Abstract
Several species of the family Orchidaceae have limited distribution and small populations, in which the probability of extinction is much higher than in broadly distributed species. As orchid seeds are very small and lack energy reserves, they require association with compatible mycorrhizal fungi that provide nutrients and carbon needed for germination and early development. Therefore, identification of putative orchid mycorrhizal partners is essential for the design of conservation strategies for these species. We investigated the mycorrhizal associations of two endangered rare species of orchids, Bipinnula apinnula and B. volckmannii, and compared our results with the mycorrhizal associations of two broadly distributed species of Bipinnula. We isolated mycorrhizal fungi and directly amplified DNA from colonized root segments and found an extremely low diversity of mycorrhizal fungi. The rarefaction curves of rare species populations saturated at a single OTU, showing a highly specialized association and considerably lower diversity than the other two species of Bipinnula. Nevertheless, symbiotic germination experiments with fungal strains of this specific fungal OTU only reached the stage of seed coat rupture in both orchid species, without formation of protocorms. Our results suggest that seeds may require additional treatment for successful germination and that fungal partners isolated from adult plants may not be the same that support the germination of seeds. Further studies are needed to improve germination of thiese orchid species and to contribute to their conservation.




Similar content being viewed by others
References
Arditti J, Ghani A (2000) Tansley review no. 110. Numerical and physical properties of orchid seeds and their biological implications. New Phytol 145:367–421 https://www.jstor.org/stable/2588806
Barthlott W, Grobe-Veldmann B, Korotkova N (2014) Orchid seed diversity: a scanning electron microscopy survey. Berlin: Englera 32, Botanic Garden and Botanical Museum Berlin-Dahlem
Batty AL, Dixon KW, Brundrett MC, Sivasithamparam K (2002) Orchid conservation and Mycorrhizal associations. In: K. Sivasithamparam, K.W. Dixon and R.L. Barrett eds. Microorganisms in plant conservation and biodiversity., the Netherlands: Kluwer academic publishers, Dordrecht, pp 195–226
Chase MW, Cameron KM, Barrett RL, Freudestein JV (2003) DNA data and Orchidaceae systematics: a new phylogenetic classification. In: Dixon KW, Kell SP, Barrett RL, Cribb PJ (eds) Orchid conservation. Sabah, Natural History Publications, Kota Kinabalu, pp 1–24
Chase MW, Cameron KM, Freudenstein JV et al (2015) An updated classification of Orchidaceae. Bot J Linn Soc 177:151–174. https://doi.org/10.1111/boj.12234
Chen J, Hu KX, Hou XQ, Guo S (2011) Endophytic fungi assemblages from 10 Dendrobium medicinal plants (Orchidaceae). World J Microbiol Biotechnol 27:1009–1016. https://doi.org/10.1007/s11274-010-0544-y
Cisternas MA, Salazar GA, Verdugo G, Novoa P, Calderón X, Negritto MA (2012) Phylogenetic analysis of Chloraeinae (Orchidaceae) based on plastid and nuclear DNA sequences. Bot J Linn Soc 168:258–277. https://doi.org/10.1111/j.1095-8339.2011.01200.x
Cribb PJ, Kell SP, Dixon KW, Barrett RL (2003) Orchid conservation: a global perspective. In: Dixon KW, Kell SP, Barrett RL, Cribb PJ (eds) Orchid conservation. Sabah, Natural History Publications, Kota Kinabalu, pp 1–24
Dahlstrom JL, Smith JE, Weber NS (2000) Mycorrhiza-like interaction by Morchellawith species of the Pinaceae in pure culture synthesis. Mycorrhiza 9:279–285. https://doi.org/10.1007/PL00009992
Davis BJ, Phillips RD, Wright M, Linde CC, Dixon K (2015) Continent-wide distribution in mycorrhizal fungi: implications for the biogeography of specialized orchids. Ann Bot 116:413–421. https://doi.org/10.1093/aob/mcv084
Dearnaley JWD, Martos F, Selosse MA (2012) Orchid mycorrhizas: molecular ecology, physiology, evolution and conservation aspects. In: Hock B (ed) The Mycota IX (fungal associations). Springer-Verlag, Berlin, pp 207–230
Devictor V, Clavel J, Julliard R, Lavergne S, Mouillot D, Thuiller W, Venail P, Villéger S, Mouquet N (2010) Defining and measuring ecological specialization. J Appl Ecol 47:15–25. https://doi.org/10.1111/j.1365-2664.2009.01744.x
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Esposito F, Jacquemyn H, Waud M, Tyteca D (2016) Mycorrhizal fungal diversity and community composition in two closely related Platanthera (Orchidaceae) species. PLoS One 11(10):e0164108. https://doi.org/10.1371/journal.pone.0164108
Fay MF, Pailler T, Dixon KW (2015) Orchid conservation: making the links. Ann Bot 116:377–379. https://doi.org/10.1093/aob/mcv142
Fracchia S, Silvani V, Flachsland E, Terada G, Sede S (2014) Symbiotic seed germination and protocorm development of Aa achalensis Schltr., a terrestrial orchid endemic from Argentina. Mycorrhiza 24:35–43. https://doi.org/10.1007/s00572-013-0510-2
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for Basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Gaston KJ (1998) Species-range size distributions: products of speciation, extinction and transformation. Phil Trans R Soc Lond B 353:219–230. https://doi.org/10.1098/rstb.1998.0204
Givinsh TJ, Spalnik D, Ames M (2016) Orchid historical biogeography, diversification, Antarctica and the paradox of orchid dispersal. J Biogeogr 43:1905–1916
Harris G, Pimm SL (2008) Range size and extinction risk in Forest birds. Conserv Biol 22:163–171. https://doi.org/10.1111/j.1523-1739.2007.00798.x
Herrera H, García-Romera I, Meneses C, Pereira G, Arriagada C (2019) Orchid Mycorrhizal interactions on the Pacific side of the Andes from Chile. A Review J Soil Sci Plant Nutr 19:187–202. https://doi.org/10.1007/s42729-019-00026-x
Jacquemyn H, Brys R, Merckx VSFT, Waud M, Lievens B, Wiegand T (2013) Coexisting orchid species have distinct mycorrhizal communities and display strong spatial segregation. New Phytol 202:616–627. https://doi.org/10.1111/nph.12640
Kartzinel TR, Trapnell DW, Shefferson RP (2013) Highly diverse and spatially heterogeneous mycorrhizal symbiosis in a rare epiphyte is unrelated to broad biogeographic or environmental features. Mol Ecol 22:5949–5961. https://doi.org/10.1111/mec.12536
Kennedy AH, Taylor DL, Watson LE (2011) Mycorrhizal specificity in the fully mycoheterotrophic Hexalectris Raf. (Orchidaceae: Epidendroideae). Mol Ecol 20:1303–1316. https://doi.org/10.1111/j.1365-294X.2011.05000.x
Lynch M, Conery J, Burger R (1995) Mutation accumulation and the extinction of small populations. Am Nat 146:489–518
Magrini S, Barreca D, Zucconi L (2019) A rapid double-staining technique to improve seed viability testing in terrestrial orchids. Plant Biosyst. https://doi.org/10.1080/11263504.2019.1587541
McCormick MK, Whigham DF, Sloan D, O’Malley K, Hodkinson B (2006) Orchid–fungus fidelity: a marriage meant to last? Ecol 87:903–911
McCormick MK, Whigham DF, Camchani-Viruet A (2018) Mycorrhizal fungi affect orchid distribution and population dynamics. New Phytol 219:1207–1215. https://doi.org/10.1111/nph.15223
McKendrick SL, Leake JR, Taylor DL, Read DJ (2002) Symbiotic germination and development of the myco-heterotrophic orchid Neottia nidus-avis in nature and its requirement for locally distributed Sebacina spp. New Phytol 154:233–247. https://doi.org/10.1046/j.1469-8137.2002.00372.x
Mitchell R (1989) Growing hardy orchids from seeds at Kew. Plantsman 11:152–169
MMA: Ministerio de Medio Ambiente, Gobierno de Chile (2011) Clasificación de especies según estado de conservación. http://www.mma.gob.cl/clasificacionespecies/
Mujica MI, Saez N, Cisternas M, Manzano M, Armesto JJ, Pérez F (2016) Relationship between soil nutrients and mycorrhizal associations of two Bipinnula species (Orchidaceae) from Central Chile. Ann Bot 118:149–158. https://doi.org/10.1093/aob/mcw082
Nguyen NH, Song Z, Bates ST, Branco S, Tedersoo L, Menke J, Schilling JS, Kennedy PG (2016) FUNGuild: an open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol 20:241–248. https://doi.org/10.1016/j.funeco.2015.06.006
Novoa P, Espejo J, Cisternas M, Rubio M, Domínguez E (2015) Guía de campo de las orquídeas chilenas. Segunda Edición, Corporación Chilena de la Madera, Concepción
Ogura-Tsujita Y, Yukawa Y (2008) High mycorrhizal specificity in a widespread mycoheterotrophic plant, Eulophia zollingeri (Orchidaceae). Am J Bot 95:93–97. https://doi.org/10.3732/ajb.95.1.93
Oliveira SF, Bocayuva MF, Gomes T, Soares DM, Canedo da Silva C, Liparini O, Kasuya MC (2014) Endophytic and mycorrhizal fungi associated with roots of endangered native orchids from the Atlantic Forest, Brazil. Mycorrhiza 24:55–64. https://doi.org/10.1007/s00572-013-0512-0
Pandey M, Sharma J, Taylor DL, Yadon VL (2013) A narrowly endemic photosynthetic orchid is non-specific in its mycorrhizal associations. Mol Ecol 22:2341–2354
Phillips RD, Barrett MD, Dixon KW, Hopper SD (2011) Do mycorrhizal symbioses cause rarity in orchids? J Ecol 99:858–869. https://doi.org/10.1111/j.1365-2745.2011.01797.x
Porras-Alfaro A, Bayman P (2007) Mycorrhizal fungi of Vanilla: diversity, specificity and effects on seed germination and plant growth. Mycol 99:510–525. https://doi.org/10.3852/mycologia.99.4.510
Rasmussen HN (1995). Terrestrial orchids. From seed to mycotrophic plant. Cambridge University Press, p. 444
Rasmussen HN, Dixon KW, Jersáková J, Tesitelová T (2015) Germination and seedling establishment in orchids: a complex of requirements. Ann bot 116:391–402
Reedeler RD, Roy R, Capell B (2002) Seed and root rots of ginseng (Panax quinquefolius L) caused by Cylindrocarpon destructans and Fusarium spp. J Ginseng Res 26:151–158
Shefferson RP, Weiß M, Kull T, Taylor DL (2005) High specificity generally characterizes mycorrhizal association in rare lady’s slipper orchids, genus Cypripedium. Mol Ecol 14:613–626. https://doi.org/10.1111/j.1365-294X.2005.02424.x
Smith SE, Read DJ (2008) Mycorrhizal symbiosis. Academic Press, Cambridge
Steinfort U, Verdugo G, Besoain X, Cisternas M (2010) Mycorrhizal association and symbiotic germination of the terrestrial orchid Bipinnula fimbriata (Poepp.) Johnst (Orchidaceae). Flora 205:811–817. https://doi.org/10.1016/j.flora.2010.01.005
Swarts ND, Dixon KW (2010) Terrestrial orchid conservation in the age of extinction. Ann Bot 104:543–556. https://doi.org/10.1093/aob/mcp025
Swarts ND, Sinclair EA, Francis A, Dixon KW (2010) Ecological specialization in mycorrhizal symbiosis leads to rarity in an endangered orchid. Mol Ecol 19:3226–3242. https://doi.org/10.1111/j.1365-294X.2010.04736.x
Swofford DL (2002) PAUP* phylogenetic analysis using parsimony (*and other methods). Version 4. Sinauer associates, Sunderland, Massachusetts
Taylor DL, McCormick MK (2008) Internal transcribed spacer primers and sequences for improved characterization of Basidiomycetous orchid mycorrhizas. New Phytol 177:1020–1033. https://doi.org/10.1111/j.1469-8137.2007.02320.x
Taylor DL, Bruns TD, Szaro TM, Hodges SA (2003) Divergence in mycorrhizal specialization within Hexalectris spicata (Orchidaceae), a nonphotosynthetic desert orchid. Am J Bot 90:1168–1179. https://doi.org/10.3732/ajb.90.8.1168
Verdugo G, Marchant J, Cisternas M, Calderón X, Peñaloza P (2007) Caracterización morfométrica de la germinación de Chloraea crispa (Orchidaceae) usando análisis de imagen. Gayan Bot 64:230–236
White TJ, Bruns TD, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, New York, pp 315–322
Willi Y, van Buskirk J, Hoffmann AA (2006) Limits to the adaptive potential of small populations. Annu Rev Ecol Evol Syst 37:433–458. https://doi.org/10.1146/annurev.ecolsys.37.091305.110145
Acknowledgements
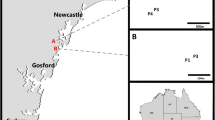
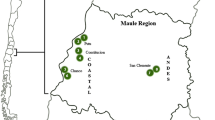
We thank all members of the Evolutionary Eco-physiology Lab of the Pontificia Universidad Católica de Chile for their help with fieldwork and data analysis. We thank Paz Montenegro for helping with the design of Fig. 1. Funding was provided by FONDECYT [1171369] and by the Institute of Ecology and Biodiversity (grants P05-002 from ICM and PFB-23 from CONICYT). M.IM. thanks CONICYT for the doctoral scholarship # 21151009. We also thank the Corporación Nacional Forestal (CONAF), Chile, for access to public protected areas.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Claro, A., Mujica, M.I., Cisternas, M. et al. Low mycorrhizal diversity in the endangered and rare orchids Bipinnula volckmannii and B. apinnula of Central Chile. Symbiosis 80, 145–154 (2020). https://doi.org/10.1007/s13199-019-00648-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-019-00648-w