Abstract
Since its original inception, a great deal has been learned about the nature, properties, and applications of the H-bond. This review summarizes some of the unexpected paths that inquiry into this phenomenon has taken researchers. The transfer of the bridging proton from one molecule to another can occur not only in the ground electronic state, but also in various excited states. Study of the latter process has developed insights into the relationships between the nature of the state, the strength of the H-bond, and the height of the transfer barrier. The enormous broadening of the range of atoms that can act as both proton donor and acceptor has led to the concept of the CH···O HB, whose properties are of immense importance in biomolecular structure and function. The idea that the central bridging proton can be replaced by any of various electronegative atoms has fostered the rapidly growing exploration of related noncovalent bonds that include halogen, chalcogen, pnicogen, and tetrel bonds.










Similar content being viewed by others
References
Latimer WM, Rodebush WH (1920) Polarity and ionization from the standpoint of the Lewis theory of valence. J Am Chem Soc 42:1419–1433
Pauling L (1940) The nature of the chemical bond. Cornell University Press, Ithaca, p 450
O’Reilly D, Stein RS, Patrascu MB, Jana SK, Kurian J, Moitessier N, Damha MJ (2018) Exploring atypical fluorine-hydrogen bonds and their effects on nucleoside conformations. Chem Eur J 24:16432–16439
Chaudhari SR, Mogurampelly S, Suryaprakash N (2013) Engagement of CF3 group in N-H…F-C hydrogen bond in the solution state: NMR spectroscopy and MD simulation studies. J Phys Chem B 117:1123–1129
Nadas J, Vukovic S, Hay BP (2012) Alkyl chlorides as hydrogen bond acceptors. Comput Theor Chem 988:75–80
Lai LL, Yang CM, Liu CC, Cheng KL, Wen YS, Hung CH, Luo TT, Kuo MY (2011) Direct evidence of a liquid-crystalline phase induced by intermolecular CH…Cl interactions on the basis of IR spectroscopy and theoretical simulations. Chem Eur J 17:111–116
Brammer L, Bruton EA, Sherwood P (2001) Understanding the behavior of halogens as hydrogen bond acceptors. Cryst Growth Des 1:277–290
Mishra KK, Singh SK, Kumar S, Singh G, Sarkar B, Madhusudhan MS, Das A (2019) Water-mediated selenium hydrogen-bonding in proteins: PDB analysis and gas-phase spectroscopy of model complexes. J Phys Chem A 123:5995–6002
Sahoo DK, Jena S, Dutta J, Rana A, Biswal HS (2019) Nature and strength of M–H···S and M–H···Se (M = Mn, Fe, & Co) hydrogen bond. J Phys Chem A 123:2227–2236
Das A, Mandal PK, Lovas FJ, Medcraft C, Walker NR, Arunan E (2018) The H2S dimer is hydrogen-bonded: direct confirmation from microwave spectroscopy. Angew Chem Int Ed 57:15199–15203
Wategaonkar S, Bhattacherjee A (2018) N–H···S interaction continues to be an enigma: experimental and computational investigations of hydrogen-bonded complexes of benzimidazole with thioethers. J Phys Chem A 122:4313–4321
Mundlapati VR, Gautam S, Sahoo DK, Ghosh A, Biswal HS (2017) Thioamide, a hydrogen bond acceptor in proteins and nucleic acids. J Phys Chem Lett 8:4573–4579
Mishra KK, Singh SK, Ghosh P, Ghosh D, Das A (2017) The nature of selenium hydrogen bonding: gas phase spectroscopy and quantum chemistry calculations. Phys Chem Chem Phys 19:24179–24187
Andersen CL, Jensen CS, Mackeprang K, Du L, Jørgensen S, Kjaergaard HG (2014) Similar strength of the NH···O and NH···S hydrogen bonds in binary complexes. J Phys Chem A 118:11074–11082
Mintz BJ, Parks JM (2012) Benchmark interaction energies for bologically relevant noncovalent complexes containing divalent sulfur. J Phys Chem A 116:1086–1092
Biswal HS, Wategaonkar S (2011) OH···X (X = O, S) hydrogen bonding in thetrahydrofuran and tetrahydrothiophene. J Chem Phys 135:134306
Biswal HS, Wategaonkar S (2009) Sulfur, not too far behind O, N, and C: SH···π hydrogen bond. J Phys Chem A 113:12774–12782
Møller KH, Hansen AS, Kjaergaard HG (2015) Gas phase detection of the NH–P hydrogen bond and importance of secondary interactions. J Phys Chem A 119:10988–10998
Viana RB, da Silva ABF (2015) Interaction between PH3 and small water clusters: understanding the electronic and spectroscopic properties. Comput Theor Chem 1059:35–44
Hansen AS, Du L, Kjaergaard HG (2014) Positively charged phosphorus as a hydrogen bond acceptor. J Phys Chem Lett 5:4225–4231
Schmidbaur H (2019) Proof of concept for hydrogen bonding to gold, Au···H − X. Angew Chem Int Ed 58:5806–5809
Wang P, Xu H-G, Cao G-J, Zhang W-J, Xu X-L, Zheng W-J (2017) Nonconventional hydrogen bonds between silver anion and nucleobases: size-selected anion photoelectron spectroscopy and density functional calculations. J Phys Chem A 121:8973–8981
Sanchez-de-Armas R, Ahlquist MSG (2015) On the nature of hydrogen bonds to platinum(II)—Which interaction can predict their strength? Phys Chem Chem Phys 17:812–816
Schmidbaur H, Raubenheimer HG, Dobrzanska L (2014) The gold-hydrogen bond, Au–H, and the hydrogen bond to gold, AuH–X. Chem Soc Rev 43:345–380
Falvello LR (2010) The hydrogen bond, front and center. Angew Chem Int Ed Engl 49:10045–10047
Rizzato S, Bergès J, Mason SA, Albinati A, Kozelka J (2010) Dispersion-driven hydrogen bonding: predicted hydrogen bond between water and patinum(II) identified by neutron diffraction. Angew Chem Int Ed Engl 49:7440–7443
Kryachko ES, Remacle F (2005) Three-gold clusters form nonconventional hydrogen bonds O–H…Au and N–H…Au with formamide and formic acid. Chem Phys Lett 404:142–149
Kozelka J (2015) Agostic and hydrogen-bonding X–H· · ·M interactions involving a d8 metal center: recent advances towards their understanding. In: Scheiner S (ed) Noncovalent forces, vol 19. Springer, Dordrecht, pp 129–158
Ventura E, Monte SAD, Fragoso W, Braga CF, Araújo RCMU (2006) Effects of π bond type, backbone size, and halogen on structural and spectroscopic properties of hydrogen-bonded complexes of the XH…π type between alkenes or alkynes and haloacids (HF and HCl). Int J Quantum Chem 106:1009–1019
Karle IL, Butcher RJ, Wolak MA, Filho DADS, Uchida M, Brédas JL, Kafafi ZH (2007) Cooperative CH···π interactions in the crystal structure of 2,5-di(3-biphenyl)-1,1-dimethyl-3,4-diphenyl-silole and Its effect on Its electronic properties. J Phys Chem C 111:9543–9547
Nishio M, Umezawa Y, Honda K, Tsuboyama S, Suezawa H (2009) CH/π hydrogen bonds in organic and organometallic chemistry. CrystEngComm 11:1757–1788
Gierszal KP, Davis JG, Hands MD, Wilcox DS, Slipchenko LV, Ben-Amotz D (2011) π-Hydrogen bonding in liquid water. J Phys Chem Lett 2:2930–2933
Kumar S, Pande V, Das A (2012) π-Hydrogen bonding wins over conventional hydrogen bonding interaction: a jet-cooled study of indole···furan heterodimer. J Phys Chem A 116:1368–1374
Mastrorilli P, Gallo V, Todisco S, Latronico M, Saielli G (2016) Uncovering intramolecular π-type hydrogen bonds in solution by NMR spectroscopy and DFT calculations. Chem Eur J 22:7964–7969
Aliev AE, Arendorf JRT, Pavlakos I, Moreno RB, Porter MJ, Rzepa HS, Motherwell WB (2015) Surfing π clouds for noncovalent interactions: Arenes versus alkenes. Angew Chem Int Ed 54:551–555
Grabowski SJ, Sokalski WA, Leszczynski J (2006) Can H…σ, π…H + …σ and σ…H + …σ interactions be classified as H-bonded? Chem Phys Lett 432:33–39
Grabowski SJ (2019) A − H…σ hydrogen bonds: dihydrogen and cycloalkanes as proton acceptors. ChemPhysChem 20:565–574
Alkorta I, Elguero J, Bene JED (2010) An ab initio investigation of the properties of H2:HX hydrogen-bonded complexes. Chem Phys Lett 489:159–163
Grabowski SJ (2013) Dihydrogen bond and X-H…σ interaction as sub-classes of hydrogen bond. J Phys Org Chem 26:452–459
Pirani F, Cappelletti D, Belpassi L, Tarantelli F (2013) Intermolecular interaction in the NH3–H2 and H2O–H2 complexes by molecular beam scattering experiments: the role of charge transfer. J Phys Chem A 117:12601–12607
Kvyatkovskaya EA, Nikitina EV, Khrustalev VN, Galmés B, Zubkov FI, Frontera A (in press) Through space “α-effect” between the O-bridge atoms in diepoxybenzo[de]isothiochromene derivatives. Eur J Org Chem. https://doi.org/10.1002/ejoc.201901169
Arunan E, Desiraju GR, Klein RA, Sadlej J, Scheiner S, Alkorta I, Clary DC, Crabtree RH, Dannenberg JJ, Hobza P, Kjaergaard HG, Legon AC, Mennucci B, Nesbitt DJ (2011) Definition of the hydrogen bond. Pure Appl Chem 83:1637–1641
Pimentel GC, McClellan AL (1960) The Hydrogen bond. Freeman, San Francisco
Vinogradov SN, Linnell RH (1971) Hydrogen bonding. Van Nostrand-Reinhold, New York
Joesten MD, Schaad LJ (1974) Hydrogen bonding. Marcel Dekker, New York, p 622
Gilli G, Gilli P (2009) The nature of the hydrogen bond. Oxford University Press, Oxford, p 313
Schuster P (1984) Hydrogen bonds, vol 120. Springer, Berlin, p 117
Schuster P, Zundel G, Sandorfy C (1976) The hydrogen bond. Recent developments in theory and experiments. North-Holland Publishing Co., Amsterdam
Grabowski SJ (2006) Hydrogen bonding—new insights. Springer, Dordrecht
Scheiner S (1997) Hydrogen bonding: a theoretical perspective. Oxford University Press, New York, p 375
Weller A (1956) Intramolecular proton transfer in excited states. Z Elektrochem 60:1144–1147
Taylor CA, El-Bayoumi MA, Kasha M (1969) Excited-state two-proton tautomerism in hydrogen-bonded N-heterocyclic base pairs. Proc Natl Acad Sci USA 63:253–260
Sengupta PK, Kasha M (1979) Excited-state proton transfer spectroscopy on 3-hyroxyflavone and quercetin. Chem Phys Lett 68:382–385
Chou PT, McMorrow D, Aartsma TJ, Kasha M (1984) The proton-transfer laser. Gain spectrum and amplification of spontaneous emission of 3-hydroxyflavone. J Phys Chem 88:4596–4599
Nishiya T, Yamauchi S, Hirota N, Baba M, Hanazaki I (1986) Fluorescence studies of the intramolecularly hydrogen-bonded molecules o-hydroxyacetophenone and salicylamide and related molecules. J Phys Chem 90:5730–5735
Nagaoka S, Fujita M, Takemura T, Baba H (1986) Fluorescence from an upper excited state of o-hydroxybenzaldehyde in the vapor phase. Chem Phys Lett 123:123–125
Ernsting NP, Nikolaus B (1986) Dye-laser pulse shortening by transient absorption following excited-state intramolecular proton transfer. Appl Phys B 39:155–164
Martinez ML, Cooper WC, Chou P-T (1992) A novel excited-state intramolecular proton transfer molecule, 10-hydroxybenzo[h]quinoline. Chem Phys Lett 193:151–154
Werner T (1979) Triplet deactivation in benzotriazole-type ultraviolet stabilizers. J Phys Chem 83:320–329
Tarkka RM, Zhang X, Jenekhe SA (1996) Electrically generated intramolecular proton transfer: electroluminescence and stimulated emission from polymers. J Am Chem Soc 118:9438–9439
Williams DL, Heller J (1970) Intramolecular proton transfer reactions in excited fluorescent compounds. J Phys Chem 74:4473–4480
Heller HJ, Blattmann HR (1973) Some aspects of stabilization of polymers against light. Pure Appl Chem 36:141–161
Latajka Z, Scheiner S (1992) Proton transfer in the ground and first excited triplet states of malonaldehyde. J Phys Chem 96:9764–9767
Luth K, Scheiner S (1994) Excited-state energetics and proton-transfer barriers in malonaldehyde. J Phys Chem 98:3582–3587
Yang Y, Li D, Li C, Liu Y, Jiang K (2019) Asymmetric substitution changes the UV-induced nonradiative decay pathway and the spectra behaviors of β-diketones. Spectrochim Acta Part A Mol Biomol Spectrosc 207:209–215
Tsutsumi T, Ono Y, Arai Z, Taketsugu T (2018) Visualization of the intrinsic reaction coordinate and global reaction route map by classical multidimensional scaling. J Chem Theory Comput 14:4263–4270
Zamastil J, Šimsa D (2017) Quantum effects and quantum chaos in multidimensional tunneling. Phys Rev E 96:062201
Nandipati KR, Kanakati AK, Singh H, Mahapatra S (2019) Controlled intramolecular H-transfer in malonaldehyde in the electronic ground state mediated through the conical intersection of 1nπ* and 1ππ* excited electronic states. Phys Chem Chem Phys 21:20018–20030
Coe JD, Martínez TJ (2006) Ab initio molecular dynamics of excited-state intramolecular proton transfer around a three-state conical intersection in malonaldehyde. J Phys Chem A 110:618–630
Sobolewski AL, Domcke W (1999) Photophysics of malonaldehyde: an ab initio study. J Phys Chem A 103:4494–4504
Luth K, Scheiner S (1995) Proton transfer in ground and excited electronic states of glyoxalmonohydrazine. J Phys Chem 99:7352–7359
Duan X, Scheiner S (1993) Ground and excited state intramolecular proton transfer in OCCNN ring. Chem Phys Lett 204:36–44
Rovira MC, Scheiner S (1995) Transfer of a proton between N atoms in excited electronic states of 1,5-diaza-1,3-pentadiene. J Phys Chem 99:9854–9861
Scheiner S, Kar T, Cuma M (1997) Excited state intramolecular proton transfer in anionic analogues of malonaldehyde. J Phys Chem A 101:5901–5909
Vener MV, Scheiner S (1995) Hydrogen bonding and proton transfer in the ground and lowest excited singlet states of o-hydroxyacetophenone. J Phys Chem 99:642–649
Cuma M, Scheiner S, Kar T (1999) Effect of adjoining aromatic ring upon excited state proton transfer. o-Hydroxybenzaldehyde. J Mol Struct (Theochem) 467:37–49
Vener MV, Scheiner S, Sokolov ND (1994) Theoretical study of hydrogen bonding and proton transfer in the ground and lowest excited singlet states of tropolone. J Chem Phys 101:9755–9765
Forés M, Scheiner S (1999) Effects of chemical substitution upon excited state proton transfer: fluoroderivatives of salicylaldimine. Chem Phys 246:65–74
Yi M, Scheiner S (1996) Proton transfer between phenol and ammonia in ground and excited electronic states. Chem Phys Lett 262:567–572
Scheiner S (2000) Theoretical studies of excited state proton transfer in small model systems. J Phys Chem A 104:5898–5909
Pang X, Jiang C, Xie W, Domcke W (2019) Photoinduced electron-driven proton transfer from water to an N-heterocyclic chromophore: nonadiabatic dynamics studies for pyridine–water clusters. Phys Chem Chem Phys 21:14073–14079
Pang X, Ehrmaier J, Wu X, Jiang C, Xie W, Sobolewski AL, Domcke W (2018) Photoinduced hydrogen-transfer reactions in pyridine-water clusters: insights from excited-state electronic-structure calculations. Chem Phys 515:550–556
Wu X, Karsili TNV, Domcke W (2017) Role of electron-driven proton-transfer processes in the ultrafast deactivation of photoexcited anionic 8-oxoGuanine-adenine and 8-oxoGuanine-cytosine base pairs. Molecules 22:135
Trung NT, Khanh PN, Carvalho AJP, Nguyen MT (2019) Remarkable shifts of Csp2-H and O–H stretching frequencies and stability of complexes of formic acid with formaldehydes and thioformaldehydes. J Comput Chem 40:1387–1400
Bednowitz AL, Post B (1966) Direct determination of the crystal structure of β-fumaric acid. Acta Cryst 21:566–571
Glasstone S (1937) The structure of some molecular complexes in the liquid phase. Trans Faraday Soc 33:200–214
Dippy JFJ (1939) The dissociation constants of monocarboxylic acids; Their measurement and their significance in theoretical organic chemistry. Chem Rev 25:151–211
Sutor DJ (1962) The C-H…O hydrogen bond in crystals. Nature 195:68–69
Karpfen A (2019) On the interaction of propynal with HNO, HF, HCl, H2O, CH3OH, and NH3: red- and blue-shifting hydrogen bonds and tetrel bonds. Comput Theor Chem 1160:1–13
Behera B, Das PK (2019) Blue-shifted hydrogen bonding in the gas phase CH/D3CN···HCCl3 complexes. J Phys Chem A 123:1830–1839
Zhang L, Li D (2019) An insight into intramolecular blue-shifting CH···π hydrogen bonds in 1,3-hexadien-5-yne and its halogen-substituted derivatives. Chem Phys 518:58–68
Scheiner S (2000) CH···O hydrogen bonding. In: Hargittai M, Hargittai I (eds) Advances in molecular structure research, vol 6. JAI Press, Stamford, pp 159–207
Masunov A, Dannenberg JJ, Contreras RH (2001) C–H bond-shortening upon hydrogen bond formation: influence of an electric field. J Phys Chem A 105:4737–4740
Pejov L, Hermansson K (2003) On the nature of blueshifting hydrogen bonds: ab initio and density functional studies of several fluoroform complexes. J Chem Phys 119:313–324
Qian W, Krimm S (2002) Vibrational spectroscopy of hydrogen bonding: origin of the different behavior of the C-H…O hydrogen bond. J Phys Chem A 106:6628–6636
Li X, Liu L, Schlegel HB (2002) On the physical origin of blue-shifted hydrogen bonds. J Am Chem Soc 124:9639–9647
Alabugin IV, Manoharan M, Peabody S, Weinhold F (2003) Electronic basis of improper hydrogen bonding: a subtle balance of hyperconjugation and rehybridization. J Am Chem Soc 125:5973–5987
Joseph J, Jemmis ED (2007) Red-, blue-, or no-shift hydrogen bonds: a unified explanation. J Am Chem Soc 129:4620–4632
Karpfen A, Kryachko ES (2009) On the intramolecular origin of the blue shift of A-H stretching frequencies: triatomic hydrides HAX. J Phys Chem A 113:5217–5223
Grabowski SJ (2011) Red- and blue-shifted hydrogen bonds: the bent rule from quantum theory of atoms in molecules perspective. J Phys Chem A 115:12789–12799
Mo Y, Wang C, Guan L, Braïda B, Hiberty PC, Wu W (2014) On the nature of blue shifting hydrogen bonds. Chem Eur J 20:8444–8452
Gu Y, Kar T, Scheiner S (1999) Fundamental properties of the CH…O interaction: Is it a true hydrogen bond? J Am Chem Soc 121:9411–9422
Scheiner S, Gu Y, Kar T (2000) Evaluation of the H-bonding properties of CH…O interactions based upon NMR spectra. J Mol Struct (Theochem) 500:441–452
Scheiner S, Grabowski SJ, Kar T (2001) Influence of hybridization and substitution upon the properties of the CH··O hydrogen bond. J Phys Chem A 105:10607–10612
Scheiner S, Kar T (2002) Red versus blue-shifting hydrogen bonds: Are there fundamental distinctions? J Phys Chem A 106:1784–1789
Gu Y, Kar T, Scheiner S (2000) Comparison of the CH…N and CH…O interactions involving substituted alkanes. J Mol Struct 552:17–31
Scheiner S, Kar T, Gu Y (2001) Strength of the CαH.O hydrogen bond of amino acid residues. J Biol Chem 276:9832–9837
Scheiner S, Kar T, Pattanayak J (2002) Comparison of various types of hydrogen bonds involving aromatic amino acids. J Am Chem Soc 124:13257–13264
Scheiner S (2006) Contributions of NH··O and CH··O H-bonds to the stability of β-sheets in proteins. J Phys Chem B 110:18670–18679
Pohl G, Plumley JA, Dannenberg JJ (2013) The interactions of phenylalanines in β-sheet-like structures from molecular orbital calculations using density functional theory (DFT), MP2, and CCSD(T) methods. J Chem Phys 138:245102
Wang C-S, Sun C-L (2010) Investigation on the individual contributions of N–H···O=C and C–H···O=C interactions to the binding energies of β-sheet models. J Comput Chem 31:1036–1044
Guo H, Gorin A, Guo H (2009) A peptide-linkage deletion procedure for estimate of energetic contributions of individual peptide groups in a complex environment: application to parallel β-sheets. Interdiscip Sci Comput Life Sci 1:12–20
Vener MV, Egorova AN, Fomin DP, Tsirel’son VG (2009) A quantum-topological analysis of noncovalent interactions in secondary polyalanine structures. Russ J Phys Chem B 3:541–547
Vener MV, Egorova AN, Fomin DP, Tsirelson VG (2007) QTAIM study of the closed-shell interactions in peptide secondary structures: a cluster treatment of oligo- and polyalanines. Chem Phys Lett 440:279–285
Parthasarathi R, Raman SS, Subramanian V, Ramasami T (2007) Bader’s electron density analysis of hydrogen bonding in secondary structural elements of proteins. J Phys Chem A 111:7141–7148
Scheiner S (2007) The strength with which a peptide group can form a hydrogen bond varies with the internal conformation of the polypeptide chain. J Phys Chem B 111:11312–11317
Scheiner S, Kar T (2005) Effect of solvent upon CH··O hydrogen bonds with implications for protein folding. J Phys Chem B 109:3681–3689
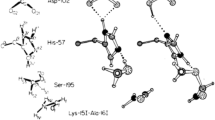
Scheiner S (2008) Analysis of catalytic mechanism of serine proteases. Viability of ring-flip hypothesis. J Phys Chem B 112:6837–6846
Horowitz S, Adhikari U, Dirk LMA, Del Rizzo PA, Mehl RA, Houtz RL, Al-Hashimi HM, Scheiner S, Trievel RC (2014) Manipulating Unconventional CH-Based Hydrogen Bonding in a Methyltransferase via Noncanonical Amino Acid Mutagenesis. ACS Chem Biol 9:1692–1697
Jones CR, Baruah PK, Thompson AL, Scheiner S, Smith MD (2012) Can a C-H···O interaction be a determinant of conformation. J Am Chem Soc 134:12064–12071
S. Scheiner, Dissection of the Factors Affecting Formation of a CH∙∙∙O H-Bond. A Case Study, Cryst. 5, 327 (2015)
Driver RW, Claridge TDW, Scheiner S, Smith MD (2016) Torsional and Electronic Factors Control the C − H···O Interaction. Chem Eur J 22:16513–16521
Adhikari U, Scheiner S (2013) The Magnitude and Mechanism of Charge Enhancement of CH··O H-bonds. J Phys Chem A 117:10551–10562
B. Nepal and S. Scheiner, Anionic CH…X− Hydrogen Bonds: Origin of Their Strength, Geometry, and Other Properties, Chem. Eur. J. 21, 1474-1481 (2015)
Nepal B, Scheiner S (2015) Microsolvation of Anions by Molecules Forming CH∙∙X− Hydrogen Bonds. Chem Phys 463:137–144
Scheiner S, Kar T (2008) Spectroscopic and structural signature of the CH–O H-bond. J Phys Chem A 112:11854–11860
Scheiner S (2009) Identification of spectroscopic patterns of CH–O H-bonds in dipeptides. J. Phys. Chem. B 113:10421–10427
Scheiner S (2010) Effect of CH…O hydrogen bond length on the geometric and spectroscopic features of the peptide unit of proteins. Int J Quantum Chem 110:2775–2783
Hassel O (1970) Structural aspects of interatomic charge-transfer bonding. Science 170:497–502
Lommerse JPM, Stone AJ, Taylor R, Allen FH (1996) The nature and geometry of intermolecular interactions between halogens and oxygen or nitrogen. J Am Chem Soc 118:3108–3116
Allen FH, Lommerse JPM, Hoy VJ, Howard JAK, Desiraju GR (1997) Halogen…O(nitro) supramolecular synthon in crystal engineering: a combined crystallographic database and ab initio molecular orbital study. Acta Cryst B53:1006–1016
Ikuta S (1990) Anisotropy of electron-density distribution around atoms in molecules: N, P, O and S atoms. J. Mol. Struct. (Theochem) 205:191–201
Hathwar VR, Row TNG (2010) Nature of Cl···Cl intermolecular interactions via experimental and theoretical charge density analysis: correlation of polar flattening effects with geometry. J Phys Chem A 114:13434–13441
Sedlak R, Kolář MH, Hobza P (2015) Polar flattening and the strength of halogen bonding. J Chem Theory Comput 11:4727–4732
Politzer P, Lane P, Concha MC, Ma Y, Murray JS (2007) An overview of halogen bonding. J Mol Model 13:305–311
Clark T, Hennemann M, Murray JS, Politzer P (2007) Halogen bonding: the σ-hole. J Mol Model 13:291–296
Solimannejad M, Gharabaghi M, Scheiner S (2011) SH…N and SH…P blue-shifting H-Bonds and N…P interactions in complexes pairing HSN with amines and phosphines. J. Chem. Phys 134:024312
Scheiner S (2011) A new noncovalent force: comparison of P…N interaction with hydrogen and halogen bonds. J. Chem. Phys 134:094315
Scheiner S (2011) Effects of multiple substitution upon the P…N noncovalent interaction. Chem Phys 387:79–84
Scheiner S (2011) Effects of substituents upon the P…N noncovalent interaction: the limits of its strength. J Phys Chem A 115:11202–11209
Scheiner S (2011) Can two trivalent N atoms engage in a direct N…N noncovalent interaction? Chem Phys Lett 514:32–35
Scheiner S (2013) The pnicogen bond: its relation to hydrogen, halogen, and other noncovalent bonds. Acc Chem Res 46:280–288
Scheiner S (2013) Detailed comparison of the pnicogen bond with chalcogen, halogen and hydrogen bonds. Int J Quantum Chem 113:1609–1620
Scheiner S (2011) On the properties of X…N noncovalent interactions for first-, second- and third-row X atoms. J Chem Phys 134:164313
Adhikari U, Scheiner S (2014) Effects of charge and substituent on the S···N Chalcogen bond. J Phys Chem A 118:3183–3192
Nziko VDPN, Scheiner S (2014) Chalcogen bonding between tetravalent SF4 and amines. J Phys Chem A 118:10849–10856
Azofra LM, Alkorta I, Scheiner S (2015) Chalcogen bonds in complexes of SOXY (X, Y = F, Cl) with nitrogen bases. J Phys Chem A 119:535–541
Nziko VDPN, Scheiner S (2015) Intramolecular S···O Chalcogen bond as stabilizing factor in geometry of substituted phenyl-SF3 molecules. J Org Chem 80:2356–2363
Nziko VDPN, Scheiner S (2015) S···π Chalcogen bonds between SF2 or SF4 and C–C multiple bonds. J Phys Chem A 119:5889–5897
Rosenfield RE, Parthasarathy R, Dunitz JD (1977) Directional preferences of nonbonded atomic contacts with divalent sulfur. 1. Electrophiles and nucleophiles. J Am Chem Soc 99:4860–4862
Burling FT, Goldstein BM (1992) Computational studies of nonbonded sulfur–oxygen and selenium–oxygen interactions in the thiazole and selenazole nucleosides. J Am Chem Soc 114:2313–2320
Iwaoka M, Tomoda S (1994) A model study on the effect of an amino group on the antioxidant activity of glutathione peroxidase. J Am Chem Soc 116:2557–2561
Del Bene JE, Alkorta I, Elguero J (2019) Probing C···S chalcogen bonds in complexes SC:SHX, for X = NO2, NC, F, Cl, CN, CCH, and NH2. Chem Phys Lett 721:86–90
Esrafili MD, Mousavian P, Mohammadian-Sabet F (2019) The influence of hydrogen- and lithium-bonding on the cooperativity of chalcogen bonds: a comparative ab initio study AU—Esrafili, Mehdi D. Mol Phys 117:726–733
Alkorta I, Legon A (2018) An ab initio investigation of the geometries and binding strengths of Tetrel-, Pnictogen-, and chalcogen-bonded complexes of CO2, N2O, and CS2 with simple lewis bases: some generalizations. Molecules 23:2250
Gleiter R, Haberhauer G, Werz DB, Rominger F, Bleiholder C (2018) From noncovalent chalcogen–chalcogen interactions to supramolecular aggregates: experiments and calculations. Chem Rev 118:2010–2041
Scheiner S (2015) Comparison of CH···O, SH···O, Chalcogen, and Tetrel bonds formed by neutral and cationic sulfur-containing compounds. J Phys Chem A 119:9189–9199
Nziko VDPN, Scheiner S (2016) Comparison of π-hole tetrel bonding with σ-hole halogen bonds in complexes of XCN (X = F, Cl, Br, I) and NH3. Phys Chem Chem Phys 18:3581–3590
Liu M, Li Q, Scheiner S (2017) Comparison of tetrel bonds in neutral and protonated complexes of pyridineTF3 and furanTF3 (T = C, Si, and Ge) with NH3. Phys Chem Chem Phys 19:5550–5559
Scheiner S (2017) Systematic elucidation of factors that influence the strength of Tetrel bonds. J Phys Chem A 121:5561–5568
Scheiner S (2018) Steric crowding in Tetrel bonds. J Phys Chem A 122:2550–2562
Zierkiewicz W, Michalczyk M, Scheiner S (2018) Implications of monomer deformation for tetrel and pnicogen bonds. Phys Chem Chem Phys 20:8832–8841
Zierkiewicz W, Michalczyk M, Wysokiński R, Scheiner S (2019) Dual geometry schemes in Tetrel bonds: complexes between TF4 (T = Si, Ge, Sn) and pyridine derivatives. Molecules 24:376
Trievel RC, Scheiner S (2018) Crystallographic and computational characterization of methyl tetrel bonding in S-adenosylmethionine-dependent methyltransferases. Molecules 23:2965–2981
Scheiner S (2018) Ability of IR and NMR spectral data to distinguish between a Tetrel bond and a hydrogen bond. J Phys Chem A 122:7852–7862
Scheiner S (2019) Dependence of NMR chemical shifts upon CH bond lengths of a methyl group involved in a Tetrel bond. Chem Phys Lett 714:61–64
Zhang J, Hu Q, Li Q, Scheiner S, Liu S (2019) Comparison of σ-hole and π-hole Tetrel bonds in complexes of borazine with TH3F and F2TO/H2TO (T = C, Si, Ge). Int J Quantum Chem 119:e25910
Zierkiewicz W, Michalczyk M, Scheiner S (2018) Comparison between Tetrel bonded complexes stabilized by σ and π hole interactions. Molecules 23:1416
Wei Y, Li Q, Scheiner S (2018) The π-tetrel bond and its influence on hydrogen bonding and proton transfer. ChemPhysChem 19:736–743
Grabowski SJ (2014) Boron and other triel lewis acid centers: from hypovalency to hypervalency. ChemPhysChem 15:2985–2993
Grabowski SJ (2015) π-hole bonds: boron and aluminum lewis acid centers. ChemPhysChem 16:1470–1479
Chi Z, Dong W, Li Q, Yang X, Scheiner S, Liu S (2019) Carbene triel bonds between TrR3 (Tr = B, Al) and N-heterocyclic carbenes. Int J Quantum Chem 119:e25867
Michalczyk M, Zierkiewicz W, Scheiner S (2018) Triel-bonded complexes between TrR3 (Tr = B, Al, Ga; R = H, F, Cl, Br, CH3) and pyrazine. ChemPhysChem 19:3122–3133
Mukherjee A, Sanz-Matias A, Velpula G, Waghray D, Ivasenko O, Bilbao N, Jeremy N, Harvey K, Mali S, De Feyter S (2019) Halogenated building blocks for 2D crystal engineering on solid surfaces: lessons from hydrogen bonding. Chem Sci 10:3881–3891
Xu C, Loh CCJ (2019) A multistage halogen bond catalyzed strain-release glycosylation unravels new hedgehog signaling inhibitors. J Am Chem Soc 141:5381–5391
Carreras L, Benet-Buchholz J, Franconetti A, Frontera A, van Leeuwen PWNM, Vidal-Ferran A (2019) Halogen bonding effects on the outcome of reactions at metal centres. Chem Commun 55:2380–2383
Hijazi H, Vacher A, Groni S, Lorcy D, Levillain E, Fave C, Schöllhorn B (2019) Electrochemically driven interfacial halogen bonding on self-assembled monolayers for anion detection. Chem Commun 55:1983–1986
Zhang X, Ren J, Tan SM, Tan D, Lee R, Tan C-H (2019) An enantioconvergent halogenophilic nucleophilic substitution (SN2X) reaction. Science 363:400–404
Wang H, Bisoyi HK, Urbas AM, Bunning TJ, Li Q (2019) The halogen bond: an emerging supramolecular tool in the design of functional mesomorphic materials. Chem Eur J 25:1369–1378
Liu C-Z, Koppireddi S, Wang H, Zhang D-W, Li Z-T (2019) Halogen bonding directed supramolecular quadruple and double helices from hydrogen-bonded arylamide foldamers. Angew Chem Int Ed 58:226–230
Serpell CJ, Kilah NL, Costa PJ, Félix V, Beer PD (2010) Halogen bond anion templated assembly of an imidazolium pseudorotaxane. Angew Chem Int Ed 49:5322–5326
Caballero A, White NG, Beer PD (2011) A bidentate halogen-bonding bromoimidazoliophane receptor for bromide ion recognition in aqueous media. Angew Chem Int Ed Engl 50:1845–1848
Walter SM, Kniep F, Rout L, Schmidtchen FP, Herdtweck E, Huber SM (2012) Isothermal calorimetric titrations on charge-assisted halogen bonds: role of entropy, counterions, solvent, and temperature. J Am Chem Soc 134:8507–8512
Gilday LC, White NG, Beer PD (2013) Halogen- and hydrogen-bonding triazole-functionalised porphyrin-based receptors for anion recognition. Dalton Trans 42:15766–15773
Borissov A, Marques I, Lim JYC, Félix V, Smith MD, Beer PD (2019) Anion recognition in water by charge-neutral halogen and chalcogen bonding foldamer receptors. J Am Chem Soc 141:4119–4129
Klein HA, Beer PD (2019) Iodide discrimination by tetra-iodotriazole halogen bonding interlocked hosts. Chem Eur J 25:3125–3130
Hein R, Borissov A, Smith MD, Beer PD, Davis JJ (2019) A halogen-bonding foldamer molecular film for selective reagentless anion sensing in water. Chem Commun 55:4849–4852
Chakraborty S, Maji S, Ghosh R, Jana R, Datta A, Ghosh P (2019) Aryl-platform-based tetrapodal 2-iodo-imidazolium as an excellent halogen bond receptor in aqueous medium. Chem Commun 55:1506–1509
Nepal B, Scheiner S (2015) competitive halide binding by halogen versus hydrogen bonding: bis-triazole pyridinium. Chem Eur J 21:13330–13335
Nepal B, Scheiner S (2015) Substituent effects on the binding of halides by neutral and dicationic bis(triazolium) receptors. J Phys Chem A 119:13064–13073
Nepal B, Scheiner S (2016) Building a better halide receptor: optimum choice of spacer, binding unit, and halosubstitution. ChemPhysChem 17:836–844
Scheiner S (2017) Assembly of effective halide receptors from components. Comparing hydrogen, halogen, and Tetrel bonds. J Phys Chem 121:3606–3615
Scheiner S (2017) Comparison of halide receptors based on H, halogen, chalcogen, pnicogen, and tetrel bonds. Faraday Disc 203:213–226
Scheiner S (2018) Tetrel bonding as a vehicle for strong and selective anion binding. Molecules 23:1147–1155
Scheiner S (2019) Differential binding of tetrel-bonding bipodal receptors to monatomic and polyatomic anions. Molecules 24:227
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Scheiner, S. The Hydrogen Bond: A Hundred Years and Counting. J Indian Inst Sci 100, 61–76 (2020). https://doi.org/10.1007/s41745-019-00142-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41745-019-00142-8