 4 h with powerful magnetic stirring (700 rpm). The chloroform phase was then separated by settling, aided by centrifugation of the dispersion if needed. The chloroform phase was then concentrated until a polymer film was observed. A small volume of methanol was added to induce precipitation and purification. The purified films were then separated from the excess methanol by vacuum filtration and dried for 6 h at 70 °C, or until constant weight, and stored in a desiccator for further analysis (see Table 5).
Rights and permissions
About this article
Cite this article
Arcos-Hernandez, M.V., Pratt, S., Laycock, B. et al. Waste Activated Sludge as Biomass for Production of Commercial-Grade Polyhydroxyalkanoate (PHA). Waste Biomass Valor 4, 117–127 (2013). https://doi.org/10.1007/s12649-012-9165-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-012-9165-z