Abstract
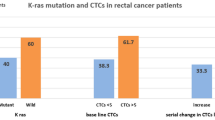
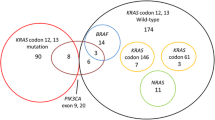
In certain cell culture studies, significant CEA expression was observed in K-ras mutant cells. However, the relationship between high CEA levels and K-ras status has not been sufficiently investigated. In the present study, we aimed to determine the prognostic role of initial CEA and CA 19–9 values in metastatic colorectal cancer patients according to the status of K-ras. Between 2000 and 2010, a total of 215 patients with metastatic colorectal cancer who were treated and followed up in our oncology center were analyzed. Smokers were excluded from the study. The clinicopathological findings and initial CEA and CA19-9 values were determined. K-ras mutation analysis was performed using quantitative PCR evaluation of the DNA from the tumor tissues. Eighty-two patients (38.1 %) were female and 133 (61.9 %) were male, with a median age of 59 years (range 27–83). Based on tumor localization, 127 patients (59 %) were classified as colon cancer patients and 88 patients (41 %) were classified as rectal cancer patients. The majority of patients (83.3 %) had pure adenocarcinoma histology, while 36 cases (16.7 %) had mucinous adenocarcinoma. The initial CEA levels were detected to be high (>5 ng/mL) in 108 of the patients (50.2 %), while high levels of initial CA 19–9 (>37 ng/mL) were found in 90 patients (41.8 %). K-ras mutations were detected in 99 of the patients (46 %). K-ras was found to be wild type in 116 patients (54 %). Significant differences were detected between the K-ras wild-type and mutant groups with respect to age and the initial serum CEA levels. Patients with K-ras mutations were younger (p = 0.04) and had higher initial CEA levels (p = 0.02) compared to patients with K-ras wild type. The median overall survival (OS) time and 3-year OS rate for patients with a high initial CEA level (>5 ng/mL) were significantly shorter than those of patients with a low initial CEA level (<5 ng/mL) (50.5 months and 61.8 % vs. 78.6 months and 79.1 %, p = 0.014). Furthermore, the patients with low initial CA 19–9 levels (<37 ng/mL) had a significant better median OS interval and 3-year OS rate (76.1 months and 80.1 %) compared to patients with high initial CA 19–9 levels (>37 ng/mL) (37.6 months and 55.7 %, p = 0.04). Multivariate analysis indicated that stage at the time of diagnosis (p < 0.001) and low initial serum CEA level (p = 0.037) were independent prognostic factors of OS. For K-ras mutant patients, the stage at diagnosis (p = 0.017), low initial serum CEA level (p = 0.001), and low initial serum CA 19–9 level were found to be independent prognostic indicators of OS. Our findings demonstrate for the first time that the presence of a K-ras mutation correlated with high initial CEA and CA 19–9 levels in patients with metastatic colorectal cancer. Patients with high initial CEA and CA 19–9 levels may potentially predict the presence of a K-ras mutation, and this prediction may guide targeted therapies in these patients.



Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013 Jan;63(1):11–30.
Benchimol S, Fuks A, Jothy S, Beauchemin N, Shirota K, Stanners CP. Carcinoembryonic antigen, a human tumor marker, functions as an intercellular adhesion molecule. Cell. 1989;57:327–34.
Berg EL, Magnani J, Warnock RA, Robinson MK, Butcher EC. Comparison of L-selectin and E-selectin ligand specificities: the L-selectin can bind the E-selectin ligands sialyl Lex and sialyl Lea. Biochem Biophys Res Commun. 1992;184:1048–55.
Hamazoe R, Maeta M, Matsui T, Shibata S, Shiota S, Kaibara N. CA72-4 compared with carcinoembryonic antigen as a tumor marker for gastric cancer. Eur J Cancer. 1992;28A:1351–4.
Hamazoe R, Maeta M, Matsui T, Shibata S, Shiota S, Kaibara N. CA72-4 compared with carcinoembryonic antigen as a tumor marker for gastric cancer. Eur J Cancer. 1992;28A:1351–4.
Formica V, Massara MC, Portarena I, Fiaschetti V, Grenga I, Del Vecchio BG, et al. Role of CA19.9 in predicting bevacizumab efficacy for metastatic colorectal cancer patients. Cancer Biomark. 2009;5(4):167–75.
Van Cutsem E, Rivera F, Berry S, et al. First BEAT investigators: safety and efficacy of first-line bevacizumab with FOLFOX, XELOX, FOLFIRI and fluoropyrimidines in metastatic colorectal cancer: the BEAT study. Ann Oncol. 2009;20:1842–7.
Vaughn CP, Zobell SD, Furtado LV, Baker CL, Samowitz WS. Frequency of K-RAS, B-RAF, and NRAS mutations in colorectal cancer. Genes Chromosomes Cancer. 2011;50:307–12.
Chao X. Zheng, Wen Hua Zhan, Ji Zong Zhao, Dong Zheng, Dong Ping Wang, et al. The prognostic value of preoperative serum levels of CEA, CA19-9 and CA72-4 in patients with colorectal cancer. World J Gastroenterol. 2001;7(3):431–4.
Paxton RJ, Mooser G, Pande H, Lee TD, Shively JE. Sequence analysis of carcinoembryonic antigen: identification of glycosylation sites and homology with the immunoglobulin supergene family. Proc Natl Acad Sci USA. 1987;84:920–4.
Lowe JB, Stoolman LM, Nair RP, Larsen RD, Berhend TL, Marks RM. ELAM-1 dependent cell adhesion to vascular endothelium determined by a transfected human fucosyltransferase cDNA. Cell. 1990;63:475–84.
Kornek GV, Depisch D, Rosen HR, Temsch EM, Scheithauer W. Comparative analysis of CA72-4, CA195 and carcinoembryonic antigen in patients with gastrointestinal malignancies. J Cancer Res Clin Oncol. 1992;118:318–20.
Ohuchi N, Takahashi K, Matoba N, Sato T, Taira Y, Sakai N, et al. Comparison of serum assays for TAG-72, CA19-9, and CEA in gastrointestinal carcinoma patients. Jpn J Clin Oncol. 1989;19:242–8.
Fucini C, Tommasi SM, Rosi S, Malatantis G, Cardona G, Panichi S, et al. Follow-up of colorectal cancer resected for cure. An experience with CEA, TPA, Ca19-9 analysis and second look surgery. Dis Colon Rectum. 1987;30:273–7.
Sato T, Nishimura G, Nonomura A, Miwa K, Miyazaki I. Serological studies on CEA, CA19-9, STn and SLX in colorectal cancer. Hepatogastroenterology. 1999;46:914–9.
Forones NM, Tanaka M. CEA and CA19 9 as prognostic indexes in colorectal cancer. Hepatogastroenterology. 1999;46:905–8.
Takada A, Ohmori K, Takahashi N, Tsuyuoka K, Yago A, Zenita K, et al. Adhesion of human cancer cells to vascular endothelium mediated by a carbohydrate antigen, sialyl Lewis A. Biochem Biophys Res Commun. 1991;179:713–9.
Wojciechowicz DC, Park PY, Datta RV, Paty PB. CEA is the major PHA-L-reactive glycoprotein in colon carcinoma cell lines and tumors: relationship between K-ras activation and beta1-6 branching of N-linked carbohydrate on CEA. Biochem Biophys Res Commun. 2000 24;273(1):147–53.
Wojciechowicz DC, Mallon RG, Picon A, Paty PB. Characterization of lectin resistant cell populations derived from human colon carcinoma: correlation of K-ras with beta1-6 branching of N-linked carbohydrate and CEA production. Biochem Biophys Res Commun. 1999 16;259(3):588–93
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Selcukbiricik, F., Bilici, A., Tural, D. et al. Are high initial CEA and CA 19–9 levels associated with the presence of K-ras mutation in patients with metastatic colorectal cancer?. Tumor Biol. 34, 2233–2239 (2013). https://doi.org/10.1007/s13277-013-0763-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-0763-6