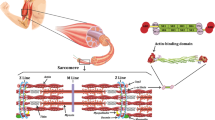
Abstract
The ability of skeletal muscles to produce force at a high velocity, which is crucial for success in power and sprint performance, is strongly influenced by genetics and without the appropriate genetic make-up, an individual reduces his/her chances of becoming an exceptional power or sprinter athlete. Several genetic variants (i.e. polymorphisms) have been associated with elite power and sprint performance in the last few years and the current paradigm is that elite performance is a polygenic trait, with minor contributions of each variant to the unique athletic phenotype. The purpose of this review is to summarize the specific knowledge in the field of genetics and elite power performance, and to provide some future directions for research in this field. Of the polymorphisms associated with elite power and sprint performance, the α-actinin-3 R577X polymorphism provides the most consistent results. ACTN3 is the only gene that shows a genotype and performance association across multiple cohorts of elite power athletes, and this association is strongly supported by mechanistic data from an Actn3 knockout mouse model. The angiotensin-1 converting enzyme insertion/deletion polymorphism (ACE I/D, registered single nucleotide polymorphism [rs]4646994), angiotensinogen (AGT Met235Thr rs699), skeletal adenosine monophosphate deaminase (AMPD1) Gln(Q)12Ter(X) [also termed C34T, rs17602729], interleukin-6 (IL-6 −174 G/C, rs1800795), endothelial nitric oxide synthase 3 (NOS3 −786 T/C, rs2070744; and Glu298Asp, rs1799983), peroxisome proliferator-activated receptor-α (PPARA Intron 7 G/C, rs4253778), and mitochondrial uncoupling protein 2 (UCP2 Ala55Val, rs660339) polymorphisms have also been associated with elite power performance, but the findings are less consistent. In general, research into the genetics of athletic performance is limited by a small sample size in individual studies and the heterogeneity of study samples, often including athletes from multiple-difference sporting disciplines. In the future, large, homogeneous, strictly defined elite power athlete cohorts need to be established though multinational collaboration, so that meaningful genome-wide association studies can be performed. Such an approach would provide unbiased identification of potential genes that influence elite athletic performance.



Similar content being viewed by others
References
Macarthur DG, North KN. Genes and human elite athletic performance. Hum Genet. 2005;116(5):331–9.
Druzhevskaya AM, Ahmetov II, Astratenkova IV, et al. Association of the ACTN3 R577X polymorphism with power athlete status in Russians. Eur J Appl Physiol. 2008;103(6):631–4.
Eynon N, Ruiz JR, Oliveira J, et al. Genes and elite athletes: a roadmap for future research. J Physiol. 2011;589(13):3063–70.
Stubbe JH, Boomsma DI, Vink JM, et al. Genetic influences on exercise participation in 37,051 twin pairs from seven countries. PloS one. 2006;1:e22.
De Moor MH, Spector TD, Cherkas LF, et al. Genome-wide linkage scan for athlete status in 700 British female DZ twin pairs. Twin Res Hum Genet. 2007;10(6):812–20.
Bouchard C, Sarzynski MA, Rice TK, et al. Genomic predictors of the maximal O uptake response to standardized exercise training programs. J Appl Physiol. 2011;110(5):1160–70.
Peeters MW, Thomis MA, Beunen GP, et al. Genetics and sports: an overview of the pre-molecular biology era. Med Sport Sci. 2009;54:28–42.
Calvo M, Rodas G, Vallejo M, et al. Heritability of explosive power and anaerobic capacity in humans. Eur J Appl Physiol. 2002;86(3):218–25.
Hagberg JM, Rankinen T, Loos RJ, et al. Advances in exercise, fitness, and performance genomics in 2010. Med Sci Sports Exerc. 2011;43(5):743–52.
Rankinen T, Roth SM, Bray MS, et al. Advances in exercise, fitness, and performance genomics. Med Sci Sports Exerc. 2010;42(5):835–46.
Roth SM, Rankinen T, Hagberg JM, et al. Advances in exercise, fitness, and performance genomics in 2011. Med Sci Sports Exerc. 2012;44(5):809–17.
Ahmetov II, Williams AG, Popov DV, et al. The combined impact of metabolic gene polymorphisms on elite endurance athlete status and related phenotypes. Hum Genet. 2009;126(6):751–61.
Ruiz JR, Gomez-Gallego F, Santiago C, et al. Is there an optimum endurance polygenic profile? J Physiol. 2009;587(Pt 7):1527–34.
Mole PA, Oscai LB, Holloszy JO. Adaptation of muscle to exercise: increase in levels of palmityl Coa synthetase, carnitine palmityltransferase, and palmityl Coa dehydrogenase, and in the capacity to oxidize fatty acids. J Clin Invest. 1971;50(11):2323–30.
Spencer MR, Gastin PB. Energy system contribution during 200- to 1500-m running in highly trained athletes. Med Sci Sports Exerc. 2001;33(1):157–62.
Ahmetov, II, Druzhevskaya AM, Lyubaeva EV, et al. The dependence of preferred competitive racing distance on muscle fibre type composition and ACTN3 genotype in speed skaters. Exp Physiol. 2011;96(12):1302–10
Van Damme R, Wilson RS, Vanhooydonck B, et al. Performance constraints in decathletes. Nature. 2002;415(6873):755–6.
Seeman E, Hopper JL, Young NR, et al. Do genetic factors explain associations between muscle strength, lean mass, and bone density? A twin study. Am J Physiol. 1996;270(2 Pt 1):E320–7.
Thomis MA, Beunen GP, Maes HH, et al. Strength training: importance of genetic factors. Med Sci Sports Exerc. 1998;30(5):724–31.
Thomis MA, Beunen GP, Van Leemputte M, et al. Inheritance of static and dynamic arm strength and some of its determinants. Acta Physiol Scand. 1998;163(1):59–71.
MacArthur DG, North KN. A gene for speed? The evolution and function of alpha-actinin-3. Bioessays. 2004;26(7):786–95.
Mills M, Yang N, Weinberger R, et al. Differential expression of the actin-binding proteins, alpha-actinin-2 and -3, in different species: implications for the evolution of functional redundancy. Hum Mol Genet. 2001;10(13):1335–46.
North KN, Yang N, Wattanasirichaigoon D, et al. A common nonsense mutation results in alpha-actinin-3 deficiency in the general population. Nat Genet. 1999;21(4):353–4.
Yang N, MacArthur DG, Gulbin JP, et al. ACTN3 genotype is associated with human elite athletic performance. Am J Hum Genet. 2003;73(3):627–31.
Eynon N, Duarte JA, Oliveira J, et al. ACTN3 R577X polymorphism and Israeli top-level athletes. Int J Sports Med. 2009;30(9):695–8.
Niemi AK, Majamaa K. Mitochondrial DNA and ACTN3 genotypes in Finnish elite endurance and sprint athletes. Eur J Hum Genet. 2005;13(8):965–9.
Doring FE, Onur S, Geisen U, et al. ACTN3 R577X and other polymorphisms are not associated with elite endurance athlete status in the Genathlete study. J Sports Sci. 2010;28(12):1355–9.
Muniesa CA, Gonzalez-Freire M, Santiago C, et al. World-class performance in lightweight rowing: is it genetically influenced? A comparison with cyclists, runners and non-athletes. Br J Sports Med. 2010;44(12):898–901.
Saunders CJ, September AV, Xenophontos SL, et al. No association of the ACTN3 gene R577X polymorphism with endurance performance in Ironman Triathlons. Ann Hum Genet. 2007;71(Pt 6):777–81.
Scott RA, Irving R, Irwin L, et al. ACTN3 and ACE genotypes in elite Jamaican and US sprinters. Med Sci Sports Exerc. 2010;42(1):107–12.
Yang N, MacArthur DG, Wolde B, et al. The ACTN3 R577X polymorphism in East and West African athletes. Med Sci Sports Exerc. 2007;39(11):1985–8.
Ruiz JR, Fernández del Valle M, Verde Z, et al. ACTN3 R577X polymorphism does not influence explosive leg muscle power in elite volleyball players. Scand J Med Sci Sports. 2011;21(6):e34–41.
Papadimitriou ID, Papadopoulos C, Kouvatsi A, et al. The ACTN3 gene in elite Greek track and field athletes. Int J Sports Med. 2008;29(4):352–5.
Gineviciene V, Pranculis A, Jakaitiene A, et al. Genetic variation of the human ACE and ACTN3 genes and their association with functional muscle properties in Lithuanian elite athletes. Medicina. 2011;47(5):284–90.
Roth SM, Walsh S, Liu D, et al. The ACTN3 R577X nonsense allele is under-represented in elite-level strength athletes. Eur J Hum Genet. 2008;16(3):391–4.
Eynon N, Ruiz JR, Femia P, et al. The ACTN3 R577X polymorphism across three groups of elite male European athletes. PloS one. 2012;7(8):e43132.
Chiu LL, Wu YF, Tang MT, et al. ACTN3 genotype and swimming performance in Taiwan. Int J Sports Med. 2011;32(6):476–80.
Eynon N, Alves AJ, Meckel Y, et al. Is the interaction between HIF1A P582S and ACTN3 R577X determinant for power/sprint performance? Metabolism. 2010;59(6):861–5.
Eynon N, Alves AJ, Yamin C, et al. Is there an ACE ID—ACTN3 R577X polymorphisms interaction that influences sprint performance? Int J Sports Med. 2009;30(12):888–91.
Ruiz JR, Arteta D, Buxens A, et al. Can we identify a power-oriented polygenic profile? J Appl Physiol. 2010;108(3):561–6.
MacArthur DG, Seto JT, Raftery JM, et al. Loss of ACTN3 gene function alters mouse muscle metabolism and shows evidence of positive selection in humans. Nat Genet. 2007;39(10):1261–5.
MacArthur DG, Seto JT, Chan S, et al. An Actn3 knockout mouse provides mechanistic insights into the association between alpha-actinin-3 deficiency and human athletic performance. Hum Mol Genet. 2008;17(8):1076–86.
Chan S, Seto JT, MacArthur DG, et al. A gene for speed: contractile properties of isolated whole EDL muscle from an alpha-actinin-3 knockout mouse. Am J Physiol Cell Physiol. 2008;295(4):C897–904.
Berman Y, North KN. A gene for speed: the emerging role of alpha-actinin-3 in muscle metabolism. Physiology. 2010;25(4):250–9.
Vincent B, De Bock K, Ramaekers M, et al. ACTN3 (R577X) genotype is associated with fiber type distribution. Physiol Genomics. 2007;32(1):58–63.
Rigat B, Hubert C, Alhenc-Gelas F, et al. An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. J Clin Invest. 1990;86(4):1343–6.
Danser AH, Schalekamp MA, Bax WA, et al. Angiotensin-converting enzyme in the human heart: effect of the deletion/insertion polymorphism. Circulation. 1995;92(6):1387–8.
Myerson S, Hemingway H, Budget R, et al. Human angiotensin I-converting enzyme gene and endurance performance. J Appl Physiol. 1999;87(4):1313–6.
Costa AM, Silva AJ, Garrido ND, et al. Association between ACE D allele and elite short distance swimming. Eur J Appl Physiol. 2009;106(6):785–90.
Juffer P, Furrer R, Gonzalez-Freire M, et al. Genotype distributions in top-level soccer players: a role for ACE? Int J Sports Med. 2009;30(5):387–92.
Nazarov IB, Woods DR, Montgomery HE, et al. The angiotensin converting enzyme I/D polymorphism in Russian athletes. Eur J Hum Genet. 2001;9(10):797–801.
Woods D, Hickman M, Jamshidi Y, et al. Elite swimmers and the D allele of the ACE I/D polymorphism. Hum Genet. 2001;108(3):230–2.
Papadimitriou ID, Papadopoulos C, Kouvatsi A, et al. The ACE I/D polymorphism in elite Greek track and field athletes. J Sports Med Phys Fitness. 2009;49(4):459–63.
Sessa F, Chetta M, Petito A, et al. Gene polymorphisms and sport attitude in Italian athletes. Genet Test Mol Biomarkers. 2011;15(4):285–90.
Amir O, Amir R, Yamin C, et al. The ACE deletion allele is associated with Israeli elite endurance athletes. Exp Physiol. 2007;92(5):881–6.
Kim CH, Cho JY, Jeon JY, et al. ACE DD genotype is unfavorable to Korean short-term muscle power athletes. Int J Sports Med. 2010;31(1):65–71.
Zoossmann-Diskin A. The association of the ACE gene and elite athletic performance in Israel may be an artifact. Exp Physiol. 2008;93(11):1220 (author reply 1).
Montgomery HE, Clarkson P, Dollery CM, et al. Association of angiotensin-converting enzyme gene I/D polymorphism with change in left ventricular mass in response to physical training. Circulation. 1997;96(3):741–7.
Sadoshima J, Xu Y, Slayter HS, et al. Autocrine release of angiotensin II mediates stretch-induced hypertrophy of cardiac myocytes in vitro. Cell. 1993;75(5):977–84.
Silva GJ, Moreira ED, Pereira AC, et al. ACE gene dosage modulates pressure-induced cardiac hypertrophy in mice and men. Physiol Genomics. 2006;27(3):237–44.
Berk BC, Vekshtein V, Gordon HM, et al. Angiotensin II-stimulated protein synthesis in cultured vascular smooth muscle cells. Hypertension. 1989;13(4):305–14.
Geisterfer AA, Peach MJ, Owens GK. Angiotensin II induces hypertrophy, not hyperplasia, of cultured rat aortic smooth muscle cells. Circ Res. 1988;62(4):749–56.
Gordon SE, Davis BS, Carlson CJ, et al. ANG II is required for optimal overload-induced skeletal muscle hypertrophy. Am J Physiol Endocrinol Metab. 2001;280(1):E150–9.
Westerkamp CM, Gordon SE. Angiotensin-converting enzyme inhibition attenuates myonuclear addition in overloaded slow-twitch skeletal muscle. Am J Physiol Regul Integr Comp Physiol. 2005;289(4):R1223–31.
Charbonneau DE, Hanson ED, Ludlow AT, et al. ACE genotype and the muscle hypertrophic and strength responses to strength training. Med Sci Sports Exerc. 2008;40(4):677–83.
Folland J, Leach B, Little T, et al. Angiotensin-converting enzyme genotype affects the response of human skeletal muscle to functional overload. Exp Physiol. 2000;85(5):575–9.
Zhang B, Tanaka H, Shono N, et al. The I allele of the angiotensin-converting enzyme gene is associated with an increased percentage of slow-twitch type I fibers in human skeletal muscle. Clin Genet. 2003;63(2):139–44.
Zhang B, Shono N, Fan P, et al. Histochemical characteristics of soleus muscle in angiotensin-converting enzyme gene knockout mice. Hypertens Res. 2005;28(8):681–8.
Bae JS, Kang BY, Lee KO, et al. Genetic variation in the renin–angiotensin system and response to endurance training. Med Princ Pract. 2007;16(2):142–6.
Karjalainen J, Kujala UM, Stolt A, et al. Angiotensinogen gene M235T polymorphism predicts left ventricular hypertrophy in endurance athletes. J Am Coll Cardiol. 1999;34(2):494–9.
Gomez-Gallego F, Santiago C, Gonzalez-Freire M, et al. The C allele of the AGT Met235Thr polymorphism is associated with power sports performance. Appl Physiol Nutr Metab. 2009;34(6):1108–11.
Norman B, Sabina RL, Jansson E. Regulation of skeletal muscle ATP catabolism by AMPD1 genotype during sprint exercise in asymptomatic subjects. J Appl Physiol. 2001;91(1):258–64.
Rubio JC, Martin MA, Rabadan M, et al. Frequency of the C34T mutation of the AMPD1 gene in world-class endurance athletes: does this mutation impair performance? J Appl Physiol. 2005;98(6):2108–12.
Morisaki T, Gross M, Morisaki H, et al. Molecular basis of AMP deaminase deficiency in skeletal muscle. Proc Natl Acad Sci USA. 1992;89(14):6457–61.
Norman B, Glenmark B, Jansson E. Muscle AMP deaminase deficiency in 2% of a healthy population. Muscle Nerve. 1995;18(2):239–41.
Norman B, Mahnke-Zizelman DK, Vallis A, et al. Genetic and other determinants of AMP deaminase activity in healthy adult skeletal muscle. J Appl Physiol. 1998;85(4):1273–8.
Lucia A, Martin MA, Esteve-Lanao J, et al. C34T mutation of the AMPD1 gene in an elite white runner. Br J Sports Med. 2006;40(3):e7.
Fischer H, Esbjornsson M, Sabina RL, et al. AMP deaminase deficiency is associated with lower sprint cycling performance in healthy subjects. J Appl Physiol. 2007;103(1):315–22.
Cieszczyk P, Ostanek M, Leonska-Duniec A, et al. Distribution of the AMPD1 C34T polymorphism in Polish power-oriented athletes. J Sports Sci. 2012;30(1):31–5.
Petersen AM, Pedersen BK. The anti-inflammatory effect of exercise. J Appl Physiol. 2005;98(4):1154–62.
Fishman D, Faulds G, Jeffery R, et al. The effect of novel polymorphisms in the interleukin-6 (IL-6) gene on IL-6 transcription and plasma IL-6 levels, and an association with systemic-onset juvenile chronic arthritis. J Clin Invest. 1998;102(7):1369–76.
Terry CF, Loukaci V, Green FR. Cooperative influence of genetic polymorphisms on interleukin 6 transcriptional regulation. J Biol Chem. 2000;275(24):18138–44.
Bennermo M, Held C, Stemme S, et al. Genetic predisposition of the interleukin-6 response to inflammation: implications for a variety of major diseases? Clin Chem. 2004;50(11):2136–40.
Yamin C, Duarte JA, Oliveira JM, et al. IL6 (−174) and TNFA (−308) promoter polymorphisms are associated with systemic creatine kinase response to eccentric exercise. Eur J Appl Physiol. 2008;104(3):579–86.
Ruiz JR, Buxens A, Artieda M, et al. The −174 G/C polymorphism of the IL6 gene is associated with elite power performance. J Sci Med Sport. 2010;13(5):549–53.
Eynon N, Ruiz JR, Meckel Y, et al. Is the −174 C/G polymorphism of the IL6 gene associated with elite power performance? A replication study with two different Caucasian cohorts. Exp Physiol. 2011;96(2):156–62.
Cooke JP, Rossitch E Jr, Andon NA, et al. Flow activates an endothelial potassium channel to release an endogenous nitrovasodilator. J Clin Invest. 1991;88(5):1663–71.
Quyyumi AA, Dakak N, Andrews NP, et al. Contribution of nitric oxide to metabolic coronary vasodilation in the human heart. Circulation. 1995;92(3):320–6.
Heydemann A, McNally E. NO more muscle fatigue. J Clin Invest. 2009;119(3):448–50.
Hickner RC, Fisher JS, Ehsani AA, et al. Role of nitric oxide in skeletal muscle blood flow at rest and during dynamic exercise in humans. Am J Physiol. 1997;273(1 Pt 2):H405–10.
McConell GK, Kingwell BA. Does nitric oxide regulate skeletal muscle glucose uptake during exercise? Exerc Sport Sci Rev. 2006;34(1):36–41.
Nakayama M, Yasue H, Yoshimura M, et al. T-786–>C mutation in the 5′-flanking region of the endothelial nitric oxide synthase gene is associated with coronary spasm. Circulation. 1999;99(22):2864–70.
Wang XL, Sim AS, Wang MX, et al. Genotype dependent and cigarette specific effects on endothelial nitric oxide synthase gene expression and enzyme activity. FEBS Lett. 2000;471(1):45–50.
Yoshimura M, Yasue H, Nakayama M, et al. A missense Glu298Asp variant in the endothelial nitric oxide synthase gene is associated with coronary spasm in the Japanese. Hum Genet. 1998;103(1):65–9.
Hand BD, McCole SD, Brown MD, et al. NOS3 gene polymorphisms and exercise hemodynamics in postmenopausal women. Int J Sports Med. 2006;27(12):951–8.
Rankinen T, Rice T, Perusse L, et al. NOS3 Glu298Asp genotype and blood pressure response to endurance training: the HERITAGE family study. Hypertension. 2000;36(5):885–9.
Gomez-Gallego F, Ruiz JR, Buxens A, et al. The −786 T/C polymorphism of the NOS3 gene is associated with elite performance in power sports. Eur J Appl Physiol. 2009;107(5):565–9.
Kawada S, Ishii N. Skeletal muscle hypertrophy after chronic restriction of venous blood flow in rats. Med Sci Sports Exerc. 2005;37(7):1144–50.
Smith LW, Smith JD, Criswell DS. Involvement of nitric oxide synthase in skeletal muscle adaptation to chronic overload. J Appl Physiol. 2002;92(5):2005–11.
Liang H, Ward WF. PGC-1alpha: a key regulator of energy metabolism. Adv Physiol Educ. 2006;30(4):145–51.
Jamshidi Y, Montgomery HE, Hense HW, et al. Peroxisome proliferator-activated receptor alpha gene regulates left ventricular growth in response to exercise and hypertension. Circulation. 2002;105(8):950–5.
Flavell DM, Jamshidi Y, Hawe E, et al. Peroxisome proliferator-activated receptor alpha gene variants influence progression of coronary atherosclerosis and risk of coronary artery disease. Circulation. 2002;105(12):1440–5.
Ahmetov II, Mozhayskaya IA, Flavell DM, et al. PPARalpha gene variation and physical performance in Russian athletes. Eur J Appl Physiol. 2006;97(1):103–8.
Eynon N, Meckel Y, Sagiv M, et al. Do PPARGC1A and PPARalpha polymorphisms influence sprint or endurance phenotypes? Scand J Med Sci Sports. 2010;20(1):e145–50.
Rance KA, Johnstone AM, Murison S, et al. Plasma leptin levels are related to body composition, sex, insulin levels and the A55V polymorphism of the UCP2 gene. Int J Obes (Lond). 2007;31(8):1311–8.
Oktavianthi S, Trimarsanto H, Febinia CA, et al. Uncoupling protein 2 gene polymorphisms are associated with obesity. Cardiovasc Diabetol. 2012;11(1):41.
Martinez-Hervas S, Mansego ML, de Marco G, et al. Polymorphisms of the UCP2 gene are associated with body fat distribution and risk of abdominal obesity in Spanish population. Eur J Clin Invest. 2012;42(2):171–8.
Moran CN, Yang N, Bailey ME, et al. Association analysis of the ACTN3 R577X polymorphism and complex quantitative body composition and performance phenotypes in adolescent Greeks. Eur J Hum Genet. 2007;15(1):88–93.
Williams AG, Folland JP. Similarity of polygenic profiles limits the potential for elite human physical performance. J Physiol. 2008;586(1):113–21.
Buxens A, Ruiz JR, Arteta D, et al. Can we predict top-level sports performance in power vs endurance events? A genetic approach. Scand J Med Sci Sports. 2011;21(4):570–9.
Clarkson PM, Devaney JM, Gordish-Dressman H, et al. ACTN3 genotype is associated with increases in muscle strength in response to resistance training in women. J Appl Physiol. 2005;99(1):154–63.
Pimenta EM, Coelho DB, Cruz IR, et al. The ACTN3 genotype in soccer players in response to acute eccentric training. Eur J Appl Physiol. 2012;112(4):1495–503.
Seto JT, Chan S, Turner N, et al. The effect of alpha-actinin-3 deficiency on muscle aging. Exp Gerontol. 2011;46(4):292–302.
Alfred T, Ben-Shlomo Y, Cooper R, et al. ACTN3 genotype, athletic status, and life course physical capability: meta-analysis of the published literature and findings from nine studies. Hum Mutat. 2011;32(9):1008–18.
Delmonico MJ, Zmuda JM, Taylor BC, et al. Association of the ACTN3 genotype and physical functioning with age in older adults. J Gerontol A Biol Sci Med Sci. 2008;63(11):1227–34.
Garatachea N, Fiuza-Luces C, Torres-Luque G, et al. Single and combined influence of ACE and ACTN3 genotypes on muscle phenotypes in octogenarians. Eur J Appl Physiol. 2012;112(7):2409–20.
Seto JT, Lek M, Quinlan KG, et al. Deficiency of alpha-actinin-3 is associated with increased susceptibility to contraction-induced damage and skeletal muscle remodeling. Hum Mol Genet. 2011;20(15):2914–27.
Clarkson PM, Hoffman EP, Zambraski E, et al. ACTN3 and MLCK genotype associations with exertional muscle damage. J Appl Physiol. 2005;99(2):564–9.
MacArthur DG, Balasubramanian S, Frankish A, et al. A systematic survey of loss-of-function variants in human protein-coding genes. Science. 2012;335(6070):823–8.
Hong E, Park J. Sample size and statistical power calculations. Genomics Inform. 2012;10(2):117–22.
Santiago C, Gonzalez-Freire M, Serratosa L, et al. ACTN3 genotype in professional soccer players. Br J Sports Med. 2008;42(1):71–3.
Official Oympic Games 2012 Website. Men’s maraton online. http://www.london2012.com/athletics/event/men-marathon/index.html. Accessed 29 Apr 2013.
Acknowledgments
No funding was received to assist in the preparation of this manuscript. The authors have no conflicts of interest to declare that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eynon, N., Hanson, E.D., Lucia, A. et al. Genes for Elite Power and Sprint Performance: ACTN3 Leads the Way. Sports Med 43, 803–817 (2013). https://doi.org/10.1007/s40279-013-0059-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-013-0059-4