Abstract
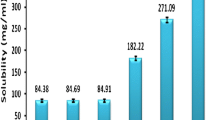
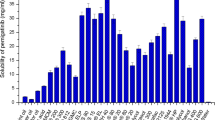
The present research was aimed at development and evaluation of self-nanoemulsifying drug delivery system (SNEDDS) for improving bioavailability of nelfinavir mesylate (NFV), a protease inhibitor exhibiting pH dependent solubility and variable oral bioavailability. Maisine 35-1, Cremophor RH-40, and Labrasol were identified as oil, surfactant, and co-surfactant that had best solubility for NFV. Scheffe’s mixture design was used to optimize the amount of components in liquid self-nanoemulsifying drug delivery system (L-SNEDDS) by taking their amounts as independent variable, whereas globule size, drug loading, and percent transmittance were taken as dependent variable. Optimized NFV-L-SNEDDS was then adsorbed on Neusilin US2 to form solid self-nanoemulsifying drug delivery system (S-SNEDDS). NFV loaded L-SNEDDS and S-SNEDDS were characterized for various physicochemical properties, and solid-state properties were determined through differential scanning calorimetry, X-ray diffraction, and scanning electron microscopy studies. In vitro dissolution using simulated gastric fluid and simulated intestinal fluid, ex vivo drug release study, and in vivo study were performed for pure NFV and NFV-S-SNEDDS. NFV-S-SNEDDS showed more than 90 % drug release in 20 min during drug release studies irrespective of pH of the dissolution medium. In vivo study revealed significant difference between release of NFV from suspension and NFV-L-SNEDDS and NFV-S-SNEDDS when given to rabbits (p < 0.001). NFV-L-SNEDDS and NFV-S-SNEDDS were subjected to stability study as per ICH guidelines, and NFV-S-SNEDDS was found to be stable during the period of study. S-SNEDDS could serve as a potential drug delivery system for NFV.












Similar content being viewed by others
References
Zhang KE, Wu E, Patick AK, Kerr B, Zorbas M, Lankford A, et al. Circulating metabolites of the human immunodeficiency virus protease inhibitor nelfinavir in humans: structural identification, levels in plasma, and antiviral activities. Antimicrob Agents Chemother. 2001;45:1086–93. doi:10.1128/AAC.45.4.1086-1093.
Yihong Q. Developing solid oral dosage forms: pharmaceutical theory and practice. New York: Elsevier; 2009.
PRVIRACEPT (2011) http://www.pfizer.ca/en/our_products/products/monograph/133. Accessed 2 July 2013
Patick AK, Mo H, Markowitz M, Appelt K, Wu B, Kalish V, et al. Antiviral and resistance studies of ag1343, an orally bioavailable inhibitor of human immunodeficiency virus protease. Antimicrob Agents Chemother. 1996;40:292–7.
Kasim NA, Whitehouse M, Ramachandran C, Bermejo M, Lennernäs H, Hussain AS, et al. Molecular properties of WHO essential drugs and provisional biopharmaceutical classification. Mol Pharmacol. 2004;1:85–96. doi:10.1021/mp034006h.
Williams GC, Sinko PJ. Oral absorption of the HIV protease inhibitors: a current update. Adv Drug Del Rev. 1999;39:211–38. doi:10.1016/S0169-409X(99)00027-7.
Sarapa N, Hsyu PH, Lappin G, Garner RC. The application of accelerator mass spectrometry to absolute bioavailability studies in humans: simultaneous administration of an intravenous microdose of 14C-nelfinavir mesylate solution and oral nelfinavir to healthy volunteers. J Clin Pharmacol. 2005;45:1198–205. doi:10.1177/0091270005280051.
Torne SJ, Torne JS, Vavia PR, Singh SK. Inclusion complexes of anti-hiv drug with β-Cyclodextrin. Chemistry and materials science. J Inc Pheno Macro Chem. 2006;56:253–59. doi:10.1007/S10847-006-9092-3.
Pouton CW. Formulation of self-emulsifying drug delivery systems. Adv Drug Del Rev. 1997;25:47–58. doi:10.1016/S0169-409X(96)00490-5.
Kale A, Patravale VB. Design and evaluation of self-emulsifying drug delivery system (SEDDS) of nimodipine. AAPS PharmSciTech. 2008;9(1):191–6. doi:10.1208/s12249-008-9037-9.
Shaji J, Jadhav D. Newer approaches to self emulsifying drug delivery system. Int J Pharm Pharm Sci. 2010;2:37–42.
Hauss D. Oral lipid-based formulations. Adv Drug Del Rev. 2007;59:667–7. doi:10.1016/j.addr.2007.05.006.
Cornaire G, Woodley J, Hermann P, Cloarec A, Arellano C, Houin G. Impact of excipients on the absorption of P-glycoprotein substrates in vitro and in vivo. Int J Pharm. 2004;278:119–31. doi:10.1016/j.ijphrm.2004.03.001.
Wandel C, Kim RB, Stein CM. “Inactive” excipients such as cremophor can affect in vivo drug disposition. Clin Pharmacol Ther. 2003;73:394–96. doi:10.1016/S0009-9236(03)00010-9.
Hauss DJ, Fogal SE, Ficorilli JV, Price CA, Roy T, Jayaraj AA, et al. Lipid-based delivery systems for improving the bioavailability and lymphatic transport of a poorly water-soluble LTB4 inhibitor. J Pharm Sci. 1998;87:164–69. doi:10.1021/js970300n.
Rege B, Kao J, Polli J. Effect of nonionic surfactants on membrane transporters in Caco-2 cell monolayers. Eur J Pharm Sci. 2002;16:237–46. doi:10.1016/S0928-0987(02)00055-6.
Elnaggar YSR, El-Massik MA, Abdallah OY. Self-nanoemulsifying drug delivery systems of tamoxifen citrate: design and optimization. Int J Pharm. 2009;380:133–41. doi:10.1016/j.ijpharm.2009.07.015.
Gupta S, Chavhan S, Sawant K. Self-nanoemulsifying drug delivery system for adefovir dipivoxil: design, characterization, in vitro and ex vivo evaluation. Colloids and Surfaces A: Physicochem Eng Aspects. 2011;392:145–55. doi:10.1016/j.colsurfa.2011.09.048.
Oh DH, Kang JH, Kim DW, Lee BJ, Kim JO, Yong CS, et al. Comparison of solid selfmicroemulsifying drug delivery system (solid SMEDDS) prepared with hydrophilic and hydrophobic solid carrier. Int J Pharm. 2011;28:412–18. doi:10.1016/j.ijpharm.2011.09.007.
Paola M, Maurizio V, Marzia C, Natascia M. New solid self microemulsifying systems to enhance dissolution rate of poorly water soluble drugs. Pharm Dev Technol. 2012;17:277–84. doi:10.3109/10837450.2010.535825.
Dong WK, Jun HK, Dong HO, Han-Gon C. Development of novel flurbiprofen-loaded solid self-microemulsifying drug delivery system using gelatin as solid carrier. J Microencapsul. 2012;29:323–30. doi:10.3109/02652048.2011.651497.
Kang JH, Oh DH, Oh YK, Yong CS, Choi HG. Effects of solid carriers on the crystalline properties, dissolution and bioavailability of flurbiprofen in solid self-nanoemulsifying drug delivery system (solid SNEDDS). Eur J Pharm Biopharm. 2012;80:289–97. doi:10.1016/j.ejpb.2011.11.005.
Venkata RK, Suresh B, Raju J, Prabhakar RV. Oral self emulsifying powder of lercanidipine hydrochloride: formulation and evaluation. Powder Tech. 2012;221:375–82. doi:10.1016/j.powtec.2012.01.032.
Shanmugam S, Baskaran R, Balakrishnan P, Thapa P, Yong CS, Yoo BK. Solid self nanoemulsifying drug delivery system (S-SNEDDS) containing phosphatidylcholine for enhanced bioavailability of highly lipophilic bioactive carotenoid lutein. Eur J Pharm Biopharm. 2011;79:250–7. doi:10.1016/j.ejpb.2011.04.012.
Saipin S, Sirima M, Narubodee P, et al. Development and evaluation of self-microemulsifying liquid and pellet formulations of curcumin, and absorption studies in rats. Eur J Pharm Biopharm. 2010;76:475–85. doi:10.1016/j.ejpb.2010.07.011.
Janolya A, Bleyzaca N, Favettab P, Gagneuc M, Aulagner G. Simple and rapid high-performance liquid chromatographic method for nelfinavir, M8 nelfinavir metabolite, ritonavir and saquinavir assay in plasma. J Chromatogr B. 2002;780:155–60. doi:10.1016/S1570-0232(02)00465-8.
Scheffe H. Experiments with mixtures. J Royal Stat Soc Ser B. 1958;20:344–60.
Colas JC, Shi W, Rao VSNM, Omri A, Mozafari MR, Singh H, et al. Microscopical investigations of nisin-loaded nanoliposomes prepared by Mozafari method and their bacterial targeting. Micron. 2007;38:841–47. doi:10.1016/j.micron.2007.06.013.
Bandivadekar M, Pancholi S, Kaul-Ghanekar R, Choudhari A, Koppikar S. Single non-ionic surfactant based self-nanoemulsifying drug delivery systems: formulation, characterization, cytotoxicity and permeability enhancement study. Drug Dev Ind Pharm. 2013;39:696–703. doi:10.3109/03639045.2012.687745.
Bhagwat DA, D’Souza JI, Intru J. Bhagwat J and D’souza Formulation and evaluation of solid self micro emulsifying drug delivery system using aerosil 200 as solid carrier. International Current Pharmaceutical Journal. 2012;1:414–19.
Chen Y, Chen C, Zheng J, Chen Z, Shi Q, Liu H. Development of a solid supersaturatable self-emulsifying drug delivery system of docetaxel with improved dissolution and bioavailability. Biol Pharm Bull. 2011;34:278–86.
Sarwar B, Jeena SS, Patra NC, Rizwan M, Swaina S, Sruti J, et al. Development of solid self-nanoemulsifying granules (SSNEGs) of ondansetron hydrochloride with enhanced bioavailability potential. Colloids Surf B: Biointerfaces. 2013;101:414–23. doi:10.1016/j.colsurfb.2012.06.031.
Smith P. Methods for evaluating intestinal permeability and metabolism in vitro. Pharm Biotechnol. 1996;8:13–34. doi:10.1007/978-1-4899-1863-5-2.
BorhadeV SS, Patravale V. Clotrimazole nanoemulsion for malaria chemotherapy. Part I: Preformulation studies, formulation design and physicochemical evaluation. Int J Pharm. 2012;431:138–48. doi:10.1016/j.ijpharm.2011.12.040.
Rangel-Yagui CO, Junior AP, Tavares LC. Micellar solubilization of drugs. J Pharm Pharm Sci. 2005;8:147–63.
Fuzheng R, Yanhui T, Yongjia S, Xinhong Y, Zhian H. Determination of nelfinavir mesylate as bulk drug and in pharmaceutical dosage form by stability indicating HPLC. J Pharm Biomed Anal. 2006;41:1065–69. doi:10.1016/j.jpba.2006.02.003.
Barot BS, Parejiya PB, Patel HK, Gohel MC, Shelat PK. Microemulsion-based gel of terbinafine for the treatment of onychomycosis: optimization of formulation using D-Optimal design. AAPS PharmSciTech. 2012;13:184–92. doi:10.1208/s12249-011-9742-7.
Ren S, Mu H, Alcher F, Chtatou A, Mullrtyz A. Optimization of self nanoemulsifying drug delivery system for poorly water-soluble drug using response surface methodology. Drug Dev Ind Pharm. 2013;39(5):799–806. doi:10.3109/03639045.2012.710634.
Dixit AR, Rajput SJ, Patel SG. Preparation and bioavailability assessment of SMEDDS containing valsartan. AAPS PharmSciTech. 2010;11:314–21. doi:10.1208/s12249-010-9385-0.
Tharwat F. Applied surfactants: principles and applications. Weinheim: Wiley-VCH; 2005
Gershanik T, Benita S. Self-dispersing lipid formulations for improving oral absorption of lipophilic drugs. Eur J Pharm Biopharm. 2000;50:179–88. doi:10.1016/S0939-6411(00)00089-8.
Parul S, Ranjana M, Bakhshib AK. Stress degradation studies of nelfinavir mesylate by Fourier transform infrared spectroscopy. J Pharm Biomed Anal. 2010;53:287–94. doi:10.1016/j.jpba.2010.03.030.
Aulton ME. Aulton’s pharmaceutics: the design and manufacture of medicines. 3rd ed. Hungary: Harcourt; 2007.
Martin HA, Wantanee P, Navnit HS, Zhang Lin; Hoffmann-La Roche Ag Pharmaceutical dosage form of amorphous nelfinavir mesylate. United States patent US7014866. 14 Nov 2002.
Sha X, Yan G, Yunjuan W. Junchan Li and Fang X. Effect of self-microemulsifying drug delivery systems containing Labrasol on tight junctions in Caco-2 cells. Eur J Pharm Sci. 2005;24:477–86.
Nerukar MM, Burton PS, Bourchart RT. The use of surfactants to enhance permeability of peptides through Caco-2 cells by inhibition of an apically polarized system. Pharm Res. 1996;13:528–34. 10.1023/A: 1016033702220.
Beg S, Swain S, Singh HP, Patra CN, Rao MB. Development, optimization, and characterization of solid self-nanoemulsifying drug delivery systems of valsartan using porous carriers. AAPS PharmSciTech 2012;13:1416–27.
Acknowledgments
The authors are thankful to Cipla Ltd. (Goa, India) for providing the gift sample of standard of Nelfinavir mesylate. The authors are also thankful to Abitec Corporation (OH, USA) Gattefosse (Lyon, France), and BASF (Ludwigshafen, Germany) for providing necessary excipients. The authors also express gratitude towards Mr. Punit Parejiya and Mr. Vinit Movaliya for helping in carrying out in vivo studies. It is a part of Ph.D. work carried out at Kadi Sarva Vishwavidyalaya (Gandhinagar, India). The authors express gratitude towards Gujarat Council on Science and Technology (GUJCOST) for funding the project under the scheme of Minor Research Project (MRP) having sanction order no. GUJCOST/MRP/201441/2009-10/3054.
Conflict of interest
The authors have no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patel, A., Shelat, P. & Lalwani, A. Development and optimization of solid self-nanoemulsifying drug delivery system (S-SNEDDS) using Scheffe’s design for improvement of oral bioavailability of nelfinavir mesylate. Drug Deliv. and Transl. Res. 4, 171–186 (2014). https://doi.org/10.1007/s13346-014-0191-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-014-0191-1