Abstract
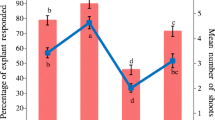
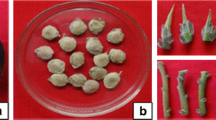
The present study reports optimization of in vitro rooting of Cattleya shoots with the aim to increase survival rate during acclimatization. Shoots (2.0–2.5 cm) which were regenerated in vitro were cultured on Murashige and Skoog (MS) medium and the effect of various parameters such as type and concentration of auxin [0.3, 0.6, 1.2 and 2.4 mg·L−1 indole butyric acid (IBA) and naphthaleneacetic acid (NAA)], medium strength (full and half strength), sucrose concentration (0, 15 and 30 g·L−1) and light intensity [photosynthetic photon flux density (PPFD) of 30, 60, 90 μmol·m−2·s−1] were tested. Half strength MS medium supplemented with 2.4 mg·L−1 IBA, 3% sucrose and 60 μmol·m−2·s−1 PPFD were found as suitable conditions for optimal induction of roots. In vitro regenerated Cattleya plantlets were successfully transferred to potting medium containing bark:perlite:peatmoss (1:1:1) and perlite:peatmoss (1:1), and 98.3 and 80.7% were survived respectively.
Similar content being viewed by others
Literature Cited
Arditti, J. 2009. Micropropagation of orchids. 2nd ed. Wiley-Blacwell, Cambridge, UK.
Chugh, S., S. Guha, and I.U. Rao. 2009. Micropropagation of orchids: A review on the potential different explants. Sci. Hort. 122:507–520.
Diaz, L.P., J.J. Namur, S.A. Bollati, and O.E.A. Arce. 2010. Acclimatization of Phalaenopsis and Cattleya obtained by micropropagation. Rev. Colomb. Biotecnol. 12:27–40.
George, E.F. and P.C. Debergh. 2008. Micropropagation: Uses and methods, p. 29–64. In: E.F. George, M.A. Hall, and G.J. De Klerk (eds.). Plant propagation by tissue culture. 3rd ed. Spinger, Dordrecht, Netherlands.
Hazarika, B.N. 2003. Acclimatization of tissue cultured plants. Curr. Sci. 85:1704–1712.
Kubota, S. and K. Yoneda. 1993. Effect of light intensity preceding day-night temperatures on the sensitivity of Phalaenopsis to flower. J. Japan. Soc. Hort. Sci. 62:595–560.
Murashige, T. and F. Skoog. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497.
Nayak, N.R., S.P. Rath, and S. Patnaik. 1997. In vitro propagation of three epiphytic orchids, Cymbidium aloifolium (L.) Sw., Dendrobium aphyllum (Roxb.) Fisch and Dendrobium moschatum (Buch.-Ham.) Sw. through thidiazuron-induced high frequency shoot proliferation. Sci. Hort. 71:243–250.
Pospisilova, J., I. Ticha, P. Kadleck, D. Haisel, and S. Plazakova. 1999. Acclimatization of micropropagated plants to ex vitro conditions. Biol. Plant. 42:481–497.
Reinert, R.A. and H.C. Mohr. 1967. Propagation of Cattleya by tissue culture of later bud meristems. Proc. Amer. Soc. Hort. Sci. 7:18.
Sheelavanthmath, S.S., H.N. Murthy, A.N. Pyati, H.G. Ashok Kumar, and B.V. Ravishankar. 2000. In vitro propagation of the endangered orchid, Geodurumdensiflorum (Lam.) Schltr. through rhizome section culture. Plant Cell Tiss. Org. Cult. 60:151–154.
Vahdati, K., C. Leslie, Z. Zamani, and G. McGranahan. 2004. Rooting and acclimatization of in vitro-grown shoot from mature trees of three Persian walnut cultivars. HortScience 39:324–327.
Van Huylenbroeck, J., A. Piqueras, and P. Debergh. 1998. Photosynthesis and carbon metabolism in leaves formed prior and during ex vitro acclimatization of micropropagated plants. Plant Sci. 134:21–30.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dewir, Y.H., El-Mahrouk, M.E., Murthy, H.N. et al. Micropropagation of Cattleya: Improved in vitro rooting and acclimatization. Hortic. Environ. Biotechnol. 56, 89–93 (2015). https://doi.org/10.1007/s13580-015-0108-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-015-0108-z