Abstract
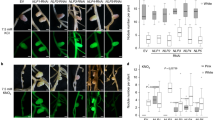
Nodulin proteins are expressed in legume root cells after infection with rhizobia. Nodulins have been classified as early and late, reflecting the developmental time points of their expression. Leghemoglobin (LegH), which is a classical example of a late nodulin, sequesters oxygen inside the nodule to protect the nitrogenase from oxygen toxicity to sustain symbiotic nitrogen fixation (SNF). Post-translational modification and/or protein–protein interaction are known to regulate activity of proteins. To elucidate the role of post-translational modification of LegH on its oxygen sequestration activity, earlier we have shown that phosphorylation at its S45 imparts most structural disruption of the porphyrin binding pocket responsible for its oxygen binding. In the present report, in an attempt to characterize the protein(s) that may interact with LegH to regulate its activity, it is demonstrated that LegH interact in vitro with Nodulin 16 of Lotus japonicus (Nlj16), another late nodulin. These two interacting proteins resulted in a bigger sized particle which shows higher diffusion coefficient as measured by dynamic light scattering. Interestingly, it was also shown that in vitro oxygen sequestration by LegH is stimulated by this interaction. Furthermore, this interaction is validated by the fact that LegH and Nlj16 could be co-immunoprecipitated from the nodule lysate. Most importantly, fluorescent immunohistochemistry of post-infected nodule sections show perceivable co-localization of these two proteins in the nodule symbiosomes. Thus, this work is a foundation for further investigation on these two interacting late nodulins as one of the plausible regulations for the oxygen sequestration by LegH during SNF.






Similar content being viewed by others
Abbreviations
- LegH:
-
Leghemoglobin
- Nlj16:
-
Nodulin Lotus Japonicus 16 kDa
- IHC:
-
Immunohistochemistry
- SNF:
-
Symbiotic Nitrogen Fixation
- LjNPP2C1:
-
Lotus Japonicus nodulin protein Phosphatase
- DLS:
-
Dynamic light scattering
- pO2 :
-
Partial pressure of O2
References
Berne BJ, Pecora R (2000) Dynamic light scattering: with applications to chemistry, biology, and physics. Dover Publications Inc, Mineola
Bhar K, Maity A, Ghosh A, Das T, Dastidar SG, Siddhanta A (2015) Phosphorylation of leghemoglobin at S45 is most effective to disrupt the molecular environment of its oxygen binding pocket. Protein J 34(2):158–167. https://doi.org/10.1007/s10930-015-9608-z
Broughton WJ, Dilworth MJ (1971) Control of leghaemoglobin synthesis in snake beans. Biochem J 125(4):1075–1080. https://doi.org/10.1042/bj1251075
Chakrabarti R, Sanyal S, Ghosh A, Bhar K, Das C, Siddhanta A (2015) Phosphatidylinositol-4-phosphate 5-Kinase 1 α modulates ribosomal RNA gene silencing through its interaction with histone H3 lysine 9 trimethylation and heterochromatin protein HP1 α. J Biol Chem 290(34):20893–20903. https://doi.org/10.1074/jbc.M114.633727
Chatterjee M, Gupta S, Bhar A, Das S (2012) Optimization of an efficient protein extraction protocol compatible with two-dimensional electrophoresis and mass sepectrometry from recalcitrant phenolic rich roots of chickpea (Cicer arietinum L.). Int J Proteomics 2012:536963. https://doi.org/10.1155/2012/536963
Cordoba E, Shishkova S, Vance CP, Hemandez G (2003) Antisense inhibition of NADH glutamate synthase impairs carbon/nitrogen assimilation in nodules of alfalfa (Medicago sativa L.). Plant J. 33(6):1037–1049. https://doi.org/10.1046/j.1365-313X.2003.01686.x
Desbrosses GJ, Stougaard J (2011) Root nodulation: a paradigm for how plant-microbe symbiosis influences host developmental pathways. Cell Host Microbe 10(4):348–358. https://doi.org/10.1016/j.chom.2011.09.005
Downie JA (2005) Legume hemoglobins: symbiotic nitrogen fixation needs bloody nodules. Curr Biol 15(6):R196–R198. https://doi.org/10.1016/j.cub.2005.03.007
Fuchsman WH, Appleby CA (1979) Separation and determination of the relative concentrations of the homogeneous components of soybean leghemoglobin by isoelectric focusing. Biochim Biophys Acta 579(2):314–324. https://doi.org/10.1016/0005-2795(79)90059-X
Grønlund M, Agalou A, Rubio MC, Lamers GEM, Roussis A, Spaink HP (2005) Embedding root and nodule tissue in plastic (BMM). In: Márquez AJ, Stougaard J (eds) Cell and molecular biology protocols, Lotus japonicus handbook. Springer, Berlin, pp 111–122
Handberg K, Stougaard J (1992) Lotus japonicus, an autogamous, diploid legume species for classical and molecular genetics. Plant J 2(4):487–496. https://doi.org/10.1111/j.1365-313X.1992.00487.x
Kapranov P, De Bruijn FJ, Szczyglowski K (1997) Novel, highly expressed late nodulin gene (LjNOD16) from Lotus japonicus. Plant Physiol 113(4):1081–1190. https://doi.org/10.1104/pp.113.4.1081
Kapranov P, Jensen TJ, Poulsen C, De Bruijn FJ, Szczyglowski K (1999) A protein phosphatase 2C gene, LjNPP2C1, from Lotus japonicus induced during root nodule development. Proc Natl Acad Sci USA 96(4):1738–1743
Kawashima K, Suganuma N, Tamaoki M, Kouchi H (2001) Two types of pea leghemoglobin genes showing different O2-binding affinities and distinct patterns of spatial expression in nodules. Plant Physiol 125(2):641–651. https://doi.org/10.1104/pp.125.2.641
Lin YW (2018) Structure and function of heme proteins regulated by diverse posttranslational modifications. Arch Biochem Biophys 641:1–30. https://doi.org/10.1016/j.abb.2018.01.009
Marx H, Minogue CE et al (2016) A proteomic atlas of the legume Medicago truncatula and its nitrogen-fixing endosymbiont Sinorhizobium meliloti. Nat Biotechnol 34(11):1198–1205. https://doi.org/10.1038/nbt.3681
Mus F, Crook MB, Garcia K et al (2016) Symbiotic nitrogen fixation and the challenges to its extension to nonlegumes. Appl Environ Microbiol 82(13):3698–3710. https://doi.org/10.1128/AEM.01055-16
Mylona P, Pawlowski K, Bisseling T (1995) Symbiotic nitrogen fixation. Plant Cell 7(7):869–885. https://doi.org/10.1105/tpc.7.7.869
Navascuésa J, Rontoméa CP, Gay M et al (2012) Leghemoglobin green derivatives with nitrated hemes evidence production of highly reactive nitrogen species during aging of legume nodules. Proc Natl Acad Sci USA 109(7):2660–2665. https://doi.org/10.1073/pnas.1116559109
Saito A, Tanabata S, Tanabata T et al (2014) Effect of nitrate on nodule and root growth of soybean (Glycine max (L.) Merr.). Int J Mol Sci 15(3):4464–4480. https://doi.org/10.3390/ijms15034464
Schultze M, Kondorosi A (1998) Regulation of symbiotic root nodule development. Annu Rev Genet 32:33–57. https://doi.org/10.1146/annurev.genet.32.1.33
Siddhanta A, Radulescu A, Stankewich MC, Shields D (2003) Fragmentation of golgi apparatus. A role for beta III spectrin and synthesis of phosphatidylinositol 4,5-bisphosphate. J Biol Chem 278:1957–1965
Stougaard J (2000) Regulators and regulation of legume root nodule development. Plant Physiol 124(2):531–540. https://doi.org/10.1104/pp.124.2.531
Van De Sande K, Bisseling T (1997) Signalling in symbiotic root nodule formation. Essays Biochem 32:127–142
Verdier J, Orres-Jerez IT, Wang M et al (2013) Establishment of the Lotus japonicus gene expression atlas (LjGEA) and its use to explore legume seed maturation. Plant J 74(2):351–362. https://doi.org/10.1111/tpj.12119
Weighardt F, Biamonti G, Riva S (1993) A simple procedure for enhancing PCR specificity. Genome Res 3:77–80
Zahran HH (1999) Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol Mol Biol Rev 63(4):968–989
Acknowledgements
We greatly appreciate the gift of Lotus japonicus late nodule (21 day old) cDNA library, L.japonicus seeds and Mesorhizobium loti (strain NZP2235) from Dr. J. Stougaard, Dept. of Molecular Biology, Aarhus University, Denmark. We purchased L. japonicus seeds from National Bioresource Project (Lotus japonicus, Glycine max) University of Miyazaki, Japan. We thank Dr. S.O Raja and Dr. Mohammad Azharuddin for technical support. This research was supported by the Centre for Modern Biology under University Potential for Excellence (UPE) and Center for Advanced Studies in Biochemistry awarded by the University Grants Commission (UGC) India. The financial assistance from Departments of Science and Technology and Biotechnology, Government of India (SR/SO/PS/82/2010; SR/SO/HS-51/2008; BT/PR 11415/BRB/10/656/2008); and Interdisciplinary Program in Life science (IPLS), Department of Biotechnology (DBT), Govt. of India also have supported this work. Amit Ghosh received his fellowship from IPLS program awarded by DBT and CSIR (SRF), Govt. of India. Kaushik Bhar was supported by research fellowship under the scheme for University Potential for Excellence (UPE).
Author information
Authors and Affiliations
Contributions
AS conceived the study and designed the experiments. AG performed the immunohistochemistry in nodule section, co-immunoprecipitation and DLS experiments. KB performed the in vitro pull down assays. AG and KB both involved in protein purification and measurements of oxygen consumption. AS and AG analyzed the data and wrote the paper. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghosh, A., Bhar, K. & Siddhanta, A. Oxygen sequestration by Leghemoglobin is positively regulated via its interaction with another late nodulin, Nlj16 of Lotus japonicus. J. Plant Biochem. Biotechnol. 28, 414–423 (2019). https://doi.org/10.1007/s13562-019-00494-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13562-019-00494-3