Abstract
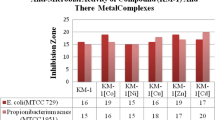
Metal complexes of Schiff base 4-[(2, 4-dihydroxy-benzylidene)-amino]-1, 5-dimethyl-2-phenyl-1, 2-dihydroxo-pyrazol-3-one (H2L) derived from condensation of 2,4-dihydroxybenzaldehyde and 4-aminoantipyrine were synthesized and characterized by FT-IR spectra, elemental analysis, 1H &13C NMR, mass spectra, UV–Visible, X-ray diffraction, molar conductance, magnetic moment and thermal analysis (TGA). The elemental analysis proved that the complexes have the general formulae of [M(HL)(X)(OH2)]2, where M = Cu(II); X = OAc− (1); NO3− (2); Cl− (3), M = Co(II) (4), Mn(II) (5); X = OAc and {[UO2(HL)]2.2(OAc)}(6). According to the FT-IR spectra data, it was found that the H2L ligand is coordinated to the metal ions in a tridentate donor sites of the nitrogen atom of azomethine group, the oxygen atom of phenolic group and the oxygen atom of carbonyl group. From the measurements of magnetic susceptibility and electronic spectral data, it is found that these complexes are octahedral geometries. The trend in g value (g║ ˃ g⊥ ˃ 2.00) suggests that the unpaired electron on copper has dx2-y2 character and the complexes have an octahedral structure. By comparing the synthesized ligand (H2L) to its metal complexes was screened for their antibacterial activity against Klebsiella pneumoniae, Escherichia coli, Pseudomonas sp., Bacillus cereus and Staphylococcus aureus. It was found that the metal complexes had more potent antimicrobial than the parent Schiff base ligand against one or more microbial species. Some complexes showed higher activity than ligand. The complex (3) has antibacterial activities against Bacillus cereus, Klebsiella pneumoniae and Staphylococcus aureus. The antibacterial activity of complex (3) was the highest ones than the ligand and other complexes as well as penicillin G against Klebsiella pneumoniae. Antifungal activities were determined against Fusarium oxysporum, Aspergillus niger and Candida albicans. The inhibition efficiency of the prepared ligand (H2L) against the corrosion of carbon steel in HCl (2 M) solution was determined by various electrochemical techniques.


















Similar content being viewed by others
References
A.Z. El-Sonbati, M.A. Diab, A.A. El-Bindary, G.G. Mohamed, Sh.M. Morgan M.I. Abou-Dobara, S.G. Nozha, J. Mol. Liq. 215, 423 (2016)
A.Z. El-Sonbati, M.A. Diab, Sh.M. Morgan, M.I. Abou-Dobara, A.A. El-Ghettany, J. Mol. Str. 1200, 127065 (2020)
A.Z. El-Sonbati, M.A. Diab, Sh.M. Morgan, H.A. Seyam, J. Mol. Str. 1154, 354 (2018)
M.A. Diab, S.G. Nozha, A.Z. El-Sonbati, M.A. El-Mogazy, Sh.M. Morgan, Appl. Organomet. Chem. 33, e5153 (2019)
M.I. Abou-Dobara, N.F. Omar, M.A. Diab, A.Z. El-Sonbati, Sh.M. Morgan, O.L. Salem, A.M. Eldesoky, Mat. Sc. Eng. C 103, 109727 (2019)
T. Rosu, E. Pahontu, C. Maxim, A.P. Gulea, Polyhedron 29, 757 (2010)
S. Bige, Z. Kilie, Z. Hayvali, T. Hokelek, S. Safran, J. Chem. Sci. 121, 989 (2009)
Sh.M. Morgan, A.Z. El-Sonbati, H.R. Eissa, J. Mol. Liq. 240, 752 (2017)
M. Mirzaei, H. Eshtiagh-Hosseini, Z. Karrabi, K. Molčanov, E. Eydizadeh, J.T. Mague, A. Bauzá, A. Frontera, Cryst. Eng. Comm. 16, 5352 (2014)
H. Temel, J. Coord. Chem. 57, 723 (2004)
Sh.M. Morgan, A.Z. El-Sonbati, M.A. El-Mogazy, Appl. Organometal. Chem. 32, e4264 (2018)
N. Tezer, N. Karakus, J. Mol. Model. 15, 223 (2009)
M.A. Diab, A.Z. El-Sonbati, Sh.M. Morgan, M.A. El-Mogazy, Appl. Organometal. Chem. 32, e4378 (2018)
M.I. Abou-Dobara, N.F. Omar, M.A. Diab, A.Z. El-Sonbati, Sh.M. Morgan, M.A. El-Mogazy, J. Cell. Biochem. 120, 1667 (2019)
A.A. Fadda, S. Bondock, R. Rabie, Monatsh. Chem. 13, 153 (2008)
B.H. Amin, M.I. Abou-Dobara, M.A. Diab, E.A. Gomaa, M.A. El-Mogazy, A.Z. El-Sonbati, M.S. EL-Ghareib, M.A. Hussien, H.M. Salama, Appl Organometl Chem 34, 5689 (2020)
S.G. Nozha, Sh.M. Morgan, S.E. Abu Ahmed, M.A. El-Mogazy, M.A. Diab, A.Z. El-Sonbati, M.I. Abou-Dobara, J. Mol. Struct. 1227, 129525 (2021)
A.Z. El-Sonbati, M.A. Diab, Sh.M. Morgan, J. Mol. Liq. 225, 195 (2017)
T.H. Al-Noor, M.R. Aziz, A.T.A.L. Jeboori, Int. J. Techn. Res. Appl. 2, 187 (2014)
G. Thiyagarajan, A. Pandey, P. Mayer, A. Thamaraichelvan, Indi. J. Chem. 53, 200 (2014)
R.O. Shaibu, G.M. Watkins, Ife J. Sci. 17, 31 (2015)
K.J. Donde, J. Chem. Pharm. Res. 7, 798 (2015)
P.A. Vigato, S. Tamburini, Coord. Chem. Rev. 248, 1717 (2004)
N.A. El-Ghamaz, A.Z. El-Sonbati, Sh.M. Morgan, J. Mol. Struct. 1027, 92 (2012)
A.Z. El-Sonbati, M.A. Diab, Sh.M. Morgan, M.Z. Balboula, Appl. Organometal. Chem. 32, e4059 (2018)
N.A. El-Ghamaz, M.A. Diab, A.Z. El-Sonbati, Sh.M. Morgan, O.L. Salem, Chem. Pap. 71, 2417 (2017)
Sh.M. Morgan, N.A. El-Ghamaz, M.A. Diab, J. Mol. Struct. 1160, 227 (2018)
R. Shirley, "The CRYSFIRE system for Automatic Powder Indexing": User's Manual, the Lattice Press, Guildford, Surrey GU2 7NL, England, (2000).
H.M. Refaat, H.A. El-Badway, Sh.M. Morgan, J. Mol. Liq. 220, 802 (2016)
G.G. Mohamed, A.A. El-Sherif, M.A. Saad, S.E.A. El-Sawy, Sh.M. Morgan, J. Mol. Liq. 223, 1311 (2016)
M.A. Diab, G.G. Mohamed, W.H. Mahmoud, A.Z. El-Sonbati, Sh.M. Morgan, S.Y. Abbas, Appl. Organometal. Chem. 33, e4945 (2019)
Sh.M. Morgan, M.A. Diab, A.Z. El-Sonbati, Appl. Organometal. Chem. 32, e4504 (2018)
M.I. Abou-Dobara, A.Z. El-Sonbati, Sh.M. Morgan, World J. Microbiol. Biotechnol. 29, 119 (2013)
A.Z. El-Sonbati, N.F. Omar, M.I. Abou-Dobara, M.A. Diab, M.A. El-Mogazy, Sh.M. Morgan, M.A. Hussien, A.A. El-Ghettany, J. Mol. Struct. 1239, 130481 (2021)
A.S. Fouda, A.M. Eldesoky, A.Z. El-Sonbati, S.F. Salam, Int. J. Electrochem. Sci. 9, 1867 (2014)
R.G. Parr, D.A. Donnelly, M. Levy, M. Palke, J. Chem. Phys. 68, 3801 (1978)
R.G. Parr, R.G. Pearson, J. Am. Chem. Soc. 105, 7512 (1983)
A. Pui, C. Pulicar, J.P. Mahy, Inorg. Chim. Acta 360, 2139 (2007)
W.J. Ceary, Coord. Chem Chem. Rev. 7, 81 (1971)
S. Chandan, D. Jain, A.K. Sharma, P. Sharma, Molecules 40, 174 (2009)
R.M. Patil, Acta Pol. Pharm Drug Res. 64, 345 (2007)
A.N. Evstopov, V.E. Yavorovskaya, E.S. Vorobev, Z.P. Kudonogova, L.N. Gritsenko, E.N. Schmidt, S.G. Medvedeva, D.V. Filimonov, T.P. Prishchep, A.S. Saratikov, Pharm. Chem. J. 26, 426 (1992)
Z.H. Abd El-Waheb, M.M. Mashaly, A.A. Faheim, Chem. Pap. 59, 25 (2005)
T. Radmakrishnan, P.T. Joseph, C.P. Prabhakaran, J. Inorg, Nucl. Chem. 38, 2217 (1976)
G. Shankar, R.R. Premkumar, S.K. Ramalingam, Polyhedron 5, 991 (1986)
R. Ramesh, P.K. Suganthy, K. Natarajan, Synth. React. Inorg. Met-Org. Chem. 26, 47 (1996)
A.Z. El-Sonbati, A.A.M. Belal, M.S. El-Gharib, Sh.M. Morgan, Spectrochim. Acta A 95, 627 (2012)
A.Z. El-Sonbati, M.A. Diab, A.A.M. Belal, Sh.M. Morgan, Spectrochim. Acta A 99, 353 (2012)
N.S. Biradar, V.B. Mahale, B.R. Havinale, Curr. Sci. 45, 124 (1976)
A. Syamal, K.S. Kale, Ind. J. Chem. 19A, 225 (1981)
M.A. Ali, A.H. Mirza, M. Nazimuddin, P.K. Dhar, R.J. Butcher, Trans. Met. Chem. 27, 27 (2002)
S.A. Ibrahim, A.M. Hammam, A.M. El-Dean Kamal, A.A. Mohamed, N.M. Rageh, Canadian J. App. Spec. 1, 38 (1993)
A.M. Abdel-Mawgoud, M.M. Hamed, H.M. Mostafa, Monatsh. Chem. 128, 553 (1997)
H.A. Dessouki, M. Gaber, A.L. El-Ansary, Egypt. J. Chem. 32, 639 (1989)
M.R. Mahmoud, S.A. Ibrahim, M.A. Hamed, Spectrochim. Acta A 39, 729 (1983)
E. Hofer, H. Hufimann, Tetrahedron Lett. 35, 3241 (1971)
N. Hassan, A.Z. El-Sonbati, M.G. El-Desouky, J. Mol. Liq. 242, 293 (2017)
A.Z. El-Sonbati, M.A. Diab, A.M. Eldesoky, Sh.M. Morgan, O.L. Salem, Appl. Organometal. Chem. 33, e4839 (2019)
A.Z. El-Sonbati, M.A. Diab, G.G. Mohamed, M.A. Saad, Sh.M. Morgan, S.E.A. El-Sawy, Appl. Organometal. Chem. 33, e4973 (2019)
R.M.N. Badger, J. Chem. Phys. 3, 710 (1935)
L.H. Jones, Spectrochim. Acta A 11, 409 (1959)
A.Z. El-Sonbati, Spetros. Lett. 30, 456 (1997)
S.P. McGlynn, J.K. Smith, W.C. Neely, J. Chem. Phys. 35, 105 (1961)
M. Mirzaei, H. Eshtiagh-Hosseini, A. Bauzá, S. Zarghami, P. Ballester, J.T. Mague, A. Frontera, Cryst. Eng. Comm. 16(2014), 6149 (2014)
F.K. Kneubuhl, J. Chem. Phys. 33, 1074 (1960)
H.R. Garmen, J.D. Swallen, J. Chem. Phys. 56, 3221 (1962)
A. Hassanpoor, M. Mirzaei, M.N. Shahrak, A.M. Majcher, Dalton Trans. 47, 13849 (2018)
A. Hassanpoor, Masoud Mirzaei, Hossein Eshtiagh-Hosseini, Anna Majcher. Cryst. Eng. Comm. 20, 3711 (2018)
A.B.P. Lever, Inorganic Electronic Spectroscopy (Elsevier, Amsterdam, 1968)
H. Eshtiagh-Hosseini, M. Mirzaei, S. Zarghami, A. Bauzá, A. Frontera, J.T. Mague, M. Habibi, M. Shamsipur, Cryst. Eng. Comm. 16, 1359 (2014)
A.H. Maki, B.R. McGarvey, J. Chem. Phys. 29, 31 (1958)
A.B.P. Lever, Mantovani. Inorg. Chem. 10, 817 (1971)
A.Z. El-Sonbati, M.A. Diab, Sh.M. Morgan, M.A. El-Mogazy, Appl Organometal Chem 32, e4530 (2018)
A.Z. El-Sonbati, M.A. Diab, Sh.M. Morgan, A.M. Eldesoky, M.Z. Balboula, Appl Organometal. Chem 32, e4207 (2018)
A.Z. El-Sonbati, W.H. Mahmoud, G.G. Mohamed, M.A. Diab, Sh.M. Morgan, S.Y. Abbas, Appl. Organometal. Chem. 33, e5048 (2019)
Sh.M. Morgan, M.A. Diab, A.Z. El-Sonbati, Appl. Organometal. Chem. 32, e4305 (2018)
Sh.M. Morgan, M.A. Diab, A.Z. El-Sonbati, Appl. Organometal. Chem. 32, e4281 (2018)
J. Laugier and B. Bochu, LMGP-suite suite of programs for the interpretation of X-ray experiments, ENSP/Laboratoire des Materiauxet du genie physique, Saint Martin d’Heres, France, 2000, pp. BP46.38042.
H. Eshtiagh-Hosseini, M. Mirzaei, M. Biabani, V. Lippolis, M. Chahkandi, C. Bazzicalupi, Cryst. Eng. Comm. 15, 6752 (2013)
A.W. Coats, J.P. Redfern, Nature 201, 68 (1964)
H.H. Horowitz, G. Metzger, Anal. Chem. 35, 1464 (1963)
M.A. Diab, A.Z. El-Sonbati, N.A. El-Ghamaz, Sh.M. Morgan, O. El-Shahat, Eur. Polym. J. 115, 268 (2019)
A.B.P. Lever, E. Mantovani, Inorg. Chem. 10, 817 (1971)
M.A. Al-Khaldi, K.Y. Al-qahtani, J. Mater. Environ. Sci. 4(5), 593 (2013)
A.S. Fouda, M.A. Diab, A.Z. El-Sonbati, S.A. Hassan, J. Electrochem. Sci. 12, 5072 (2017)
D.D. Macdonald, M.C.H. Mckubre, "Impedance measurements in electrochemical systems" Modern Aspects of Electrochemistry, J.O’M. Bockris, B.E. Conway, R.E. White, Eds., Plenum Press, New York 14 1982 pp. 61–150.
F. Mansfeld, Corrosion 36, 301 (1981)
C. Gabrielli, “Identification of Electrochemical processes by Frequency Response Analysis” Solarton Instrumentation Group, 1980.
M. El Achouri, S. Kertit, H.M. Gouttaya, B. Nciri, Y. Bensouda, L. Perez, M.R. Infante, K. Elkacemi, Prog. Org. Coat. 43, 267 (2001)
A. Anejjar, A. Zarrouk, R. Salghi, H. Zarrok, D. Ben Hmamou, B. Hammouti, B. Elmahi, S.S. Al-Deyab, the ecidenc. J Mater Environ Sci 4, 583 (2013)
S.F. Mertens, C. Xhoffer, B.C. Decooman, E. Temmerman, Corrosion 53, 381 (1997)
G. Trabanelli, C. Montecelli, V. Grassi, A. Frignani, J. Cem. Concr. Res. 35, 1804 (2005)
F.m. Reis, H.G. de Melo, I. Costa, J. Electrochem. Acta 51 (2006) 1780.
E. McCafferty, N. Hackerman, J. Electrochem. Soc. 119, 999 (1972)
A.S. Fouda, M.A. Diab, A.Z. El-Sonbati, Sh.A. Hassan, Int. J. Electrochem. Sci. 12, 5072 (2017)
H. Ma, S. Chen, L. Niu, S. Zhao, S. Li, D. Li, J. Appl. Electrochem. 32, 65 (2002)
E. Kuş, F. Mansfeld, Corros. Sci. 48, 965 (2006)
G.A. Caigman, S.K. Metcalf, E.M. Holt, J. Chem. Cryst. 30, 415 (2000)
Acknowledgements
The authors are grateful to Mr. Mohamed Marzouk El-Zahed, Assistant Lecturer of Microbiology at Botany and Microbiology Department, Faculty of Science, Damietta University, for his effort for detecting antimicrobial activities of the synthesized compounds.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
El-Sonbati, A.Z., Diab, M.A., Abou-Dobara, M.I. et al. Synthesis, characterization, electrochemical studies and antimicrobial activities of metal complexes. J IRAN CHEM SOC 19, 979–1002 (2022). https://doi.org/10.1007/s13738-021-02354-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02354-1