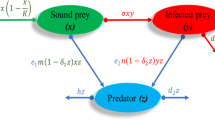
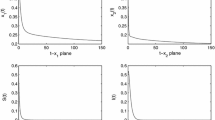
Abstract
In this research work, a delay-induced eco-epidemic model using a reconstructed Leslie–Gower-type growth rate is formulated and analyzed. An extended qualitative nature of the solutions of the model system like boundedness, strong uniform persistence, and permanence is examined to secure the longstanding viability of the system. The stability of the system is investigated at different stationary points, and sufficient conditions are obtained for the local as well as global stability. The dynamics of the delay-induced model system, including the Hopf bifurcation phenomenon, is rigorously studied around the coexisting equilibrium using the normal form method and center manifold theorem. Also, the length of the delay to preserve the stability of the coexisting equilibrium is evaluated. It is observed that the effect of infection on the total harvest is negligible, but the effort to harvest can reduce the infection and preserve the system’s stability. The results may help to determine the point of reference for disease persistence and extinction. Based on our analytical results, several numerical simulations are also performed.







Similar content being viewed by others
References
Auger P, Mchich R, Chowdhury T, Sallet G, Tchuente M, Chattopadhyay J (2009) Effects of a disease affecting a predator on the dynamics of a predator–prey system. J Theor Bio 258(3):344–351
Aziz-Alaoui MA (2002) Study of a Leslie–Gower-type tri-trophic population model. Chaos Solitons Fractals 14(8):1275–1293
Aziz-Alaoui MA, Okiye MD (2003) Boundedness and global stability for a predator-prey model with modified Leslie–Gower and Holling-type II schemes. Appl Math Lett 16(7):1069–1075
Bai Y, Li Y (2019) Stability and Hopf bifurcation for a stage-structured predator–prey model incorporating refuge for prey and additional food for predator. Adv Differ Equ 2019(1):42
Chakraborty K, Haldar S, Kar TK (2013) Global stability and bifurcation analysis of a delay-induced prey–predator system with stage structure. Non Dyn 73(3):1307–1325
Chakraborty K, Das K, Haldar S, Kar TK (2015) A mathematical study of an eco-epidemiological system on disease persistence and extinction perspective. Appl Math Comp 254:99–112
Chattopadhyay J, Arino O (1999) A predator–prey model with disease in the prey. Nonlinear Anal Theory Methods Appl 36(6):747–766
Chattopadhyay J, Bairagi N (2001) Pelicans at risk in Salton Sea—an eco-epidemiological model. Ecol Model 136(2–3):103–112
Chen F (2005) On a nonlinear nonautonomous predator–prey model with diffusion and distributed delay. J Comp Appl Math 180(1):33–49
Das KP (2016) Complex dynamics and its stabilization in an eco-epidemiological model with alternative food. Model Earth Syst Environ 2(4):1–12
Freedman HL, Rao VSH (1983) The trade-off between mutual interference and time lags in predator-prey systems. Bull Math Biol 45(6):991–1004
Gopalsamy K (2013) Stability and oscillations in delay differential equations of population dynamics, vol 74. Springer Science & Business Media, Berlin
Guo Y, Ji N, Niu B (2019) Hopf bifurcation analysis in a predator–prey model with time delay and food subsidies. Adv Differ Equ 2019(1):99
Haldar S, Chakraborty K, Kar TK (2015) Controllability of an eco-epidemiological system with disease transmission delay: a theoretical study. Appl Appl Math 10(1):382–420
Haldar S, Das K, Kar TK (2020) Dynamics of an exploited prey predator model induced by sigmoidal functional response in strong allee effect. Int J Ecol Dev 35(1):15–34
Hale JK, Lunel SMV (2013) Introduction to functional differential equations, vol 99. Springer Science & Business Media, New york
Haque M, Venturino E (2007) An eco-epidemiological model with disease in predator: the ratio-dependent case. Math Methods Appl Sci 30(14):1791–1809
Haque M, Zhen J, Venturino E (2009) An eco-epidemiological predator-prey model with standard disease incidence. Math Methods Appl Sci 32(7):875–898
Hassard BD, Kazarinoff ND, Wan YH (1981) Theory and application of Hopf bifurcation. Cambridge University Press, Cambridge
Hsieh YH, Hsiao CK (2008) Predator–prey model with disease infection in both populations. Math Med Biol A J IMA 25(3):247–266
Jana S, Kar TK (2013) Modeling and analysis of a prey–predator system with disease in the prey. Chaos Solitons Fractals 47:42–53
Kang A, Xue Y, Jin Z (2008) Dynamic behavior of an eco-epidemic system with impulsive birth. J Math Anal Appl 345(2):783–795
Kar TK, Jana S (2012) Stability and bifurcation analysis of a stage structured predator prey model with time delay. Appl Math Comput 219(8):3779–3792
Kar TK, Matsuda H (2006) Controllability of a harvested prey–predator system with time delay. J Biol Syst 14(02):243–254
Kar TK, Pahari UK (2006) Non-selective harvesting in prey–predator models with delay. Commun Nonlinear Sci Numer Simul 11(4):499–509
Khan QJ, Al Lawatia M, Al-Kharousi FA (2016) Predator–prey harvesting model with fatal disease in prey. Math Methods Appl Sci 39(10):2647–2658
Kooi BW, Venturino E (2016) Ecoepidemic predator–prey model with feeding satiation, prey herd behavior and abandoned infected prey. Math Biosci 274:58–72
Kot M (2001) Elements of mathematical ecology. Cambridge University Press, Cambridge
Kuang Y (1993) Delay differential equations: with applications in population dynamics, vol 191. Academic Press, Boston
Kumar S, Kharbanda H (2019) Chaotic behavior of predator–prey model with group defense and non-linear harvesting in prey. Chaos Solitons Fractals 119:19–28
Kundu S, Maitra S (2018) Dynamics of a delayed predator–prey system with stage structure and cooperation for preys. Chaos, Solitons Fractals 114:453–460
Kuznetsov YA (1997) Elements of applied bifurcation theory. Springer, New York
Lotka AJ (1925) Elements of physical biology. Williams and Wilkins, Baltimore
Maiti A, Bera SP, Samanta GP (2008) A prey–predator model with microparasite infection in the predator. J Biol Syst 16(02):219–239
Maiti AP, Dubey B, Tushar J (2017) A delayed prey–predator model with Crowley–Martin type functional response including prey refuge. Math Methods Appl Sci 40(16):5792–5809
Martin A, Ruan S (2001) Predator–prey models with delay and prey harvesting. J Math Biol 43(3):247–267
Meng XY, Huo HF, Zhang XB (2011) Stability and global Hopf bifurcation in a delayed food web consisting of a prey and two predators. Commun Nonlinear Sci Numer Simul 16(11):4335–4348
Mishra P, Raw SN, Tiwari B (2019) Study of a Leslie–Gower predator-prey model with prey defense and mutual interference of predators. Chaos Solitons Fractals 120:1–16
Mukhopadhyay B, Bhattacharyya R (2009) Role of predator switching in an eco-epidemiological model with disease in the prey. Ecol Model 220(7):931–939
Murray JD (2002) Mathematical biology. Springer, Berlin
Pathak S, Maiti A, Bera SP (2011) Effect of time-delay on a prey–predator model with microparasite infection in the predator. J Biol Syst 19(2):365–387
Peng M, Zhang Z (2018) Hopf bifurcation analysis in a predator–prey model with two time delays and stage structure for the prey. Adv Diff Equ 2018(1):251
Sahoo B, Poria S (2016) Effects of additional food in a susceptible-exposed-infected prey–predator model. Model Earth Syst Environ 2(3):160
Saito Y, Takeuchi Y (2003) A time-delay model for prey–predator growth with stage structure. Can Appl Math Q 11(3):293–302
Song Y, Yin T, Shu H (2017) Dynamics of ratio-dependent stage structured predator–prey model with delay. Math Meth Appl Sci 40(18):6451–6467
Venturino E (1994) The influence of diseases on Lotka–Volterra systems. Rocky Mt J Math 24:381–402
Venturino E (1995) Epidemics in predator-prey models: diseases in the prey. Math Popul Dyn Anal Heterog 1:381–393
Venturino E (2002) Epidemics in predator–prey models: disease in the predators. Math Med Biol 19(3):185–205
Volterra V (1926) Variazioni e fluttuazioni del número d’individui in specie animali conviventi. Mem Acad Lincei Roma 2:31–113
Xiao Y, Chen L (2001) Modeling and analysis of a predator–prey model with disease in the prey. Math Biosci 171(1):59–82
Xiao Y, Van Den Bosch F (2003) The dynamics of an eco-epidemic model with biological control. Ecol Model 168(1–2):203–214
Xu R, Ma Z (2008) Stability and Hopf bifurcation in a ratio-dependent predator–prey system with stage structure. Chaos Solitons Fractals 38(3):669–684
Yan XP, Zhang CH (2008) Hopf bifurcation in a delayed Lokta–Volterra predator–prey system. Nonlinear Anal Real World Appl 9(1):114–127
Zhang L, Zhang C (2010) Rich dynamic of a stage-structured prey–predator model with cannibalism and periodic attacking rate. Commun Nonlinear Sci Numer Simul 15(12):4029–4040
Acknowledgements
Dr. S. Haldar is very much thankful to Dr. Kunal Chakraborty, Scientist-D, INCOIS, Hyderabad, for his unconditional support. The research work of Anupam Khatua is financially supported by Department of Science and Technology-INSPIRE, Government of India (No. DST/INSPIRE Fellowship/2016/IF160667 dated 21st September 2016). Research of T. K. Kar is supported by the Council of Scientific and Industrial Research (CSIR), India (File No. 25(300)/19/EMR-II, dated: 16th May, 2019). We are also grateful to the anonymous reviewers and editor for their valuable comments and useful suggestions to improve the quality and presentation of the manuscript significantly.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A
Let \(x\) be the amount of susceptible prey that are contaminated by some infectious disease and \(\tau\) be the time to become infected from susceptible after contamination. This \(x\) amount does not increase the biomass of the infected class. There are some losses due to external hazards within the time delay \(\tau\). Let \(\gamma\) be the decay rate of that loss at any instant t. Then, after one unit of time, it would be \(x - \gamma x = \left( {1 - \gamma } \right)x\). After two unit time, it would be \(\left( {1 - \gamma } \right)x - \gamma \left( {1 - \gamma } \right)x = \left( {1 - \gamma } \right)^{2} x\), and that will be \(\left( {1 - \gamma } \right)^{\tau } x\) after \(\tau\) units of time. If the decay rate is calculated section wise by two times in one unit of time, then after \(\tau\) times, it would be \(\left( {1 - \frac{\gamma }{2}} \right)^{2\tau } x\). If the decay rate is calculated section wise by three times in one unit of time, then after \(\tau\) times, it would be \(\left( {1 - \frac{\gamma }{3}} \right)^{3\tau } x\). Similarly, if the decay rate is calculated section wise by \(m\) times in one unit of time, then after \(\tau\) times, it would be \(\left( {1 - \frac{\gamma }{m}} \right)^{m\tau } x\). Thus, letting \(m \to \infty\), we get continuous compounding amounts as \(\mathop {\lim }\limits_{m \to \infty } \left( {1 - \frac{\gamma }{m}} \right)^{m\tau } x = e^{ - \gamma \tau } x\).
Here, \(x = \frac{{\beta I\left( {t - \tau } \right)S\left( {t - \tau } \right)}}{{\left( {S\left( {t - \tau } \right) + I\left( {t - \tau } \right) + M} \right)}}\), and hence, \(e^{ - \gamma \tau } x = e^{ - \gamma \tau } \frac{{\beta I\left( {t - \tau } \right)S\left( {t - \tau } \right)}}{{\left( {S\left( {t - \tau } \right) + I\left( {t - \tau } \right) + M} \right)}}\).
Appendix B (proof of positivity)
From the system (1), and using the initial conditions, we have:
and \(P\left( t \right) = P\left( 0 \right)\exp \left( {\int\limits_{0}^{t} {\aleph_{3} {\text{d}}u} } \right) > 0\)
where \(\aleph_{1} = r - r\frac{S\left( u \right) + I\left( u \right)}{K} - \frac{\beta I\left( u \right)}{S\left( u \right) + I\left( u \right) + M} - \frac{\alpha P\left( u \right)}{a + S\left( u \right)} - q_{1} E\),
and \(\aleph_{3} = s - \frac{sP\left( u \right)}{\eta S\left( u \right) + \omega I\left( u \right) + L} - q_{3} E.\)
Therefore, all the solutions of the system (1) are positive. Hence, the desired result is obtained.
Appendix C (Proof of Proposition 3.1):
First, we state a result due to Chen (2005) as follows:
Lemma C.1
If \(a > 0,b > 0\) and \(\dot{x}(t) \ge ( \le )x(t)(b - ax^{\alpha } (t))\), (\(\alpha > 0\)), then for \(t \ge 0\) and \(x\left( 0 \right) > 0\), we have \(x\left( t \right) \ge \left( \le \right)\left( {\frac{b}{a}} \right)^{{\frac{1}{\alpha }}} \left[ {1 + \left( {\frac{{bx^{ - \alpha } \left( 0 \right)}}{a} - 1} \right)e^{ - b\alpha t} } \right]^{{ - \frac{1}{\alpha }}}\).
Now, using the first equation of (1), we have for all \(t > 0\):
According to the Lemma C.1, we get \(\mathop {\lim \sup }\limits_{t \to + \infty } S\left( t \right) \le \frac{{K\left( {r - q_{1} E} \right)}}{r} = Q_{1}\) (say).
Thus, we may conclude that, for any \(\varepsilon > 0\), \(\exists\) \(T_{1} > 0\), such that:\(S\left( t \right) < Q_{1} + \varepsilon ,\quad t > T_{1}\).
From the second equation of (1), we can write, \(\dot{I}\left( t \right) \ge - \left( {d + q_{2} E} \right)I\left( t \right)\).
Integrating between \(t - \tau\) to \(t\), we have \(I\left( {t - \tau } \right) \le I\left( t \right)e^{{\left( {d + q_{2} E} \right)\tau }}\).
Now, from Eq. (1), using the above inequality, we find:
Now, if we assume that \(\left( {d + q_{2} E} \right) > \beta e^{{\left( {d + q_{2} E - \gamma } \right)\tau }}\), then for any \(\varepsilon > 0\), \(\exists\) \(T_{2} > T_{1} > 0\) and \(Q_{2} > 0\), such that \(I\left( t \right) < Q_{2} + \varepsilon ,\quad t > T_{2}\), where \(\mathop {\lim \sup }\limits_{t \to + \infty } I\left( t \right) \le Q_{2}\).
Again, from the last equation of (1), we have:
Following Lemma C.1, we obtain \(\lim \sup_{t \to + \infty } P(t) \le (s - q_{3} E)(\eta Q_{1} + \omega Q_{2} + L) = Q_{3}\).
Therefore, for any \(\varepsilon > 0\), \(\exists\) \(T_{3} > T_{2} > 0\), such that \(P\left( t \right) < Q_{3} + \varepsilon ,\quad t > T_{3}\).
Furthermore, from the first equation of (1), we have:
By Lemma C.1, we can conclude that:
Then, we can say that, for any \(\varepsilon > 0\), there exists \(T_{4} > T_{3} > 0\), such that: \(S(t) > R_{1} + \varepsilon ,\quad t > T_{4}\).
From second equation of (1), we have, \(\dot{I}(t) > - (\sigma Q_{3} + d + q_{2} E)I(t)\).
Since \((\sigma Q_{3} + d + q_{2} E) > 0\) always, so we can say for any \(\varepsilon > 0\), there exists \(T_{5} > T_{4} > 0\) and \(R_{2} > 0\), such that, \(I(t) > R_{2} + \varepsilon ,\quad t > T_{5}\), where \(\lim \inf_{t \to + \infty } I(t) \ge R_{2}\).
Finally, from last equation of (1), we find \(\dot{P}\left( t \right) \ge P\left( t \right)\left( {\left( {s - q_{3} E} \right) - \frac{P\left( t \right)}{{\eta R_{1} + \omega R_{2} + L}}} \right)\).
Following Lemma C.1, we obtain \(\mathop {\lim \inf }\limits_{t \to + \infty } P\left( t \right) \ge \left( {s - q_{3} E} \right)\left( {\eta R_{1} + \omega R_{2} + L} \right) = R_{3}\).
Therefore, it can be said that, for any \(\varepsilon > 0\), there exists \(T_{6} > T_{5} > 0\), such that \(P\left( t \right) > R_{3} + \varepsilon ,\quad t > T_{6}\).
Hence, the theorem.
Appendix D: Permanence (Proof of the Theorem 3.2)
It is obvious that \(\lim \inf < \lim \sup\).
Therefore, \(\hbox{min} \left( {\lim \inf } \right) < \hbox{max} \left( {\lim \sup } \right)\) which implies that \(\nu < \vartheta\).
Meanwhile, we have established that for the persistence of the system, that all the constants \(Q_{i} ,R_{i} (i = 1,2,3)\) are greater than zero.
Therefore, \(\hbox{min} \left\{ {R_{1} ,R_{2} ,R_{3} } \right\} \ge 0\) and \(\hbox{max} \left\{ {Q_{1} ,Q_{2} ,Q_{3} } \right\} \ge 0\).
Hence, the desired results are done automatically. Therefore, the system is permanent.
Appendix E (stability of infection-free equilibrium)
One eigenvalue of the Jacobian matrix at \(E^{3} (S_{3} ,0,P_{3} )\) is given as \(\lambda_{1} = \frac{{\beta S^{*} (S^{*} + M)}}{{(S^{*} + M)^{2} }} - \sigma P^{*} - \tilde{d}\).
Now, \(\lambda_{1}\) will be negative if (i)\(\beta S^{*} (S^{*} + M) < (\sigma P^{*} + \tilde{d})(S^{*} + M)^{2} .\)
The other two eigenvalues are obtained from the following matrix:
The two characteristic roots of \(J_{3}\) will have negative real parts if \(Tr\left( {J_{1} } \right) < 0\) and \(\det \left( {J_{1} } \right) > 0\). Now, the above two conditions will be satisfied if the following two inequalities hold:
(2) \(\tilde{r} + \tilde{s} < \frac{{2rS^{*} }}{K} + \frac{{a\alpha P^{*} }}{{(a + S^{*} )^{2} }} + \frac{{2sP^{*} }}{{(\eta S^{*} + L)}}\)
(3) \(\tilde{r}\tilde{s} + \frac{{2sP^{*} }}{{(\eta S^{*} + L)}}\left( {\frac{{2rS^{*} }}{K} + \frac{{a\alpha P^{*} }}{{(a + S^{*} )^{2} }}} \right) + \frac{{\alpha s\eta S^{*} P^{*2} }}{{(\eta S^{*} + L)(a + S^{*} )}} > \frac{{2s\tilde{r}P^{*} }}{{(\eta S^{*} + L)}} + \tilde{s}\left( {\frac{{2rS^{*} }}{K} + \frac{{a\alpha P^{*} }}{{(a + S^{*} )^{2} }}} \right)\).
Hence, following the Routh–Hurwitz criteria, we may conclude that the system will be LAS at the infection-free stationary state if the inequalities (1), (2), and (3) are satisfied.
Rights and permissions
About this article
Cite this article
Haldar, S., Khatua, A., Das, K. et al. Modeling and analysis of a predator–prey type eco-epidemic system with time delay. Model. Earth Syst. Environ. 7, 1753–1768 (2021). https://doi.org/10.1007/s40808-020-00893-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40808-020-00893-9