Abstract
Purpose
As epigenetic modifications like chromatin histone modifications have been suggested to play a role in the pathophysiology of Diabetic Nephropathy (DN) and are also found to be regulated by microRNAs. Our main purpose was to explore the role of microRNA in histone modulations associated with DN. There is downregulation of miR-29b due to advanced glycation end products in diabetes. Histone Deacetylase-4 (HDAC4) is amongst the histone modulators which promotes podocytes’ impairment and upregulates transforming growth factor-1 (TGF-β1) leading to renal fibrosis. Moreover, macrophage infiltration causes podocytes’ apoptosis and IL-6 mediated inflammation. As miR-29b is downregulated in diabetes and HDAC4, TGF-β1 and IL-6 could be the possible therapeutic targets in DN, our study was focussed on unveiling the role of miR-29b in modulation of HDAC4 and hence, in podocyte dysfunction and renal fibrosis in DN.
Methods
In silico analysis and luciferase assay were done to study the interaction between miR-29b and HDAC4. In-vitro DN model was developed in podocytes and miR-29b mimics were transfected. Also, podocytes were co-cultured with macrophage and miR-29b mimics were transfected. At the end, in-vivo DN model was generated in C57BL/6 J male mice and the effect of miR-29b mimics was reconfirmed.
Results
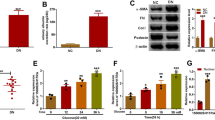
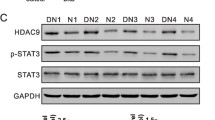
It was found that miR-29b targets the 3′ untranslated region of HDAC4. In both in-vitro and in-vivo DN model, downregulation of miR-29b and subsequent increase in HDAC4 expression was observed. The miR-29b mimics suppressed podocytes’ inflammation mediated through macrophages and attenuated HDAC4 expression, glomerular damage and renal fibrosis.
Conclusion
This study concludes that miR-29b regulates the expression of HDAC4 which plays a role in controlling renal fibrosis and podocytes’ impairment in DN.








Similar content being viewed by others
References
Siegerist F, et al. Acute podocyte injury is not a stimulus for podocytes to migrate along the glomerular basement membrane in zebrafish larvae. Sci Rep. 2017;7:43655.
Nagata M. Podocyte injury and its consequences. Kidney Int. 2016;89(6):1221–30.
Lin C-L, et al. MicroRNA-29a promotion of Nephrin acetylation ameliorates hyperglycemia-induced Podocyte dysfunction. J Am Soc Nephrol. 2014;25(8):1698–709.
Kato M, Natarajan R. Diabetic nephropathy—emerging epigenetic mechanisms. Nat Rev Nephrol. 2014;10(9):517–30.
Gilbert RE, et al. Histone deacetylase inhibition attenuates diabetes-associated kidney growth: potential role for epigenetic modification of the epidermal growth factor receptor. Kidney Int. 2011;79(12):1312–21.
de Groh ED, Swanhart LM, Cosentino CC, Jackson RL, Dai W, Kitchens CA, et al. Inhibition of histone deacetylase expands the renal progenitor cell population. J Am Soc Nephrol. 2010;21(5):794–802.
Jiang M, et al. Autophagy in proximal tubules protects against acute kidney injury. Kidney Int. 2012;82(12):1271–83.
Fang L, Zhou Y, Cao H, Wen P, Jiang L, He W, et al. Autophagy attenuates diabetic glomerular damage through protection of hyperglycemia-induced podocyte injury. PLoS One. 2013;8(4):e60546.
Wang X, et al. Histone deacetylase 4 selectively contributes to podocyte injury in diabetic nephropathy. Kidney Int. 2014;86(4):712–25.
Wang B, Komers R, Carew R, Winbanks CE, Xu B, Herman-Edelstein M, et al. Suppression of microRNA-29 expression by TGF-β1 promotes collagen expression and renal fibrosis. J Am Soc Nephrol. 2012;23(2):252–65.
Chen H-Y, Zhong X, Huang XR, Meng XM, You Y, Chung AC, et al. MicroRNA-29b inhibits diabetic nephropathy in db/db mice. Mol Ther. 2014;22(4):842–53.
Mannaerts I, Eysackers N, Onyema OO, van Beneden K, Valente S, Mai A, et al. Class II HDAC inhibition hampers hepatic stellate cell activation by induction of microRNA-29. PLoS One. 2013;8(1):e55786.
Zhou L, Wang L, Lu L, Jiang P, Sun H, Wang H. A novel target of microRNA-29, Ring1 and YY1-binding protein (Rybp), negatively regulates skeletal myogenesis. J Biol Chem. 2012;287(30):25255–65.
Bechtel W, Helmstädter M, Balica J, Hartleben B, Kiefer B, Hrnjic F, et al. Vps34 deficiency reveals the importance of endocytosis for podocyte homeostasis. J Am Soc Nephrol. 2013;24(5):727–43.
Amodio N, Stamato MA, Gullà AM, Morelli E, Romeo E, Raimondi L, et al. Therapeutic targeting of miR-29b/HDAC4 epigenetic loop in multiple myeloma. Mol Cancer Ther. 2016;15(6):1364–75.
Gondaliya P, et al. miR29b regulates aberrant methylation in In-Vitro diabetic nephropathy model of renal proximal tubular cells. PLOS ONE. 2018;13(11):e0208044.
You H, Gao T, Cooper TK, Brian Reeves W, Awad AS. Macrophages directly mediate diabetic renal injury. American Journal of Physiology-Renal Physiology. 2013;305(12):F1719–27.
Guo Y, Song Z, Zhou M, Yang Y, Zhao Y, Liu B, et al. Infiltrating macrophages in diabetic nephropathy promote podocytes apoptosis via TNF-α-ROS-p38MAPK pathway. Oncotarget. 2017;8(32):53276–87.
Das Gupta K, Shakespear MR, Iyer A, Fairlie DP, Sweet MJ. Histone deacetylases in monocyte/macrophage development, activation and metabolism: refining HDAC targets for inflammatory and infectious diseases. Clinical & translational immunology. 2016;5(1):e62–2.
Wang B, Liu TY, Lai CH, Rao YH, Choi MC, Chi JT, et al. Glycolysis-dependent histone deacetylase 4 degradation regulates inflammatory cytokine production. Mol Biol Cell. 2014;25(21):3300–7.
Lian X, Chen X, Sun J, An G, Li X, Wang Y, et al. MicroRNA-29b inhibits supernatants from silica-treated macrophages from inducing extracellular matrix synthesis in lung fibroblasts. Toxicology Research. 2017;6(6):878–88.
Liu BC, Song X, Lu XY, Li DT, Eaton DC, Shen BZ, et al. High glucose induces podocyte apoptosis by stimulating TRPC6 via elevation of reactive oxygen species. Biochim Biophys Acta. 2013;1833(6):1434–42.
Wing MR, et al. DNA methylation profile associated with rapid decline in kidney function: findings from the CRIC study. Nephrology Dialysis Transplantation. 2014;29(4):864–72.
Wei Q, Dong Z. HDAC4 blocks autophagy to trigger podocyte injury: non-epigenetic action in diabetic nephropathy. Kidney Int. 2014;86(4):666–8.
Wolf G, Chen S, Ziyadeh FN. From the periphery of the glomerular capillary wall toward the center of disease: podocyte injury comes of age in diabetic nephropathy. Diabetes. 2005;54(6):1626–34.
Mundel P, Shankland SJ. Podocyte biology and response to injury. J Am Soc Nephrol. 2002;13(12):3005–15.
Khullar, M., B.S. Cheema, and S.K. Raut, Emerging Evidence of Epigenetic Modifications in Vascular Complication of Diabetes. Front Endocrinol, 2017. 8(237).
Yi C, Yu L. How does acetylation regulate autophagy? Autophagy. 2012;8(10):1529–30.
Sankrityayan H, Kulkarni YA, Gaikwad AB. Diabetic nephropathy: the regulatory interplay between epigenetics and microRNAs. Pharmacol Res. 2019;141:574–85.
Tanji N, Markowitz GS, Fu C, Kislinger T, Taguchi A, Pischetsrieder M, et al. Expression of advanced glycation end products and their cellular receptor RAGE in diabetic nephropathy and nondiabetic renal disease. J Am Soc Nephrol. 2000;11(9):1656–66.
Benigni A, Gagliardini E, Tomasoni S, Abbate M, Ruggenenti P, Kalluri R, et al. Selective impairment of gene expression and assembly of nephrin in human diabetic nephropathy. Kidney Int. 2004;65(6):2193–200.
Inoki K, Mori H, Wang J, Suzuki T, Hong S, Yoshida S, et al. mTORC1 activation in podocytes is a critical step in the development of diabetic nephropathy in mice. J Clin Invest. 2011;121(6):2181–96.
Schoeler D, Grützkau A, Henz BM, Küchler J, Krüger-Krasagakis S. Interleukin-6 enhances whereas tumor necrosis factor α and interferons inhibit integrin expression and adhesion of human mast cells to extracellular matrix proteins. J Investig Dermatol. 2003;120(5):795–801.
Zhang XL, Topley N, Ito T, Phillips A. Interleukin-6 regulation of transforming growth factor (TGF)-β receptor compartmentalization and turnover enhances TGF-β1 signaling. J Biol Chem. 2005;280(13):12239–45.
Villiger P, et al. IL-1 beta and IL-6 selectively induce transforming growth factor-beta isoforms in human articular chondrocytes. J Immunol. 1993;151(6):3337–44.
Zhang Y, et al. Deletion of interleukin-6 alleviated interstitial fibrosis in streptozotocin-induced diabetic cardiomyopathy of mice through affecting TGFβ1 and miR-29 pathways. Sci Rep. 2016;6:23010.
Nogueira, A., M.J. Pires, and P.A. Oliveira, Pathophysiological Mechanisms of Renal Fibrosis: A Review of Animal Models and Therapeutic Strategies. In vivo (Athens, Greece), 2017. 31(1): p. 1–22.
Hsu Y-C, et al. Protective effects of miR-29a on diabetic glomerular dysfunction by modulation of DKK1/Wnt/β-catenin signaling. Sci Rep. 2016;6:30575.
Chow FY, et al. Macrophages in streptozotocin-induced diabetic nephropathy: potential role in renal fibrosis. Nephrology Dialysis Transplantation. 2004;19(12):2987–96.
Simpson K, Wonnacott A, Fraser DJ, Bowen T. MicroRNAs in diabetic nephropathy: from biomarkers to therapy. Current diabetes reports. 2016;16(3):35–5.
Wang G, et al. Urinary miR-21, miR-29, and miR-93: novel biomarkers of fibrosis. Am J Nephrol. 2012;36(5):412–8.
Zhong X, Chung AC, Chen HY, Dong Y, Meng XM, Li R, et al. miR-21 is a key therapeutic target for renal injury in a mouse model of type 2 diabetes. Diabetologia. 2013;56(3):663–74.
Acknowledgments
The research was carried out at National Institute of Pharmaceutical Education and Research- Ahmedabad with the financial support from the Department of Pharmaceuticals, Ministry of Chemicals and Fertilizers, Government of India.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Animals studies
All applicable institutional guidelines for the care and use of animals were followed. All procedures performed in this study involving animals were in accordance with the ethical standards of the institution at which the study was conducted (NIPER-A institutional animal ethics committee, NIPER-A/IAEC/2017/032).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 498 kb)
Rights and permissions
About this article
Cite this article
Gondaliya, P., P. Dasare, A., Jash, K. et al. miR-29b attenuates histone deacetylase-4 mediated podocyte dysfunction and renal fibrosis in diabetic nephropathy. J Diabetes Metab Disord 19, 13–27 (2020). https://doi.org/10.1007/s40200-019-00469-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-019-00469-0