Abstract
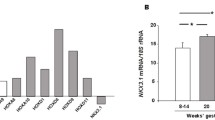
Fetal growth restriction (FGR) affects up to 5 % of pregnancies worldwide, and trophoblast function plays a significant role on the outcome. An epidemiological study has linked vitamin D deficiency to adverse perinatal outcomes, which include decreased birth weight. The placenta as an important source of vitamin D regulates its metabolism through the vitamin D receptor (VDR), but the mechanism by which VDR regulates trophoblast function is poorly understood. Our study aimed at determining placental VDR expression in FGR and gestation-matched control (GMC) pregnancies and identifying the actions of VDR in trophoblast differentiation and apoptosis. Placentae were collected from a well-defined cohort of idiopathic FGR and GMC pregnancies. VDR mRNA and protein expressions were determined by PCR, immunohistochemistry and immunoblotting, while functional consequences of VDR inactivation in vitro were determined on BeWo cells by determining changes in differentiation, attachment and apoptosis. Significant decreases in VDR mRNA expression (p = 0.0005) and protein expression (p = 0.0003) were observed in the FGR samples, while VDR inactivation, which showed markers for differentiation, cell attachment and apoptosis, was significantly increased. Thus, decreased placental VDR may contribute to uncontrolled premature differentiation and apoptosis of trophoblasts that are characteristics of idiopathic FGR pregnancies.
Key message
-
Fetal growth restriction (FGR) affects up to 5 % of all pregnancies worldwide.
-
FGR is the second highest cause of perinatal mortality and morbidity.
-
The placenta plays a pivotal role in vitamin D metabolism during pregnancy.
-
Vitamin D deficiency is associated with adverse pregnancy outcomes.
-
Placental vitamin D receptor expression is decreased in FGR.






Similar content being viewed by others
References
Bikle DD (2011) Vitamin D regulation of immune function. Vitam Horm 86:1–21
Lagishetty V, Liu NQ, Hewison M (2011) Vitamin D metabolism and innate immunity. Mol Cell Endocrinol 347:97–105
Wei SQ, Qi HP, Luo ZC, Fraser WD (2013) Maternal vitamin D status and adverse pregnancy outcomes: a systematic review and meta-analysis. J J Matern Fetal Neonatal Med Off J Eur Assoc Perinatal Med Fed Asia Ocean Perinatal Soc Int Soc Perinatal Obstet 26:889–899
Robinson CJ, Wagner CL, Hollis BW, Baatz JE, Johnson DD (2011) Maternal vitamin D and fetal growth in early-onset severe preeclampsia. Am J Obstet Gynecol 204(556):e551–554
Kumar R, Cohen WR, Silva P, Epstein FH (1979) Elevated 1,25-dihydroxyvitamin D plasma levels in normal human pregnancy and lactation. J Clin Invest 63:342–344
Liu NQ, Hewison M (2012) Vitamin D, the placenta and pregnancy. Arch Biochem Biophys 523:37–47
Robinson CJ, Alanis MC, Wagner CL, Hollis BW, Johnson DD (2010) Plasma 25-hydroxyvitamin D levels in early-onset severe preeclampsia. Am J Obstet Gynecol 203(366):e361–366
Thorne-Lyman A, Fawzi WW (2012) Vitamin D during pregnancy and maternal, neonatal and infant health outcomes: a systematic review and meta-analysis. Paediatr Perinat Epidemiol 26(Suppl 1):75–90
De-Regil LM, Palacios C, Ansary A, Kulier R, Pena-Rosas JP (2012) Vitamin D supplementation for women during pregnancy. Cochrane Database Syst Rev 2:CD008873
Goyal R, Zhang L, Blood AB, Baylink DJ, Longo LD, Oshiro B, Mata-Greenwood E (2014) Characterization of an animal model of pregnancy-induced vitamin D deficiency due to metabolic gene dysregulation. Am J Physiol Endocrinol Metab 306:E256–266
Awad AB, Alappat L, Valerio M (2012) Vitamin d and metabolic syndrome risk factors: evidence and mechanisms. Crit Rev Food Sci Nutr 52:103–112
Fleet JC, DeSmet M, Johnson R, Li Y (2012) Vitamin D and cancer: a review of molecular mechanisms. Biochem J 441:61–76
Motiwala SR, Wang TJ (2011) Vitamin D and cardiovascular disease. Curr Opin Nephrol Hypertens 20:345–353
Ponsonby AL, Lucas RM, Lewis S, Halliday J (2010) Vitamin D status during pregnancy and aspects of offspring health. Nutrients 2:389–407
Pludowski P, Holick MF, Pilz S, Wagner CL, Hollis BW, Grant WB, Shoenfeld Y, Lerchbaum E, Llewellyn DJ, Kienreich K et al (2013) Vitamin D effects on musculoskeletal health, immunity, autoimmunity, cardiovascular disease, cancer, fertility, pregnancy, dementia and mortality-a review of recent evidence. Autoimmun Rev 12:976–989
McIntire DD, Bloom SL, Casey BM, Leveno KJ (1999) Birth weight in relation to morbidity and mortality among newborn infants. N Engl J Med 340:1234–1238
Scifres CM, Nelson DM (2009) Intrauterine growth restriction, human placental development and trophoblast cell death. J Physiol 587:3453–3458
Mongelli M, Gardosi J (2000) Fetal growth. Curr Opin Obstet Gynecol 12:111–115
Godfrey KM, Barker DJ (2001) Fetal programming and adult health. Public Health Nutr 4:611–624
Barker DJ, Eriksson JG, Forsen T, Osmond C (2002) Fetal origins of adult disease: strength of effects and biological basis. Int J Epidemiol 31:1235–1239
Steffensen FH, Sorensen HT, Gillman MW, Rothman KJ, Sabroe S, Fischer P, Olsen J (2000) Low birth weight and preterm delivery as risk factors for asthma and atopic dermatitis in young adult males. Epidemiology 11:185–188
Rosso IM, Cannon TD, Huttunen T, Huttunen MO, Lonnqvist J, Gasperoni TL (2000) Obstetric risk factors for early-onset schizophrenia in a Finnish birth cohort. Am J Psychiatry 157:801–807
Gale CR, Martyn CN (2004) Birth weight and later risk of depression in a national birth cohort. Br J Psychiatry 184:28–33
Frisk V, Amsel R, Whyte HE (2002) The importance of head growth patterns in predicting the cognitive abilities and literacy skills of small-for-gestational-age children. Dev Neuropsychol 22:565–593
Sankaran S, Kyle PM (2009) Aetiology and pathogenesis of IUGR. Best Pract Res Clin Obstet Gynaecol 23:765–777
Handwerger S (2010) New insights into the regulation of human cytotrophoblast cell differentiation. Mol Cell Endocrinol 323:94–104
Ma R, Gu Y, Zhao S, Sun J, Groome LJ, Wang Y (2012) Expressions of vitamin D metabolic components VDBP, CYP2R1, CYP27B1, CYP24A1, and VDR in placentas from normal and preeclamptic pregnancies. Am J Physiol Endocrinol Metab 303:E928–935
Murthi P, Said JM, Doherty VL, Donath S, Nowell CJ, Brennecke SP, Kalionis B (2006) Homeobox gene DLX4 expression is increased in idiopathic human fetal growth restriction. Mol Hum Reprod 12:763–769
Murthi P, Doherty VL, Said JM, Donath S, Brennecke SP, Kalionis B (2006) Homeobox gene ESX1L expression is decreased in human pre-term idiopathic fetal growth restriction. Mol Hum Reprod 12:335–340
Murthi P, Doherty V, Said J, Donath S, Brennecke SP, Kalionis B (2006) Homeobox gene HLX1 expression is decreased in idiopathic human fetal growth restriction. Am J Pathol 168:511–518
Rajaraman G, Murthi P, Leo B, Brennecke SP, Kalionis B (2007) Homeobox gene HLX1 is a regulator of colony stimulating factor-1 dependent trophoblast cell proliferation. Placenta 28:991–998
Rajaraman G, Murthi P, Pathirage N, Brennecke SP, Kalionis B (2010) Downstream targets of homeobox gene HLX show altered expression in human idiopathic fetal growth restriction. Am J Pathol 176:278–287
Pathirage NA, Cocquebert M, Sadovsky Y, Abumaree M, Manuelpillai U, Borg A, Keogh RJ, Brennecke SP, Evain-Brion D, Fournier T et al (2013) Homeobox gene transforming growth factor beta-induced factor-1 (TGIF-1) is a regulator of villous trophoblast differentiation and its expression is increased in human idiopathic fetal growth restriction. Mol Hum Reprod 19:665–675
Guaran RL, Wein P, Sheedy M, Walstab J, Beischer NA (1994) Update of growth percentiles for infants born in an Australian population. Aust N Z J Obstet Gynaecol 34:39–50
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Keogh RJ (2010) New technology for investigating trophoblast function. Placenta 31:347–350
Chen CP, Bajoria R, Aplin JD (2002) Decreased vascularization and cell proliferation in placentas of intrauterine growth-restricted fetuses with abnormal umbilical artery flow velocity waveforms. Am J Obstet Gynecol 187:764–769
Kingdom J, Huppertz B, Seaward G, Kaufmann P (2000) Development of the placental villous tree and its consequences for fetal growth. Eur J Obstet Gynecol Reprod Biol 92:35–43
Avila E, Diaz L, Halhali A, Larrea F (2004) Regulation of 25-hydroxyvitamin D3 1alpha-hydroxylase, 1,25-dihydroxyvitamin D3 24-hydroxylase and vitamin D receptor gene expression by 8-bromo cyclic AMP in cultured human syncytiotrophoblast cells. J Steroid Biochem Mol Biol 89–90:115–119
Barrera D, Avila E, Hernandez G, Mendez I, Gonzalez L, Halhali A, Larrea F, Morales A, Diaz L (2008) Calcitriol affects hCG gene transcription in cultured human syncytiotrophoblasts. Reprod Biol Endocrinol RB&E 6:3
Stephanou A, Ross R, Handwerger S (1994) Regulation of human placental lactogen expression by 1,25-dihydroxyvitamin D3. Endocrinology 135:2651–2656
Tuan RS, Moore CJ, Brittingham JW, Kirwin JJ, Akins RE, Wong M (1991) In vitro study of placental trophoblast calcium uptake using JEG-3 human choriocarcinoma cells. J Cell Sci 98(Pt 3):333–342
Kovacs CS, Kronenberg HM (1997) Maternal-fetal calcium and bone metabolism during pregnancy, puerperium, and lactation. Endocr Rev 18:832–872
Evans KN, Bulmer JN, Kilby MD, Hewison M (2004) Vitamin D and placental-decidual function. J Soc Gynecol Investig 11:263–271
Kovacs CS (2008) Vitamin D in pregnancy and lactation: maternal, fetal, and neonatal outcomes from human and animal studies. Am J Clin Nutr 88:520S–528S
Zehnder D, Evans KN, Kilby MD, Bulmer JN, Innes BA, Stewart PM, Hewison M (2002) The ontogeny of 25-hydroxyvitamin D(3) 1alpha-hydroxylase expression in human placenta and decidua. Am J Pathol 161:105–114
Samuel S, Sitrin MD (2008) Vitamin D’s role in cell proliferation and differentiation. Nutr Rev 66:S116–124
Shahbazi M, Jeddi-Tehrani M, Zareie M, Salek-Moghaddam A, Akhondi MM, Bahmanpoor M, Sadeghi MR, Zarnani AH (2011) Expression profiling of vitamin D receptor in placenta, decidua and ovary of pregnant mice. Placenta 32:657–664
Sandovici I, Hoelle K, Angiolini E, Constancia M (2012) Placental adaptations to the maternal-fetal environment: implications for fetal growth and developmental programming. Reprod Biomed Online 25:68–89
Novakovic B, Sibson M, Ng HK, Manuelpillai U, Rakyan V, Down T, Beck S, Fournier T, Evain-Brion D, Dimitriadis E et al (2009) Placenta-specific methylation of the vitamin D 24-hydroxylase gene: implications for feedback autoregulation of active vitamin D levels at the fetomaternal interface. J Biol Chem 284:14838–14848
Saffery R, Ellis J, Morley R (2009) A convergent model for placental dysfunction encompassing combined sub-optimal one-carbon donor and vitamin D bioavailability. Med Hypotheses 73:1023–1028
Jurutka PW, Whitfield GK, Hsieh JC, Thompson PD, Haussler CA, Haussler MR (2001) Molecular nature of the vitamin D receptor and its role in regulation of gene expression. Rev Endocr Metab Disord 2:203–216
Whitfield GK, Remus LS, Jurutka PW, Zitzer H, Oza AK, Dang HT, Haussler CA, Galligan MA, Thatcher ML, Encinas Dominguez C et al (2001) Functionally relevant polymorphisms in the human nuclear vitamin D receptor gene. Mol Cell Endocrinol 177:145–159
Ingles SA, Ross RK, Yu MC, Irvine RA, La Pera G, Haile RW, Coetzee GA (1997) Association of prostate cancer risk with genetic polymorphisms in vitamin D receptor and androgen receptor. J Natl Cancer Inst 89:166–170
Niimi T, Tomita H, Sato S, Kawaguchi H, Akita K, Maeda H, Sugiura Y, Ueda R (1999) Vitamin D receptor gene polymorphism in patients with sarcoidosis. Am J Respir Crit Care Med 160:1107–1109
Morley R, Carlin JB, Pasco JA, Wark JD, Ponsonby AL (2009) Maternal 25-hydroxyvitamin D concentration and offspring birth size: effect modification by infant VDR genotype. Eur J Clin Nutr 63:802–804
Kaufmann P, Black S, Huppertz B (2003) Endovascular trophoblast invasion: implications for the pathogenesis of intrauterine growth retardation and preeclampsia. Biol Reprod 69:1–7
Crocker IP, Cooper S, Ong SC, Baker PN (2003) Differences in apoptotic susceptibility of cytotrophoblasts and syncytiotrophoblasts in normal pregnancy to those complicated with preeclampsia and intrauterine growth restriction. Am J Pathol 162:637–643
Newhouse SM, Davidge ST, Winkler-Lowen B, Demianczuk N, Guilbert LJ (2007) In vitro differentiation of villous trophoblasts from pregnancies complicated by intrauterine growth restriction with and without pre-eclampsia. Placenta 28:999–1003
Huppertz B (2003) Extravillous trophoblast: proliferation and invasion during pregnancy. Pathologica 95:231–232
Mi S, Lee X, Li X, Veldman GM, Finnerty H, Racie L, LaVallie E, Tang XY, Edouard P, Howes S et al (2000) Syncytin is a captive retroviral envelope protein involved in human placental morphogenesis. Nature 403:785–789
Vargas A, Moreau J, Le Bellego F, Lafond J, Barbeau B (2008) Induction of trophoblast cell fusion by a protein tyrosine phosphatase inhibitor. Placenta 29:170–174
Frendo JL, Cronier L, Bertin G, Guibourdenche J, Vidaud M, Evain-Brion D, Malassine A (2003) Involvement of connexin 43 in human trophoblast cell fusion and differentiation. J Cell Sci 116:3413–3421
Evseenko DA, Murthi P, Paxton JW, Reid G, Emerald BS, Mohankumar KM, Lobie PE, Brennecke SP, Kalionis B, Keelan JA (2007) The ABC transporter BCRP/ABCG2 is a placental survival factor, and its expression is reduced in idiopathic human fetal growth restriction. FASEB J 21:3592–3605
Yung HW, Hemberger M, Watson ED, Senner CE, Jones CP, Kaufman RJ, Charnock-Jones DS, Burton GJ (2012) Endoplasmic reticulum stress disrupts placental morphogenesis: implications for human intrauterine growth restriction. J Pathol 228:554–564
Lala PK, Lee BP, Xu G, Chakraborty C (2002) Human placental trophoblast as an in vitro model for tumor progression. Can J Physiol Pharmacol 80:142–149
Huppertz B, Kadyrov M, Kingdom JC (2006) Apoptosis and its role in the trophoblast. Am J Obstet Gynecol 195:29–39
Smith SC, Baker PN (1999) Placental apoptosis is increased in post-term pregnancies. Br J Obstet Gynaecol 106:861–862
Author contributions
T.P.H.N. contributed by performing the laboratory work, acquisition of data, analysis and interpretation, drafting and revision of the manuscript.
H.E.J.Y. assisted with the data analysis and interpretation, and revision of the manuscript.
T.C. contributed by assisting with the optimisation of cell culture experiments, data analysis and revision of the manuscript.
A.J.B. assisted with the real-time PCR analyses, data acquisition and revision of the manuscript.
S.P.B. contributed to the clinical characterisation of patient samples and critical revision of the manuscript for its intellectual content.
P.M. contributed substantially to the study concept and experimental design, data acquisition, analysis and interpretation and critical revision of the manuscript for its intellectual content.
Conflict of interest
The authors have nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nguyen, T.P.H., Yong, H.E.J., Chollangi, T. et al. Placental vitamin D receptor expression is decreased in human idiopathic fetal growth restriction. J Mol Med 93, 795–805 (2015). https://doi.org/10.1007/s00109-015-1267-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-015-1267-1