Abstract
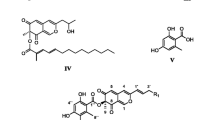
Seven new azaphilone pigments named lenormandins A–G were isolated from stromata (fruiting bodies) of the xylariaceous fungus, Hypoxylon lenormandii using preparative High Performance Liquid Chromatography (HPLC) and their structures were elucidated by NMR spectroscopy, high resolution mass spectrometry and other spectral methods. Their occurrence in herbarium specimens (including various type materials collected in the 19th and early 20th century) and several fresh collections was studied by analytical HPLC with diode array and mass spectrometric detection (HPLC-DAD/MS), revealing that this group of pigments is specific for Hypoxylon lenormandii from various geographic regions and otherwise only occurs in closely related taxa, i.e. H. sublenormandii and an undescribed fungus from Sri Lanka, for which we propose the name H. jaklitschii. The status of the new species was established by using molecular phylogenetic data based on ITS and partial beta-tubulin sequences, and by detailed morphological studies.








Similar content being viewed by others
References
Bitzer J, Köpcke B, Stadler M, Hellwig V, Ju YM, Seip S, Henkel T (2007) Accelerated dereplication of natural products, supported by reference libraries. Chimia 51:332–338
Bitzer J, Læssøe T, Fournier J, Kummer V, Decock C, Tichy HV, Piepenbring M, Peršoh D, Stadler M (2008) Affinities of Phylacia and the daldinoid Xylariaceae, inferred from chemotypes of cultures and ribosomal DNA sequences. Mycol Res 112:251–270
Clark RC, Lee SY, Boger DL (2008) Total synthesis of chlorofusin, its seven chromophore diastereomers, and key partial structures. J Am Chem Soc 130:12355–12369
Davis AL, McNeill GP, Caswell DC (1999) Analysis of conjugated linoleic acid isomers by 13C NMR spectroscopy. Chem Phys Lipids 97:155–165
Dennis RWG (1963) Hypoxyloideae of Congo. Bull Jardin Bot Brux 33:317–343
Fournier J, Köpcke B, Stadler M (2010) New species of Hypoxylon from Western Europe and Ethiopia. Mycotaxon 113:209–235
Gao JM, Yang SX, Qin JC (2013) Azaphilones: chemistry and biology. Chem Rev 113:4755–4811
Halecker S, Surup F, Kuhnert E, Mohr KI, Brock NL, Dickschat JS, Junker C, Schulz B, Stadler M (2014) Hymenosetin, a 3-decalinoyltetramic acid antibiotic from cultures of the ash dieback pathogen, Hymenoscyphus pseudoalbidus. Phytochemistry, 100:86–91
Hashimoto T, Tahara S, Takaoka S, Tori M, Asakawa Y (1994) Structures of daldinins A~C, three novel azaphilone derivatives from ascomyceteous fungus Daldinia concentrica. Chem Pharm Bull 42:2397–2399
Hellwig V, Ju YM, Rogers JD, Fournier J, Stadler M (2005) Hypomiltin, a novel azaphilone from Hypoxylon hypomiltum, and chemotypes in Hypoxylon sect. Hypoxylon as inferred from analytical HPLC profiling. Mycol Progr 4:39–54
Hladki AI, Romero IA (2009) Taxonomic and nomenclatural aspects of Hypoxylon taxa from southern South America proposed by Spegazzini. Mycologia 101:733–744
Hsieh HM, Ju YM, Rogers JD (2005) Molecular phylogeny of Hypoxylon and closely related genera. Mycologia 97:844–865
Hsieh HM, Lin CR, Fang MJ, Rogers JD, Fournier J, Lechat C, Ju YM. (2010) Phylogenetic status of Xylaria subgenus Pseudoxylaria among taxa of the subfamily Xylarioideae (Xylariaceae) and phylogeny of the taxa involved in the subfamily. Mol Phylogenet Evol 54:957–969
Ju YM, Rogers JD (1996) A revision of the genus Hypoxylon. Mycologia Memoir no. 20. APS Press, St. Paul, 365 pp
Kuhnert E, Fournier J, Peršoh D, Luangsa-ard JJ, Stadler M (2014a) New Hypoxylon species from Martinique and new evidence on the molecular phylogeny of Hypoxylon based on ITS rDNA and β-tubulin data. Fungal Divers 64:181–203
Kuhnert E, Heitkämper S, Fournier J, Surup F, Stadler M (2014b) Hypoxyvermelhotins A–C, new pigments from Hypoxylon lechatii sp. nov. Fungal Biol 118:242–252
Læssøe T, Srikitikulchai P, Luangsa-ard JJD, Stadler M (2013) Theissenia reconsidered, including molecular phylogeny of the type species T. pyrenocrata and a new genus Durotheca (Xylariaceae, Ascomycota). IMA Fungus 4:57–69
Læssøe T, Srikitikulchai P, Fournier J, Köpcke B, Stadler M (2010) Lepraric acid derivatives as chemotaxonomic markers in Hypoxylon aeruginosum, Chlorostroma subcubisporum and C. cyaninum, sp. nov. Fungal Biol 114:481–489
Martin P (1969) Studies in the Xylariaceae. Part V. Euhypoxylon. J South Afr Bot 35:149–206
Mühlbauer A, Triebel D, Peršoh D, Wollweber H, Seip S, Stadler M (2002) Macrocarpones, novel metabolites from stromata of Hypoxylon macrocarpum and new evidence on the chemotaxonomy of Hypoxylon. Mycol Prog 1:235–248
Okanya PW, Mohr KI, Gerth K, Jansen R, Müller R (2011) Marinoquinolines A - F, pyrroloquinolines from Ohtaekwangia kribbensis (Bacteroidetes). J Nat Prod 74:603–608
Pažoutová S, Follert S, Bitzer J, Keck M, Surup F, Šrůtka P, Holuša J, Stadler M (2013) A new endophytic insect-associated Daldinia species, recognised from a comparison of secondary metabolite profiles and molecular phylogeny. Fungal Divers 60:107–123
Quang DN, Hashimoto T, Tanaka M, Baumgartner M, Stadler M, Asakawa Y (2002) Chemical constituents of the ascomycete Daldinia concentrica. J Nat Prod 65:1869–1874
Quang DN, Hashimoto T, Stadler M, Asakawa Y (2004a) New azaphilones from the inedible mushroom Hypoxylon rubiginosum. J Nat Prod 67:1152–1155
Quang DN, Hashimoto T, Tanaka M, Stadler M, Asakawa Y (2004b) Cyclic azaphilones daldinins E and F from the ascomycete fungus Hypoxylon fuscum (Xylariaceae). Phytochemistry 65:469–473
Quang DN, Hashimoto T, Fournier J, Stadler M, Radulović N, Asakawa Y (2005a) Sassafrins A–D, new antimicrobial azaphilones from the fungus Creosphaeria sassafras. Tetrahedron 61:1743–1748
Quang DN, Hashimoto T, Nomura Y, Wollweber H, Hellwig V, Fournier J, Stadler M, Asakawa Y (2005b) Cohaerins A and B, azaphilones from the fungus Hypoxylon cohaerens, and comparison of HPLC-based metabolite profiles in Hypoxylon sect. Annulata. Phytochemistry 66:797–809
Quang DN, Stadler M, Fournier J, Asakawa Y (2006a) Carneic acids A and B, two chemotaxonomically significant antimicrobial agents from the xylariaceous ascomycete, Hypoxylon carneum. J Nat Prod 69:1198–1202
Quang DN, Stadler M, Fournier J, Tomita A, Hashimoto T (2006b) Cohaerins C–F, four azaphilones from the xylariaceous fungus Annulohypoxylon cohaerens. Tetrahedron 62:6349–6354
Rayner RW (1970) A mycological colour chart. Commonwealth Mycological Institute, Kew and British Mycological Society
San Martin F, Rogers JD, Lavin P, Perez-Silva E, Esqueda M (1999) Two new species of Hypoxylon from Mexico. Mycotaxon 72:159–162
Sánchez-Ballesteros J, González V, Salazar O, Acero J, Portal MA, Julián M, Rubio V (2000) Phylogenetic study of Hypoxylon and related genera based on ribosomal ITS sequences. Mycologia 92:964–977
Stadler M, Fournier J (2006) Pigment chemistry, taxonomy and phylogeny of the Hypoxyloideae (Xylariaceae). Rev Iberoam Micol 23:160–170
Stadler M, Hellwig V (2005) Chemotaxonomy of the Xylariaceae and remarkable bioactive compounds from Xylariales and their associated asexual stages. Recent Res Dev Phytochem 9:41–93
Stadler M, Fournier J, Gardt S, Peršoh D (2010) The phylogenetic position of Rhopalostroma as inferred from a polythetic approach. Persoonia 25:11–21
Stadler M, Baumgartner M, Grothe T, Mühlbauer A, Seip S, Wollweber H (2001a) Concentricol, a taxonomically significant triterpenoid from Daldinia concentrica. Phytochemistry 56:787–793
Stadler M, Wollweber H, Mühlbauer A, Henkel T, Asakawa Y, Hashimoto T, Rogers JD, Ju YM, Wetzstein HG, Tichy HV (2001b) Secondary metabolite profiles, genetic fingerprints and taxonomy of Daldinia and allies. Mycotaxon 77:379–429
Stadler M, Fournier J, Quang DN, Akulov AY (2007) Metabolomic studies on the chemical ecology of the Xylariaceae (Ascomycota). Nat Prod Commun 2:287–304
Stadler M, Kuhnert E, Peršoh D, Fournier J (2013) The Xylariaceae as model example for a unified nomenclature following the “One Fungus-One Name” (1F1N) Concept. Mycol Int J Fungal Biol 4:5–21
Stadler M, Læssøe T, Fournier J, Decock C, Schmieschek B, Tichy HV, Peršoh D (2014) A polyphasic taxonomy of Daldinia (Xylariaceae). Stud Mycol 77:1–143
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690
Steglich W, Klaar M, Furtner W (1974) Mitorubrin derivatives from Hypoxylon fragiforme. Phytochemistry 13:2874–2875
Steyn PS, Vleggaar R (1976) The structure of dihydrodeoxy-8-epi-austdiol and the absolute configuration of the azaphilones. J Chem Soc Perkin Trans, pp 204–206
Surup F, Mohr KI, Jansen R, Stadler M (2013) Cohaerins G-K, azaphilone pigments from Annulohypoxylon cohaerens and absolute stereochemistry of cohaerins C–K. Phytochemistry 95:252–258
Suwannasai N, Rodtong, S,Thienhirun S, Whalley AJS (2006) New species and phylogenetic relationships of Hypoxylon species found in Thailand inferred from the internal transcribed spacer regions of ribosomal DNA sequences. Mycotaxon 94:303–324
Triebel D, Peršoh D, Wollweber H, Stadler M (2005) Phylogenetic relationships among Daldinia, Entonaema and Hypoxylon as inferred from ITS nrDNA sequences. Nova Hedw 80:25–43
Van der Gucht K (1995) Illustrations and descriptions of xylariaceous fungi collected in Papua New Guinea. Bull Jard Bot Brux 64:219–403
Whalley WB, Ferguson G, Marsh WC, Restivo RJ (1976) The chemistry of fungi. Part LXVIII. The absolute configuration of (+)-sclerotiorin and of the azaphilones. Journal of the Chemical Society. Perkin Trans, pp 1366–1369
Yamakoshi S, Hayashi N, Suzuki T, Nakada M (2009) Asymmetric total synthesis of (+)-carneic acid A and structure revision of its natural form. Tetrahedron Lett 50:5372–5375
Acknowledgments
We gratefully acknowledge support from the curators of various international culture collections and herbaria, above all BCC, CBS, K, MFLUCC, MUCL and S, who provided important specimens for the present study. We also thank Markus Scholler (KR) for mediating international loan transactions. Prasert Srikitikulchai and J. Jennifer Luangsa-ard are thanked for their continuous work in finding important specimens during his field trips in Thailand and Jacques Fournier for his invaluable work on the taxonomy of Hypoxylon. The field sampling in Thailand benefited from a joint TRF-DAAD PPP academic exchange grant by the German Academic Exchange Service (DAAD) and the Thai Royal Golden Ph.D. Jubilee-Industry program (RGJ) to Kevin D. Hyde, Eric Kuhnert and Marc Stadler. The DAAD and the Argentina Ministerio de Ciencia, Tecnología e Innovación Productiva are also gratefully acknowledged for an academic exchange program involving Adriana Hladki, Eric Kuhnert, Esteban Benjamin Sir and Marc Stadler. Furthermore, we are grateful to various colleagues at the HZI: Kathrin I. Mohr and Wera Collisi for conducting the bioassays and Christel Kakoschke for recording NMR spectra. Manfred Rohde is thanked for SEM recordings. For measurements of the HPLC–HRMS data we are grateful to Aileen Teichmann. We also thank Philine Wotsch for technical assistance in culture maintenance and Rolf Jansen for proofreading the chemical part.
The authors would also like to express their gratitude to Administration of National Parks of Argentina, Ministry of environment of Salta province and Provincial Direction of Biodiversity of Jujuy province for authorization of collection.
Author information
Authors and Affiliations
Corresponding author
Additional information
Taxonomic Novelties
Hypoxylon jaklitschii Sir & Kuhnert, sp. nov.
Rights and permissions
About this article
Cite this article
Kuhnert, E., Surup, F., Sir, E.B. et al. Lenormandins A—G, new azaphilones from Hypoxylon lenormandii and Hypoxylon jaklitschii sp. nov., recognised by chemotaxonomic data. Fungal Diversity 71, 165–184 (2015). https://doi.org/10.1007/s13225-014-0318-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13225-014-0318-1