Abstract
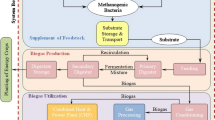
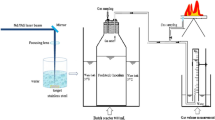
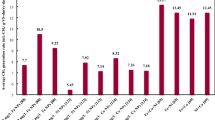
This study investigated the simultaneous effects of nanoparticles (NPs) addition to rumen fluid (archaea source) and laser irradiation of the mixture on biogas production from anaerobic digestion of dairy manure. Where, the previous study reported that the addition of 2 mg L−1 nickel nanoparticles (Ni NPs) significantly (p < 0.05) increased the biogas and methane volumes by 1.74 and 2.01 times compared to the control, respectively. The results indicated that the most efficient irradiation time was 2 h laser with the addition of 2 mg L−1 Ni NPs (p < 0.05), which minimized the lag phase from 4 days to 1 day and the Hydraulic Retention Time (HRT) to attain the peak of biogas production in comparison to the control from 28 days to 16 days. The combination of laser irradiation and nanoparticles addition yielded the highest significant value of the specific biogas and methane production, compared to all treatments (incandescent light, control), which were 679.5 mL biogas g−1 VS and 453.3 mL CH4 g−1 VS. Furthermore, Laser photocatalysis of Ni NPs enhances the photo-reduction/photo-oxidation of CH4 formation pathways. Consequently, this treatment increased the biogas and methane volumes by 1.9 and 2.32 times the biogas and methane volumes resulted from the control, respectively.






Similar content being viewed by others
References
Feng, Y., Zhang, Y., Quan, X., Chen, S.: Enhanced anaerobic digestion of waste activated sludge digestion by the addition of zero valent iron. Water Res. 52, 242–250 (2014)
Slimane, K., Fathya, S., Assia, K., Hamza, M.: Influence of inoculums/substrate ratios (ISRs) on the mesophilic anaerobic digestion of slaughterhouse waste in batch mode: process stability and biogas production. Energy Procedia 50, 57–63 (2014)
El-Mashad, H.M., Van Loon, K.P., Wilko, G., Zeeman, G.P.A., Bot, G., Lettinga, G.: Reuse potential of agricultural wastes in semi-arid regions: Egypt as a case study. Environ. Sci. Technol. 2, 53–66 (2003)
Ravuri, H.K.: Role of factors influencing on anaerobic process for production of bio hydrogen. Future Fuel. Int. J. Adv. Chem. 1(2), 31–38 (2013)
Amaya, O.M., Barragán, M.T.C., Tapia, F.J.A.: Microbial biomass in batch and continuous system, biomass now. In: Matovic, M. D. (ed.) Sustainable Growth and Use. InTech, London (2013). ISBN 978-953-51-1105-4, https://doi.org/10.5772/55303
Chen, J.L., Ortiz, R., Steele, T.W.J., Stuckey, D.C.: Toxicants inhibiting anaerobic digestion: a review. Biotechnol. Adv. 32, 1523–1534 (2014)
Tada, C., Sawayama, S.: Photoenhancement of biogas production from thermophilic anaerobic digestion. J. Biosci. Bioeng. 98, 387–390 (2004)
Abdelsalam, E.: Application of laser and nanotechnology to increase biogas production. PhD dissertation, National Institute of Laser Enhanced Sciences (NILES), Cairo University, Egypt (2015)
Olson, K.D., Mcmathon, C.W., Wolfe, R.S.: Light sensitivity of methanogenic archaebacteria. Appl. Environ. Microbiol. 57, 2683–2686 (1991)
Fedoseyeva, G.E., Karu, T.I., Lyapunova, T.S., Pomoshnikova, N.A., Meissel, M.N.: The activation of yeast metabolism with He–Ne laser radiation. I. Protein synthesis in various cultures. Lasers Life Sci. 2, 137–146 (1988)
Fedoseyeva, G.E., Karu, T.I., Lyapunova, T.S., Pomoshnikova, N.A., Meissel, M.N.: The activation of yeast metabolism with He-Ne laser. II. Activity of enzymes of oxidative and phosphorus metabolism. Lasers Life Sci. 2, 147–154 (1988)
Karu, T.I.: Effects of visible radiation on cultured cells. Photochem. Photobiol. B 52, 1089–1098 (1990)
Tiphlova, O.A., Karu, T.I.: Effect of argon laser radiation and noncoherent blue light on Escherichia coli. Radiobiologia 26, 829–832 (1986)
Karu, T.I., Tiphlova, O.A., Letokhov, V.S., Lobko, V.V.: Stimulation of E. coli growth by laser and incoherent red light. Nuovo Cimento D 2, 1138–1144 (1983)
Argun, H., Kargi, F.: Effects of light source, intensity and lighting regime on bio-hydrogen production from ground wheat starch by combined dark and photo-fermentations. Int. J. Hydrogen Energy 35, 1604–1612 (2010)
Olson, K.D., Mcmathon, C.W., Wolfe, R.S.: Photoactivation of the 2-(methylthio)ethanesulfonic acid reductase from Methanobacterium. Proc. Natl. Acad. Sci. USA 88: 4013–4099 (1991)
Ellermann, J., Hedderich, R., Bocher, R., Thauer, R.K.: The final step in methane formation. Investigations with highly purified methyl-CoM reductase (component C) from Methanobacterium thermoautotrophicum (strain Marburg). Eur. J. Biochem. 172, 669–677 (1988)
Karu, T., Pyatibrat, L., Kalendo, G.: Irradiation with He-Ne laser increases ATP level in cells cultivated in vitro. Photochem. Photobiol. B 27, 219–223 (1995)
Kato, M., Shmzawa, K., Yoshikawa, S.: Cytochrome oxidase is a possible photoreceptor in mitochondria. Photochem. Photobiol. B 2, 263–269 (1981)
Vekshm, N.L., Mironov, G.P.: Flavin-dependent oxygen uptake in mitochondria under illumination. Biofizika 27, 537–539 (1982)
Passarella, S., Casamassima, F., Molmari, S., Pastore, D., Quagliariello, E., Catalano, I.M., Cmgolam, A.: Increase of proton electrochemical potential and ATP synthesis in rat liver mitochondria irradiated in vitro by He-Ne laser. FEBS Lett. 175, 95–99 (1984)
Passarella, S., Perlmo, E., Quagliariello, E., Baldassarre, L., Catalano, I.M., Cmgolam, A.: Evidence of changes induced by He-Ne laser irradiation in the biochemical properties of rat liver mitochondria. Bioelectrochem. Bioenerg. 70, 185–198 (1983)
Passarella, S., Ostum, A., Atlante, A., Quagliariello, E.: Increase in the ADP/ATP exchange in rate liver mitochondria irradiated in vitro by He-Ne laser. Biochem. Biophys. Res. Commun. 156, 978–986 (1988)
Karu, T.I.: Primary and secondary mechanisms of action of visible to Near-IR radiation on cells. Photochem. Photobiol. B 49, 1–17 (1999)
Pastore, D., Di Martino, C., Bosko, G., Passarella, S.: Stimulation of ATP synthesis via oxidative phosphorylation in wheat mitochondria irradiated with helium-neon laser. Biochem. Mol. Biol. Int. J. 39, 149–157 (1996)
Chhabra, A., Manjunath, K., Panigrahy, S., Parihar, J.: Spatial pattern of methane emissions from Indian livestock. Curr. Sci. 96(5), 683–692 (2009)
Nusbaum, N.J.: Dairy livestock methane remediation and global warming. J. Commun. Health 35(5), 500–502 (2010)
Ma, J., Zhao, Q.B., Laurens, L.L.M., Jarvis, E.E., Nagle, N.J., Chen, S., Frear, C.S.: Mechanism, kinetics and microbiology of inhibition caused by long-chain fatty acids in anaerobic digestion of algal biomass. Biotechnol. Biofuels 8, 141 (2015)
Yıldırım, E., Ince, O., Aydin, S., Ince, B.: Improvement of biogas potential of anaerobic digesters using rumen fungi. Renew. Energy 109, 346–353 (2017)
Budiyono, B., Widiasa, I.N., Johari, S., Sunarso, S.: Influence of inoculum content on performance of anaerobic reactors for treating cattle manure using rumen fluid inoculum. Int. J. Eng. Technol. 1(3), 109–116 (2009)
Sunarso, S., Johari, S., Widiasa, I.N., Budiyono, B.: The effect of feed to inoculums ratio on biogas production rate from cattle manure using rumen fluid as inoculums. Int. J. Sci. Eng. 1(2), 41–45 (2010)
Qiang, H., Lang, D.-L., Li, Y.-Y.: High-solid mesophilic methane fermentation of food waste with an emphasis on iron, cobalt, and nickel requirements. Bioresour. Technol. 103, 21–27 (2012)
Abdelsalam, E., Samer, M., Abdel-Hadi, M.A., Hassan, H.E., Badr, Y.: Effects of CoCl2, NiCl2 and FeCl3 additives on biogas production. Misr J. Agric. Eng. 32(2), 843–862 (2015)
Gustavsson, J., Yekta, S.S., Sundberg, C., Karlsson, A., Ejlertsson, J., Skyllberg, U., et al.: Bioavailability of cobalt and nickel during anaerobic digestion of sulfurrich stillage for biogas formation. Appl. Energy 112, 473–477 (2013)
Facchin, V., Cavinato, C., Fatone, F., Pavan, P., Cecchi, F., Bolzonella, D.: Effect of trace element supplementation on the mesophilic anaerobic digestion of food waste in batch trials: the influence of inoculum origin. Biochem. Eng. J. 70, 71–77 (2013)
Parisi, C., Vigani, M., Rodríguez-Cerezo, E.: Agricultural nanotechnologies: what are the current possibilities? Nano Today 10(2), 124–127 (2015)
Powell, J.J., Faria, N., Thomas-McKay, E., Pele, L.C.: Origin and fate of dietary nanoparticles and microparticles in the gastrointestinal tract. J. Autoimmun. 34(3), J226–J233 (2010)
Nassar, N.N.: Rapid removal and recovery of Pb(II) from wastewater by magnetic nanoadsorbents. J. Hazard. Mater. 184, 538–546 (2010)
Nassar, N.N.: Iron oxide nanoadsorbents for removal of various pollutants from wastewater: an overview. In: Bhatnagar, A. (ed.), Application of Adsorbents for Water Pollution Control. Bentham Science Publishers, Oak Park (2012)
Zhang, W.: Nanoscale iron particles for environmental remediation: an overview. J. Nanopart. Res. 5, 323–332 (2003)
Abdelsalam, E., Samer, M., Attia, Y., Abdel-Hadi, M.A., Hassan, H.E., Badr, Y.: Comparison of nanoparticles effects on biogas and methane production from anaerobic digestion of cattle dung slurry. Renew. Energ. 87(1), 592–598 (2016)
Abdelsalam, E., Samer, M., Attia, Y., Abdel-Hadi, M.A., Hassan, H.E., Badr, Y.: Influence of zero valent iron nanoparticles and magnetic iron oxide nanoparticles on biogas and methane production from anaerobic digestion of manure. Energy 120, 842–853 (2017)
Abdelsalam, E., Samer, M., Attia, Y., Abdel-Hadi, M.A., Hassan, H.E., Badr, Y.: Effects of Co and Ni nanoparticles on biogas and methane production from anaerobic digestion of slurry. Energ. Convers. Manag. 14, 108–119 (2017)
Subramanyam, K., Sreelekha, N., Amaranatha Reddy, D., Murali, G., Rahul Varma, K., Vijayalakshmi, R.P.: Chemical synthesis, structural, optical, magnetic characteristics and enhanced visible light active photocatalysis of Ni doped CuS nanoparticles. Solid State Sci. 65, 68–78 (2017)
Yamada, Y., Miyahigashi, T., Kotani, H., Ohkubo, K., Fukuzum, S.: Photocatalytic hydrogen evolution with Ni nanoparticles by using 2-phenyl-4-(1-naphthyl)quinolinium ion as a photocatalyst. Energy Environ. Sci. 5, 6111–6118 (2012)
EPA: Total, Fixed, and Volatile Solids. Method 1684, January 2001, p. 20460. U.S. Environmental protection Agency, Engineering and Analysis Division (4303), Washington, DC (2001)
Cuetos, M.J., Fernández, C., Gómez, X., Morán, A.: Anaerobic co-digestion of swine manure with energy crop residues. Biotechnol. Bioprocess Eng. 16, 1044–1052 (2011)
Samer, M.: A software program for planning and designing biogas plants. Trans. ASABE 53(4), 1277–1285 (2010)
Samer, M.: Biogas plant constructions. In: Kumar, S. (ed.) Biogas, pp. 343–368. InTech, Rijeka (2012). ISBN 978-953-51-0204-5, https://doi.org/10.5772/31887
Yang, Y., Tsukahara, K., Zhang, Z., Sugiura, N., Sawayama, S.: Optimization of illumination time for the production of methane using carbon felt fluidized bed bioreactor in thermophilic anaerobic digestion. Biochem. Eng. J. 44, 131–135 (2009)
Snedecor, G.W., Cochran, W.G.: Statistical Methods, 8th edn. Iowa State University Press, Ames (1989)
Abdelsalam, E., Samer, M., Abdel-Hadi, M.A., Hassan, H.E., Badr, Y.: The effect of buffalo dung treatment with paunch fluid on biogas production. Misr J. Agric. Eng. 32(2), 807–826 (2015)
Lee, S.S., Choi, C.K., Ahna, B.H., Moonb, Y.H.: In vitro stimulation of rumen microbial fermentation by a rumen anaerobic fungal culture. Anim. Feed Sci. Technol. 115, 215–226 (2004)
Hu, Z.-H., Yu, H.-Q.: Anaerobic digestion of cattail by rumen cultures. Waste Manag. 26, 1222–1228 (2006)
Daquiado, A.R., Cho, K.M., Kim, T.Y., Kim, S.C., Chang, H.-H., Lee, Y.B.: Methanogenic archaea diversity in Hanwoo (Bos taurus coreanae) rumen fluid, rectal dung, and barn floor manure using a culture independent method based on mcrA gene sequences. Anaerobe 27, 77–81 (2014)
Bożym, M., Florczak, I., Zdanowska, P., Wojdalski, J., Klimkiewicz, M.: An analysis of metal concentrations in food wastes for biogas production. Renewable Energy 77, 467–472 (2015)
Tada, C., Tsukahara, K., Sawayama, S.: Illumination enhances methane production from thermophilic anaerobic digestion. Appl. Microbiol. Biotechnol. 71, 363–368 (2006)
Tayade, U.S., Borse, A.U., Meshram, J.S.: First report on butea monosperma flower extract based nickel nanoparticles green synthesis and characterization. Int. J. Sci. Res. Sci. Eng. Technol. 4(3), 43–49 (2018). http://ijsrset.com/IJSRSET184312.php
Ferry, J.G.: How to make a living by exhaling methane. Annu. Rev. Microbiol. 64, 453–473 (2010)
Rodionov, D.A., Hebbeln, P., Gelfand, M.S., Eitinger, T.: Comparative and functional genomic analysis of prokaryotic nickel and cobalt uptake transporters: evidence for a novel group of ATP-binding cassette transporters. J. Bacteriol. 188, 317–327 (2006)
Zhang, Y., Rodionov, D.A., Gelfand, M.S., Gladyshev, V.N.: Comparative genomic analyses of nickel, cobalt and vitamin B12 utilization. BMC Genomics. 10, 1–26 (2009). https://doi.org/10.1186/1471-2164-10-78
Lin, D.G., Nishio, N., Mazumder, T.K., Nagai, S.: Influence of Co2+, Ni2+ and Fe2+ on the production of tetrapyrroles by Methanosarcina barkeri. Appl. Microbiol. Biotechnol. 30, 196–200 (1989)
Kavitha, S., Kannah, R.Y., Ick Tae, Y., Khac-Uan, D.: J. Rajesh Banu, Combined thermo-chemo-sonic disintegration of waste activated sludge for biogas production. Biores. Technol. 197, 383–392 (2015)
Acknowledgements
This study was conducted at the National Institute of Laser Enhanced Sciences (NILES), and funded by Cairo University, Egypt. Therefore, we acknowledge NILES and Cairo University. Sincere gratitude to Dr. Ibrahim Yacoub, Assistant Professor of Agronomy at the Faculty of Agriculture (Cairo University) for his hard work in performing the statistical analysis.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Abdelsalam, E.M., Samer, M., Attia, Y.A. et al. Effects of Laser Irradiation and Ni Nanoparticles on Biogas Production from Anaerobic Digestion of Slurry. Waste Biomass Valor 10, 3251–3262 (2019). https://doi.org/10.1007/s12649-018-0374-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-018-0374-y