Abstract
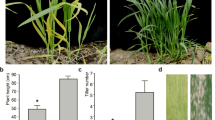
Chlorophyll-deficiency mutants are useful as genetic markers and as materials for studying the photosynthesis process. We characterized the inheritance of the gene controlling an opaque leaf (OL) trait in mungbean (Vigna radiata (L.) Wilczek). An F2 population was developed from the cross between the OL mutant and ‘Berken’, an Australian mungbean cultivar, to study inheritance and molecular tagging. The population was advanced by selfing to produce F5 lines from which two normal lines and two OL lines were randomly chosen to study chlorophyll content, seed growth and development, and seed cell morphology. The chlorophyll content in opaque leaf was lower than normal and thus was expected to have lower photosynthetic activity. This resulted in yellowish and shrinking pods and seeds within 15 days after flowering, while those from normal plants extended their growth up to 18 days. The cotyledon transfer cells of the OL plants deformed at 12 days and deteriorated at 14 days after flowering. The OL trait was controlled by a single recessive ol gene which was independent from the genes controlling petiole color and growth habit. We used 193 AFLP primer combinations to tag this gene and found that the marker AGG/ATA was linked with the ol gene at a distance of 3.4 cM.
Similar content being viewed by others
References
Ahloowalia BS, Maluszynski M. 2001. Induced mutations — A new paradigm in plant breeding. Euphytica 118: 167–173
Bahl JR, Gupta PK. 1982. Chlorophyll mutations in mung bean (Vigna radiata (L.) Wilczek). Theor. Appl. Genet. 63: 23–26
Boyer C. 1996. Biochemical genetics of carbohydrate metabolism in source and sink tissue. In E Zamski, AA Schaffer, ed, Photoassimilate Distribution in Plants and Crops, Source-Sink Relationship, Marcel Dekker, New York, pp 341–367
Brooks RM, Bradley MV, Anderson TI. 1950. Plant Microtechnique Manual, University of California, Davis
Corbineau F, Picard MA, Fougereux JA, Ladonne F, Côme D. 2000. Effects of dehydration conditions on desiccation tolerance of developing pea seeds as related to oligosaccharide content and cell membrane properties. Seed Sci. 10: 329–339
Dahiya BS. 1973. Improvement of mungbean through induced mutations. Indian J. Genet. Plant Breed. 33: 460–468
Duthion C, Pigeaive A. 1991. Seed lengths corresponding to the final stage in seed abortion of three grain legumes. Crop Sci. 31: 1579–1583
Fraser J, Egli DB, Leggett JE. 1982. Pod and seed development in soybean cultivars with differences in seed size. Agron. J. 74: 81–85
Gupta PK. 1996. Mutation Breeding in Mungbean. In AN Asthana, DH Kim, ed, Recent Advances in Mungbean Research, Indian Society of Pulses Research, IIPR, Kanpur, pp 124–136
Hermsen JG, Ramanna T, Vogel J. 1973. The location of a recessive gene for chlorophyll deficiency in diploid Solanum tuberosum by means of trisomic analysis. Can. J. Genet. Cytol. 15: 807–813
Keck RW, Dilley RA, Ke B. 1970. Photochemical characteristics in a soybean mutant. Plant Physiol. 46: 699–704
Klindworth DL, Williams ND, Duysen M.E. 1995. Genetic analysis of Chlorina mutants of durum wheat. Crop Sci. 35: 431–436
Kosambi DD. 1944. The estimation of map distance from recombination values. Ann. Eugen. 12: 172–175
Lambrides CJ, Lawn RJ, Godwin ID, Manners J, Imrie BC. 2000. Two genetic linkage maps of mungbean using RFLP and RAPD markers. Aust. J. Agric. Res. 51: 415–425
Lander ES, Green P, Abrahamson J, Barlow A, Daly MJ, Lincoln SE, Newburg L. 1987. Mapmaker: An interactive computer package for constructing primary genetic linkage maps of experimental and natural populations. Genomics 1: 174–181
Le Deunff Y, Rachidian Z. 1998. Interruption of water delivery at physiological maturity is essential for seed development, germination and seedling growth in pea. J. Exp. Bot. 39: 1221–1230
Mather K. 1951. The Measurement of Linkage in Heredity, John Wiley and Sons, Inc., New York
Moran R. 1982. Formulae for determination of chlorophyll pigments extracted with N,N-dimethylformamide. Plant Physiol. 69: 1376–1381
Nair SK, Tomar SMS. 2001. Genetical and anatomical analyses of a leaf flecking mutant in Triticum aestivum L. Euphytica 121: 53–58
Nelson OE. 1967. Biochemical genetics of higher plants. Annu. Rev. Genet. 1: 245–268
Ney B, Duthion C, Fontaine E. 1993. Timing of reproductive abortions in relation to cell division, water content, and growth of pea seeds. Crop Sci. 33: 267–270
Nobel PS. 2000. Plant Physiology, Academic Press, San Diego, 1999
Rascher U, Liebig M, Lüttge U. 2000. Evaluation of instant light-response curves of chlorophyll fluorescence parameters obtained with a portable chlorophyll fluorometer on site in the field. Plant Cell Environ. 23: 1397–1405
Sangsiri C, Sorajjapinun W, Srinives P. 2005. Gamma radiation induced mutations in mungbean. Sci. Asia 31: 251–255
SAS. 1989. SAS/STAT Guide for Personal Computers. Version 6. Cary: SAS Institute
Specht JE, Haskins FA, Gorz HJ. 1975. Contents of chlorophyll a and b in chlorophyll-deficient mutants of sweet clover. Crop Sci. 15: 851–853
Srinives P, Hual-alai N, Saengchot S, Ngampongsai S. 2000. The use of wild relatives and gamma radiation in mung bean and blackgram breeding. Proc. 7th MAFF Int. Workshop Genet. Res. Part 1. Wild Legumes, AFFRC and NIAR, Japan, pp 205–218
Subramanian D. 1980. Effect of gamma radiation on Vigna. Indian J. Genet. Plant Breed. 40: 187–194
Tekrony DM, Egli DB, Balles J, Pfeiffer T, Fellows RJ. 1979. Physiological maturity in soybean. Agron. J. 71: 771–775
Vogelmann TC, Evans JR. 2002. Profiles of light absorption and chlorophyll within spinach leaves from chlorophyll fluorescence. Plant Cell Environ. 25: 1313–1323
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabeau M. 1995. AFLP: A new technique for DNA fingerprinting. Nucleic Acids Res. 23: 4407–4414
Weber H, Heim U, Golombek S, Borisjuk L, Wobus U. 1998. Assimilate uptake and the regulation of seed development. Seed Sci. Res. 8: 331–345
Wettstein DV. 1961. Nuclear and cytoplasmic factors in development of protoplast structure and function. Can. J. Bot. 39: 1537–1545
Williams ND, Joppa LR, Duysen ME, Freeman TP. 1985. Inheritance of three chlorophyll-deficient mutants of common wheat. Crop Sci. 5: 1023–1025
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rungnoi, O., Chanprem, S., Toojinda, T. et al. Characterization, inheritance, and molecular study of opaque leaf mutant in mungbean (Vigna radiata (L.) Wilczek). J. Crop Sci. Biotechnol. 13, 219–226 (2010). https://doi.org/10.1007/s12892-010-0059-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-010-0059-y