Abstract
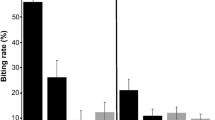
Host choice by mosquitoes affects the transmission dynamics of vector-borne infectious diseases. Although asymmetries in mosquito attraction to vertebrate species have been reported, the relative importance of host characteristics in mosquito blood-feeding behavior is still poorly studied. Here, we investigate the relationship between avian phenotypic traits—in particular, morphometry, plumage coloration, and nesting and roosting behavior—and the blood-feeding patterns in two common Culex mosquito species on a North American avian community. Forage ratios of the mosquito species were unrelated to the phylogenetic relationships among bird species. Culex pipiens fed preferably on birds with lighter-colored plumage and longer tarsi; furthermore, solitary roosting avian species were both bitten by Cx. pipiens and Cx. restuans more often than expected. These associations may be explained by greater mosquito attraction towards larger birds with a greater color contrast against the background. Although communally roosting birds may release more cues and attract more mosquitoes, individuals may in fact receive fewer bites due to the encounter-dilution effect. Mosquito feeding behavior is a highly complex phenomenon, and our results may improve understanding of the non-random interaction between birds and mosquitoes in natural communities.

Similar content being viewed by others
References
Allan SA (1994) Physics of mosquito vision—an overview. J Am Mosq Control Assoc 10:266–271
Allan SA, Day JF, Edman JD (1987) Visual ecology of biting flies. Annu Rev Entomol 32:297–314. https://doi.org/10.1146/annurev.en.32.010187.001501
Bartoń K (2013) MuMIn: multi-model inference R package version 1
Becker N, Petrić D, Boase C, Lane J, Zgomba M, Dahl C, Kaiser A (2010) Mosquitoes and their control vol 2, 2nd edn. Springer. https://doi.org/10.1007/978-3-540-92874-4
Bidlingmayer W (1971) Mosquito flight paths in relation to the environment. 1. Illumination levels, orientation, and resting areas. Ann Entomol Soc Am 64:1121–1131
Blackmore JS, Dow RP (1958) Differential feeding of Culex tarsalis on nestling and adult birds. Mosq News 18:15–17
Brett G (1938) On the relative attractiveness to Aedes aegypti of certain coloured cloths. Trans R Soc Trop Med Hyg 32:113–124. https://doi.org/10.1016/S0035-9203(38)90101-4
van Breugel F, Riffell J, Fairhall A, Dickinson MH (2015) Mosquitoes use vision to associate odor plumes with thermal targets. Curr Biol 25:2123–2129. https://doi.org/10.1016/j.cub.2015.06.046
Brown A (1954) Studies on the responses of the female aëdes mosquito. Part VI—the attractiveness of coloured cloths to Canadian species. Bull Entomol Res 45:67–78. https://doi.org/10.1017/S0007485300026808
Browne SM, Bennett GF (1981) Response of mosquitoes (Diptera: Culicidae) to visual stimuli. J Med Entomol 18:505–521. https://doi.org/10.1093/jmedent/18.6.505
Burkett-Cadena ND, McClure CJ, Ligon RA, Graham SP, Guyer C, Hill GE, Ditchkoff SS, Eubanks MD, Hassan HK, Unnasch TR (2011) Host reproductive phenology drives seasonal patterns of host use in mosquitoes. PLoS One 6:e17681
Burnham KP, Anderson DR (2003) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer Science & Business Media, New York. https://doi.org/10.1007/b97636
Burnham KP, Anderson DR (2004) Multimodel inference understanding AIC and BIC in model selection. Socio Meth Res 33:261–304. https://doi.org/10.1177/0049124104268644
Cardé RT (2015) Multi-cue integration: how female mosquitoes locate a human host. Curr Biol 25:R793–R795
Clements K (2007) The encyclopedia of birds: 6 volume set. Facts On File, New York
Conway DJ, McBride JS (1991) Genetic evidence for the importance of interrupted feeding by mosquitoes in the transmission of malaria. Trans R Soc Trop Med Hyg 85:454–456
Core Development Team R (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Cresswell W (1994) Flocking is an effective anti-predation strategy in redshanks, Tringa totanus. Anim Behav 47:433–442
Davies C (1990) Interrupted feeding of blood-sucking insects: causes and effects. Parasitol Today 6:19–22
del Hoyo J, Elliott A, Christie DA (2011) The handbook of the birds of the world-volume 16 tanagers to new world blackbirds. Lynx Edicions, Barcelona
Diuk-Wasser MA, Molaei G, Simpson JE, Folsom-O’Keefe CM, Armstrong PM, Andreadis TG (2010) Avian communal roosts as amplification foci for West Nile virus in urban areas in northeastern United States. Am J Trop Med Hyg 82:337–343
Edman JD, Scott TW (1987) Host defensive behaviour and the feeding success of mosquitoes. Int J Trop Insect Sci 8:617–622. https://doi.org/10.1017/S1742758400022694
Estep LK, McClure CJ, Burkett-Cadena ND, Hassan HK, Hicks TL, Unnasch TR, Hill GE (2011) A multi-year study of mosquito feeding patterns on avian hosts in a southeastern focus of eastern equine encephalitis virus. Am J Trop Med Hyg 84:718–726
Estep LK, McClure CJ, Burkett-Cadena ND, Hassan HK, Unnasch TR, Hill GE (2012) Developing models for the forage ratios of Culiseta melanura and Culex erraticus using species characteristics for avian hosts. J Med Entomol 49:378–387
Farajollahi A, Fonseca DM, Kramer LD, Kilpatrick AM (2011) “Bird biting” mosquitoes and human disease: a review of the role of Culex pipiens complex mosquitoes in epidemiology. Infect Genet Evol 11:1577–1585. https://doi.org/10.1016/j.meegid.2011.08.013
Ferraguti M, Martínez-de la Puente J, Muñoz J, Roiz D, Ruiz S, Soriguer R, Figuerola J (2013) Avian Plasmodium in Culex and Ochlerotatus mosquitoes from southern Spain: effects of season and host-feeding source on parasite dynamics. PLoS One 8:e66237. https://doi.org/10.1371/journal.pone.0066237
Figuerola J, Jiménez-Clavero MA, López G, Rubio C, Soriguer R, Gómez-Tejedor C, Tenorio A (2008) Size matters: West Nile Virus neutralizing antibodies in resident and migratory birds in Spain. Vet Microbiol 132:39–46. https://doi.org/10.1016/j.vetmic.2008.04.023
Fox J, Weisberg S, Bates D (2010) Car: companion to applied regression. R package version 2.1–2
Gilbert I, Gouck H (1957) Influence of surface color on mosquito landing rates. J Econ Entomol 50:678–680. https://doi.org/10.1093/jee/50.5.678
Green AJ (2001) Mass/length residuals: measures of body condition or generators of spurious results? Ecology 82:1473–1483
Grueber C, Nakagawa S, Laws R, Jamieson I (2011) Multimodel inference in ecology and evolution: challenges and solutions. J Evol Biol 24:699–711
Hamer GL, Kitron UD, Goldberg TL, Brawn JD, Loss SR, Ruiz MO, Hayes DB, Walker ED (2009) Host selection by Culex pipiens mosquitoes and West Nile virus amplification. Am J Trop Med Hyg 80:268–278
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387. https://doi.org/10.1126/science.7123238
Hassan HK, Cupp EW, Hill GE, Katholi CR, Klingler K, Unnasch TR (2003) Avian host preference by vectors of eastern equine encephalomyelitis virus. Am J Trop Med Hyg 69:641–647
Hawkes F, Gibson G (2016) Seeing is believing: the nocturnal malarial mosquito Anopheles coluzzii responds to visual host-cues when odour indicates a host is nearby. Parasit Vectors 9:1
Janousek WM, Marra PP, Kilpatrick AM (2014) Avian roosting behavior influences vector–host interactions for West Nile virus hosts. Parasit Vectors 7:1–11
Jaramillo A, Burke P (1999) New world blackbirds: the Icterids. Princeton University Press, Princeton
Kamilar JM, Cooper N (2013) Phylogenetic signal in primate behaviour, ecology and life history. Philos Trans R Soc B 368:20120341
Kilpatrick AM, Daszak P, Jones MJ, Marra PP, Kramer LD (2006a) Host heterogeneity dominates West Nile virus transmission. Proc R Soc B 273:2327–2333. https://doi.org/10.1098/rspb.2006.3575
Kilpatrick AM, Kramer LD, Jones MJ, Marra PP, Daszak P (2006b) West Nile virus epidemics in North America are driven by shifts in mosquito feeding behavior. PLoS Biol 4:e82
Kleiber M (1947) Body size and metabolic rate. Physiol Rev 27:511–541
Krebs BL, Anderson TK, Goldberg TL, Hamer GL, Kitron UD, Newman CM, Ruiz MO, Walker ED, Brawn JD (2014) Host group formation decreases exposure to vector-borne disease: a field experiment in a ‘hotspot’of West Nile virus transmission. Proc R Soc B 281:20141586
Lehane MJ (2005) The biology of blood-sucking in insects, 2nd edn. Cambridge University Press, New York
Lislevand T, Figuerola J, Szekely T (2007) Avian body sizes in relation to fecundity, mating system, display behavior, and resource sharing. Ecology 88:1605–1605. https://doi.org/10.1890/06-2054
Llopis IV, Tomassone L, Grego E, Serrano E, Mosca A, Vaschetti G, Andrade D, Rossi L (2016) Evaluating the feeding preferences of West Nile virus mosquito vectors using bird-baited traps. Parasit Vectors 9:479
Long C, Flint J, Lepper P (2011) Insect attraction to wind turbines: does colour play a role? Eur J Wildl Res 57:323–331
Malmqvist B, Strasevicius D, Hellgren O, Adler PH, Bensch S (2004) Vertebrate host specificity of wild–caught blackflies revealed by mitochondrial DNA in blood. Proc R Soc Lond B (Suppl) 271:S152–S155. https://doi.org/10.1098/rsbl.2003.0120
Marquardt WH (2004) Biology of disease vectors. Academic Press
Martínez-Abraín A, Esparza B, Oro D (2004) Lack of blood parasites in bird species: does absence of blood parasite vectors explain it all. Ardeola 51:225–232
Martínez-de la Puente J, Merino S, Tomás G, Moreno J, Morales J, Lobato E, Taravera S, Sarto I, Monteys V (2009) Factors affecting Culicoides species composition and abundance in avian nests. Parasitology 136:1033–1041. https://doi.org/10.1017/S0031182009006374
Martínez-de la Puente J, Merino S, Lobato E, Rivero-de Aguilar J, del Cerro S, Ruiz-de-Castañeda R, Moreno J (2010) Nest-climatic factors affect the abundance of biting flies and their effects on nestling condition. Acta Oecol 36:543–547. https://doi.org/10.1016/j.actao.2010.07.008
Mendenhall I, Tello S, Neira L, Castillo L, Ocampo C, Wesson D (2012) Host preference of the arbovirus vector Culex erraticus (Diptera: Culicidae) at Sonso Lake, Cauca Valley department, Colombia. J Med Entomol 49:1092–1102
Mooring MS, Benjamin JE, Harte CR, Herzog NB (2000) Testing the interspecific body size principle in ungulates: the smaller they come, the harder they groom. Anim Behav 60:35–45. https://doi.org/10.1006/anbe.2000.1461
Muñoz J, Eritja R, Alcaide M, Montalvo T, Soriguer RC, Figuerola J (2011) Host-feeding patterns of native Culex pipiens and invasive Aedes albopictus mosquitoes (Diptera: Culicidae) in urban zones from Barcelona, Spain. J Med Entomol 48:956–960. https://doi.org/10.1603/ME11016
Muñoz J, Ruiz S, Soriguer R, Alcaide M, Viana DS, Roiz D, Vázquez A, Figuerola J (2012) Feeding patterns of potential West Nile virus vectors in south-west Spain. PLoS One 7:1–9. https://doi.org/10.1371/journal.pone.0039549
O’brien RM (2007) A caution regarding rules of thumb for variance inflation factors. Qual Quant 41:673–690
Orme D (2013) The caper package: comparative analysis of phylogenetics and evolution in R version 0.5.2
Pagel M (1999) Inferring the historical patterns of biological evolution. Nature 401:877–884. https://doi.org/10.1038/44766
Paradis E, Claude J, Strimmer K (2004) APE: analyses of phylogenetics and evolution in R language version 3.5. Bioinformatics 20:289–290
Piersma T (1997) Do global patterns of habitat use and migration strategies co-evolve with relative investments in immunocompetence due to spatial variation in parasite pressure? Oikos:623–631
Rizzoli A, Bolzoni L, Chadwick EA, Capelli G, Montarsi F, Grisenti M, Martínez-de la Puente J, Muñoz J, Figuerola J, Soriguer R (2015) Understanding West Nile virus ecology in Europe: Culex pipiens host feeding preference in a hotspot of virus emergence. Parasit Vectors 8:213. https://doi.org/10.1186/s13071-015-0831-4
Rodewald P (2015) The birds of North America. Cornell Laboratory of Ornithology, Ithaca. Online: http://bna.birds.cornell.edu/BNA/. Accessed August 2015
Rubolini D, Liker A, Garamszegi LZ, Møller AP, Saino N (2015) Using the BirdTree.org website to obtain robust phylogenies for avian comparative studies: a primer. Curr Zool 61:959–965. https://doi.org/10.1093/czoolo/61.6.959
Sawabe K, Isawa H, Hoshino K, Sasaki T, Roychoudhury S, Higa Y, Kasai S, Tsuda Y, Nishiumi I, Hisai N (2010) Host-feeding habits of Culex pipiens and Aedes albopictus (Diptera: Culicidae) collected at the urban and suburban residential areas of Japan. J Med Entomol 47:442–450. https://doi.org/10.1603/ME09256
Scott TW, Chow E, Strickman D, Kittayapong P, Wirtz RA, Lorenz LH, Edman JD (1993) Blood-feeding patterns of Aedes aegypti (Diptera: Culicidae) collected in a rural Thai village. J Med Entomol 30:922–927
Senar J, Pascual J (1997) Keel and tarsus length may provide a good predictor of avian body size. Ardea Wagening 85:269–274
Simpson JE, Hurtado PJ, Medlock J, Molaei G, Andreadis TG, Galvani AP, Diuk-Wasser MA (2012) Vector host-feeding preferences drive transmission of multi-host pathogens: West Nile virus as a model system. Proc R Soc B 279:925–933. https://doi.org/10.1098/rspb.2011.1282
Suom C, Ginsberg HS, Bernick A, Klein C, Buckley P, Salvatore C, LeBrun RA (2010) Host-seeking activity and avian host preferences of mosquitoes associated with West Nile virus transmission in the northeastern USA. J Vector Ecol 35:69–74
Takken W, Verhulst NO (2013) Host preferences of blood-feeding mosquitoes. Annu Rev Entomol 58:433–453. https://doi.org/10.1146/annurev-ento-120811-153618
Tempelis C, Reeves W, Bellamy R, Lofy M (1965) A three-year study of the feeding habits of Culex tarsalis in Kern County, California. Am J Trop Med Hyg 14:170–177
Thiemann TC, Reisen WK (2012) Evaluating sampling method bias in Culex tarsalis and Culex quinquefasciatus (Diptera: Culicidae) bloodmeal identification studies. J Med Entomol 49:143–149
Thiemann TC, Wheeler SS, Barker CM, Reisen WK (2011) Mosquito host selection varies seasonally with host availability and mosquito density. PLoS Negl Trop Dis 5:e1452
Vuilleumier F (2011) Birds of North America: western region. Dorling Kindersley Publishing, New York
Walker ED, Edman JD (1985) Feeding-site selection and blood-feeding behavior of Aedes triseriatus (Diptera: Culicidae) on rodent (Sciuridae) hosts. J Med Entomol 22:287–294. https://doi.org/10.1093/jmedent/22.3.287
Yezerinac SM, Weatherhead PJ (1995) Plumage coloration, differential attraction of vectors and hematozoa infections in birds. J Anim Ecol 64:528–537. https://doi.org/10.2307/5655
Acknowledgements
We are grateful to GL Hamer, UD Kitron, TL Goldberg, JD Brawn, SR Loss, MO Ruiz, DB Hayes, and ED Walker for publishing the FR data used in their paper. We thank the four anonymous reviewers whose comments helped improve earlier drafts of the manuscript.
Funding information
This study was funded by the projects CGL2012-30759 and CGL2015-65055-P from the Spanish Ministry of Economy and Competitiveness and the European Regional Development Fund (FEDER). JY was supported by the State Scholarship Fund from the China Scholarship Council, LG by a contract from the Excellence Projects of the Junta de Andalucía (RNM-7800), and JMP by the Juan de la Cierva program.
Author contributions
J.Y. conducted the literature review, carried out the analyses, and drafted a first version of the manuscript; L.G. coordinated the analyses and helped collect data and draft the manuscript; J.M.P. helped collect data and draft the manuscript; R.S. coordinated and designed the study; J.F. coordinated and designed the study, and helped in manuscript drafting. All authors have given their approval for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Communicated by: Pamela C. Rasmussen
Electronic supplementary material
Table S1
Phenotypic attributes for the 49 avian species included in this study. The forage ratio is given for mosquitoes of the species Cx. pipiens and Cx. restuans. Abbreviations used: Mass: body mass (g); Tarsus: tarsus length (mm); Bill: bill length (mm); Phac: the percentage of highly attractive colors; Pmac: the percentage of moderately attractive colors; Psac: the percentage of slightly attractive colors; Roosting: roosting behavior (C-communal, S-solitary); Nest: nest type (C-close cup, H-hole or cavity-nester, O-open cup/nest); Cx. pipiens: forage ratio for Cx. pipiens; Cx. restuans: forage ratio for Cx. restuans. (DOC 123 kb)
Fig S1
Consensus trees obtained from 1000 trees generated on BirdTree ( http://birdtree.org ). The trees generated by adopting a 50% majority-rule consensus tree (SumTrees 3.3.1 in DendroPy 3.12.2) using the code described by Rubolini et al. (2015 Curr Zool 61). Working phylogenies for PGLS analyses included a) 49 avian species with the estimated forage ratio for Cx. pipiens; b) 44 avian species with the estimated forage ratio for Cx. restuans. (GIF 45 kb)
Table S2
Summary statistics of the averaged model derived from the set of top GLM models (∆i(AICc) < 2) explaining variation in the feeding patterns of Cx. pipiens after omitting two raptor species. (DOC 31 kb)
Table S3
Summary statistics of the averaged model derived from the set of top GLM models (∆i(AICc) < 2) explaining variation in the feeding patterns of Cx. pipiens derived from gravid trap. (DOC 32 kb)
Rights and permissions
About this article
Cite this article
Yan, J., Gangoso, L., Martínez-de la Puente, J. et al. Avian phenotypic traits related to feeding preferences in two Culex mosquitoes. Sci Nat 104, 76 (2017). https://doi.org/10.1007/s00114-017-1497-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-017-1497-x