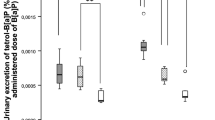
Abstract
In order to survey changes and activities in the polycyclic aromatic hydrocarbon (PAH)-metabolizing enzymes implicated in lung cancer susceptibility studies, we investigated enzyme induction by 2–5-ring-sized ‘biomarker’ PAHs in rat liver and lung, and the activities in five human lung specimens. Naphthalene, phenanthrene, pyrene, chrysene, and benzo[a]pyrene (BaP) were administered to rats for 3 days (25–128 mg/kg/day) and the responses compared with those of model inducers. PAH treatment increased the CYP1A-catalyzed activity of pyrene 1-hydroxylation and 7-ethoxyresorufin O-deethylation in rat liver by up to 28- and 279-fold, and in rat lung by up to 22- and 51-fold, respectively. 1-Naphthol (hUGT1A6), 1-hydroxypyrene (hUGT1A6/1A9), and entacapone (hUGT1A9) are markers of PAH-glucuronidating human uridine diphosphate-glucuronosyltransferases (UGT). These activities increased up to 6.4-fold in rat liver and up to 1.9-fold in rat lung. NADPH:quinone oxidoreductase 1 (NQO1) and glutathione S-transferase activities increased up to 5.3- and 1.6-fold (liver), and up to 4.4- and 1.4-fold (lung), respectively. CYP1A showed the best liver-to-lung relationship (R 2 = 0.90). The inducing efficiency by PAHs differed extensively: control ≤ naphthalene < phenanthrene, pyrene << chrysene < BaP. In human lung (non-smokers), the marker activities of CYP1A1, UGT1A6/1A9, and NQO1 were lower than those in rat lung. Epoxide hydrolase activity was 1,000-fold higher than the pulmonary CYP1A1 activities. Human UGT and NQO1 displayed large variations (>60-fold), many times greater than the experimental (inducible/constitutive) variation in the rat. Kinetics of 1-hydroxypyrene glucuronidation showed two low-K m forms both in rat and human lung. Since the 2–4-ring PAHs (major constituents) were poor enzyme inducers, it appears that the PAH-metabolizing pathways are mainly induced by BaP-type minor constituents. Gene–environmental interactions which magnify polymorphic variability in pulmonary bioactivation/detoxification capacity probably play a key role in individual susceptibility to (or protection against) chemically induced lung cancer. Hence, human exposure to PAH mixtures with high content of BaP-type hydrocarbons confers a potentially higher health risk than PAH mixtures with low content of procarcinogens.



Similar content being viewed by others
Abbreviations
- ALT:
-
Alanine aminotransaminase
- ANF:
-
α-Naphthoflavone
- BNF:
-
β-Naphthoflavone
- BaP:
-
Benzo[a]pyrene
- BSO:
-
l-Buthionine-[S,R]-sulfoximine
- CDNB:
-
1-Chloro-2,4-dinitrobenzene
- CYP or P450:
-
Cytochrome P450
- DCPIP:
-
2,6-Dichlorophenolindophenol
- EQ:
-
Ethoxyquin
- EROD:
-
7-Ethoxyresorufin O-deethylase
- E-UGT:
-
Entacapone UGT
- GST:
-
Glutathione S-transferase
- HPLC:
-
High-performance liquid chromatography
- MC:
-
3-Methylcholanthrene
- mEH:
-
Microsomal epoxide hydrolase
- NQO1:
-
NADPH:quinone oxidoreductase 1 [NMO1, quinone reductase, DT-diaphorase]
- NP-oxide (EPNP):
-
1,2-Epoxy-3-(p-nitrophenoxy)propane
- NP-diol:
-
3-[p-Nitrophenoxy]-1,2-propane diol
- N-UGT:
-
1-Naphthol UGT
- 1-OHP:
-
1-Hydroxypyrene
- PH:
-
Pyrene 1-hydroxylase
- PAH:
-
Polycyclic aromatic hydrocarbon
- UDP:
-
Uridine diphosphate
- UDPGA:
-
UDP-glucuronic acid
- UGT:
-
UDP-glucuronosyltransferase
References
ACGIH (2005) Polycyclic aromatic hydrocarbons (PAHs): BEI®, 7th edn. Documentation ACGIH®. Publication #7DOC-735. American Conference of Governmental Industrial Hygienists, Cincinnati, 17 pp
Angerer J, Mannschreck C, Gundel J (1997) Biological monitoring and biochemical effect monitoring of exposure to polycyclic aromatic hydrocarbons (a review). Int Arch Occup Environ Health 70:365–377
Anttila S, Lei X-D, Elovaara E, Karjalainen A, Sun W, Vainio H, Hankinson O (2000) An uncommon phenotype of poor inducibility of CYP1A1 in human lung is not ascribable to polymorphisms in the AHR, ARNT, or CYP1A1 genes. Pharmacogenetics 10:741–751
Anttila S, Tuominen P, Hirvonen A, Nurminen M, Karjalainen A, Hankinson O, Elovaara E (2001) CYP1A1 levels in lung tissue of tobacco smokers and polymorphisms of CYP1A1 and aromatic hydrocarbon receptor. Pharmacogenetics 11:501–509
Asher G, Lotem J, Sachs L, Shaul Y (2004) p53-dependent apoptosis and NAD(P)H:quinone oxidoreductase 1. Meth Enzymol 382(part B):278–293
Bock KW, Gschaidmeier H, Heel H, Lehmkoster T, Munzel PA, Raschko F, Bock-Hennig B (1998) AH receptor-controlled transcriptional regulation and function of rat and human UDP-glucuronosyltransferase isoforms. Adv Enzyme Regul 38:207–222
Bouchard M, Viau C (1999) Urinary 1-hydroxypyrene as a biomarker of exposure to polycyclic aromatic hydrocarbons: biological monitoring strategies and methodology for determining biological exposure indices for various work environments. Biomarkers 4:159–187, and references therein
Buetler TM, Gallagher EP, Wang C, Stahl DL, Hayes JD, Eaton DL (1995) Induction of phase I and phase II drug-metabolizing enzyme mRNA, protein, and activity by BHA, ethoxyquin, and oltipraz. Toxicol Appl Pharmacol 135:45–57
Burchell B, Brierley CH, Monaghan CH, Clarke DJ (1998) The structure and function of the UDP-glucuronosyltransferase gene family. Adv Pharmacol 42:335–338
Conney AH (1982) Induction of microsomal enzymes by foreign chemicals and carcinogen by polycyclic aromatic hydrocarbons: GHA Clowes Memorial Lecture. Cancer Res 42:4875–4917
Elovaara E, Engström K, Nakajima T, Park SS, Gelboin HV, Vainio H (1991) Metabolism of inhaled styrene in acetone-, phenobarbital-, and 3-methylcholanthrene pretreated rats: stimulation and stereochemical effects by induction of cytochromes P450IIE1, P450IIB and P450IA. Xenobiotica 21:651–661
Elovaara E, Heikkilä P, Pyy L, Mutanen P, Riihimäki V (1995a) Significance of dermal and respiratory uptake in creosote workers: exposure to polycyclic aromatic hydrocarbons and urinary excretion of 1-hydroxypyrene. Occup Environ Med 52:196–203
Elovaara E, Raunio H, Pelkonen O, Vainio H (1995b) Oxidation of pyrene in human liver and lungs. Hum Exp Toxicol 14:821
Elovaara E, Karjalainen A, Vanhala E, Anttila S, Luukkanen L, Vainio H (1997) Effect of asbestos on PAH-metabolizing enzyme activities in lungs from smoking and non-smoking lung cancer surgery patients. In: ISSX proceedings (6th European ISSX Meeting), Göteborg, Sweden, vol. 11, p 64
Elovaara E, Mikkola J, Luukkanen L, Antonio L, Fournel-Gigleux S, Burchell B, Magdalou J, Taskinen J (2004) Assessment of catechol induction and glucuronidation in rat liver microsomes. Drug Metab Dispos 32:1426–1433
Ernster L (1967) DT diaphorase. Meth Enzymol 10:309–317
Gelboin HV (1980) Benzo(alpha)pyrene metabolism, activation and carcinogenesis: role and regulation of mixed-function oxidases and related enzymes. Physiol Rev 60:1107–1166
Giuliano KA, Lau EP, Fall RR (1980) Simplified liquid chromatographic assay for epoxide hydrolase. J Chromatogr 202:447–452
Grainger J, Huang W, Li Z, Selvin E, Walcott C, Smith C, Turner WE, Wang R, Patterson DG Jr (2004) PAH reference range levels in the U.S. population by measurement of urinary monohydroxy metabolites. Polycyclic Aromatic Comp 24:385–404
Grimmer G, Jacob J, Dettbarn G, Naujack KW (1997) Determination of urinary metabolites of polycyclic aromatic hydrocarbons (PAH) for the risk assessment of PAH-exposed workers. Int Arch Occup Environ Health 69:231–239
Guengerich FP (2000) Metabolism of chemical carcinogens. Carcinogenesis 21:345–351
Gundel J, Schaller KH, Angerer J (2000) Occupational exposure to polycyclic aromatic hydrocarbons in a fireproof stone producing plant: biological monitoring of 1-hydroxypyrene, 1-, 2-, 3- and 4-hydroxyphenanthrene, 3-hydroxybenz(a)anthracene and 3-hydroxybenzo(a)pyrene. Int Arch Occup Environ Health 73:270–274
Habig WH, Pabst MJ, Jacoby WB (1974) Glutathione S-transferases. The first step in mercapturic acid formation. J Biol Chem 249:7130–7139
Hayes JD, Pulford DJ (1995) The glutathione S-transferase supergene family. Regulation of GST and the contribution of the isozymes to cancer chemoprotection and drug resistance. Crit Rev Biochem Mol Biol 30:455–600
Hayes JD, Flanagan JU, Jowsey IR (2005) Glutathione transferases. Annu Rev Pharmacol Toxicol 45:51–88
Hecht SS (2002) Human urinary carcinogen metabolites: biomarkers for investigating tobacco and cancer. Carcinogenesis 23:907–922
Huang W, Grainger J, Patterson DG Jr, Turner WE, Caudill SP, Needham LL, Pirkle JL, Sampson EJ (2004) Comparison of 1-hydroxypyrene exposure in the US population with that in occupational exposure studies. Int Arch Occup Environ Health 77:491–498
Hukkanen J, Pelkonen O, Hakkola J, Raunio H (2002) Expression and regulation of xenobiotic-metabolizing enzymes cytochrome P450 (CYP) enzymes in human lung. Crit Rev Toxicol 32:391–411
IARC (1985) IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans, vol. 35, Polynuclear aromatic compounds. Part 4, Bitumens, coal-tars and derived products, shale oils and soots. IARC, Lyon, France
Jacob J, Schmoldt A, Grimmer G (1982) Influence of monooxygenase inducers on the metabolic profile of phenanthrene in rat liver microsomes. Toxicology 25:333–343
Jaiswal AK (2000) Regulation of genes encoding NAD(P)H:quinone oxidoreductases. Free Radic Biol Med 29:254–262
Jinno H, Saeki M, Saito Y, Tanaka-Kagawa T, Hanioka N, Sai K, Kaniwa N, Ando M, Shirao K, Minami H, Ohtsu A, Yoshida T, Saijo N, Ozawa S, Sawada J (2003) Functional characterization of human UDP-glucuronosyltransferase 1A9 variant, D256N, found in Japanese cancer patients. J Pharmacol Exp Ther 306:688–693
Joseph P, Jaiswal AK (1994) NAD(P)H:quinone oxidoreductase1 (DT diaphorase) specifically prevents the formation of benzo[a]pyrene quinone-DNA adducts generated by cytochrome P4501A1 and P450 reductase. Proc Natl Acad Sci USA 91:8413–8417
Keski-Hynnilä H, Raanaa K, Forsberg M, Männistö P, Taskinen J, Kostiainen R (2001) Quantitation of entacapone glucuronide in rat plasma by on-line coupled restricted access media column and liquid chromatography–tandem mass spectrometry. J Chromatogr B Biomed Sci Appl 759:227–236
Kim JH, Sherman ME, Curriero FC, Guengerich FP, Strickland PT, Sutter TR (2004a) Expression of cytochromes P450 1A1 and 1B1 in human lung from smokers, non-smokers, and ex-smokers. Toxicol Appl Pharmacol 199:210–219
Kim YD, Todoroki H, Oyama T, Isse T, Matsumoto A, Yamaguchi T, Kim H, Uchiyama I, Kawamoto T (2004b) Identification of cytochrome P450 isoforms involved in 1-hydroxylation of pyrene. Environ Res 94:262–266
Kiyohara C, Otsu A, Shirakawa T, Fukuda S, Hopkin JM (2002) Genetic polymorphisms and lung cancer susceptibility: a review. Lung Cancer 37:241–256
Koley AP, Buters JTM, Robinson RC, Markowitz A, Friedman FK (1997) Differential mechanisms of cytochrome P450 inhibition and activation by alpha-naphthoflavone. J Biol Chem 272:3149–3152
Lowry O, Rosebrough N, Farr L, Randall R (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Luukkanen L, Elovaara E, Lautala P, Taskinen J, Vainio H (1997) Characterization of 1-hydroxypyrene as a novel marker substrate of 3-methylcholanthrene-inducible phenol UDP-glucuronosyltransferase(s). Pharmacol Toxicol 80:152–158
Luukkanen L, Mikkola J, Forsman T, Taavitsainen P, Taskinen J, Elovaara E (2001) Glucuronidation of 1-hydroxypyrene by human liver microsomes and human UDP-glucuronosyltransferases UGT1A6, UGT1A7, and UGT1A9: development of a high-sensitivity glucuronidation assay for human tissue. Drug Metab Dispos 29:1096–1101
Luukkanen L, Taskinen J, Kurkela M, Kostiainen R, Hirvonen J, Finel M (2005) Kinetic characterization of the 1A subfamily of recombinant human UDP-glucuronosyltransferases. Drug Metab Dispos 33:1017–1026
Manning BW, Franklin MR (1990) Induction of rat UDP-glucuronosyltransferase and glutathione S-transferase activities by l-buthionine-S,R-sulfoximine without induction of cytochrome P-450. Toxicology 65:149–159
Myllynen P, Pasanen M, Pelkonen O (2005) Human placenta: a human organ for developmental toxicology research and biomonitoring. Placenta 26:361–371 (review)
Nakajima T, Elovaara E, Anttila S, Hirvonen A, Camus A-M, Hayes JD, Ketterer B, Vainio H (1995) Expression and polymorphism of glutathione S-transferases in human lungs: risk factors in smoking-related lung cancer. Carcinogenesis 16:707–711
Nebert DW, Dalton TP, Okey AB, Gonzalez FJ (2004) Role of aryl hydrocarbon receptor-mediated induction of the CYP1 enzymes in environmental toxicity and cancer. J Biol Chem 23:23847–23850
Nioi P, Hayes JD (2004) Contribution of NAD(P)H:quinone oxidoreductase 1 to protection against carcinogenesis, and regulation of its gene by Nrf2 basic-region zipper and the arylhydrocarbon receptor basic helix-loop-helix transcription factors. Mutat Res 555:149–171
Paakki P, Stockmann H, Kantola M, Wagner P, Lauper U, Huch R, Elovaara E, Kirkinen P, Pasanen M (2000) Maternal drug abuse and human placental term xenobiotic and steroid metabolizing enzymes in vitro. Environ Health Perspect 108:141–145
Pelkonen O, Nebert D (1982) Metabolism of polycyclic aromatic hydrocarbons: etiologic role in carcinogenesis. Pharmacol Rev 34:189–221
Pelkonen O, Pasanen M, Kuha H, Gachalyi B, Kairaluoma M, Sotaniemi EA, Park SS, Friedman FK, Gelboin HV (1986) The effect of cigarette smoking on 7-ethoxycoumarin O-deethylase and other monooxygenase activities in human liver: analyses with monoclonal antibodies. Br J Clin Pharmacol 22:125–134
Pelkonen O, Raunio H, Rautio A, Lang M (1999) Xenobiotic-metabolizing enzymes and cancer risk: correspondence between genotype and phenotype. IARC Sci Publ 148:77–88 (review)
Penning TM, Burczynski ME, Hung CF, McCoull KD, Palackal NT, Tsuruda LS (1999) Dihydrodiol dehydrogenases and polycyclic aromatic hydrocarbon activation: generation of reactive and redox active o-quinones. Chem Res Toxicol 12:1–18 (review)
Prough RA, Burke MD, Mayer RT (1978) Direct fluorometric methods for measuring mixed-function oxidase activity. Meth Enzymol 52(part C):373–377
Ross D, Siegel D (2004) NAD(P)H:quinone oxidoreductase 1 (NQO1, DT-diaphorase) functions and pharmacogenetics. Meth Enzymol 382(part B):115–144
Schlager JJ, Powis G (1990) Cytosolic NAD(P)H:(quinone-acceptor)oxidoreductase in human normal and tumor tissue: effects of cigarette smoking and alcohol. Int J Cancer 45:403–409
Shimada T, Fujii-Kuriyama Y (2004) Metabolic activation of polycyclic aromatic hydrocarbons to carcinogens by cytochrome P450 1A1 and 1B1. Cancer Sci 95:1–6
Shimada T, Yun CH, Yamazaki H, Gautier JC, Beaune PH, Guengerich FP (1992) Characterization of human lung microsomal cytochrome P-450 1A1 and its role in the oxidation of chemical carcinogens. Mol Pharmacol 41:856–864
Shou M, Grogan J, Mancewicz JA, Krausz KW, Gonzalez FJ, Gelboin HV, Korzekwa KR (1994) Activation of CYP3A4: evidence for the simultaneous binding of two substrates in a cytochrome P450 active site. Biochemistry 33:6450–6455
Thier R, Bruning T, Roos PH, Rihs H-P, Golka K, Ko Y, Bolt HM (2003) Markers of genetic susceptibility in human environmental hygiene and toxicology: the role of selected CYP, NAT and GST genes. Int J Hyg Environ Health 206:149–171
UBA (1998) Umweltbundesamt: Umwelt-Survey, Berlin 1998. http://www.umweltbundesamt.de/survey/us98/pak.htm
Vainio H, Elovaara E, Luukkanen L, Anttila S, Ulmanen I, Fournel-Gigleux S, Ouzzine M, Pillot T, Magdalou M (1995) Expression and co-induction of CYP1A1 and UGT1*6 in human lungs. Eur J Drug Metab Pharmacokinet 20:47–48
Vineis P (2004) Individual susceptibility to carcinogens. Oncogene 23:6477–6483
Wells PG, Mackenzie PI, Chowdhury JR, Guillemette C, Gregory PA, Ishii Y, Hansen AJ, Kessler FK, Kim PM, Chowdhury NR, Ritter JK (2004) Glucuronidation and the UDP-glucuronosyltransferases in health and disease. Drug Metab Dispos 32:281–290 (review)
Yang M, Koga M, Katoh T, Kawamoto T (1999) A study for the proper application of urinary naphthols, new biomarkers for airborne polycyclic aromatic hydrocarbons. Arch Environ Contam Toxicol 36:99–108
Ye Z, Song H, Higgins JP, Pharoah P, Danesh J (2006) Five glutathione S-transferase gene variants in 23,452 cases of lung cancer and 30,397 controls: meta-analysis of 130 studies. PLoS Med 3(4):e91 [Epub 2006 March 7]
Zheng Z, Fang JL, Lazarus P (2002) Glucuronidation: an important mechanism for detoxification of benzo[a]pyrene in aerodigestive tract tissues. Drug Metab Dispos 30:397–403
Acknowledgments
We wish to thank Ms. Maria Pihlaja and Ms. Ulla Peltonen for skillful technical assistance. UGT studies were associated with the BMH4-CT97-2621 project supported by the Commission of the European Communities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elovaara, E., Mikkola, J., Stockmann-Juvala, H. et al. Polycyclic aromatic hydrocarbon (PAH) metabolizing enzyme activities in human lung, and their inducibility by exposure to naphthalene, phenanthrene, pyrene, chrysene, and benzo(a)pyrene as shown in the rat lung and liver. Arch Toxicol 81, 169–182 (2007). https://doi.org/10.1007/s00204-006-0135-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-006-0135-8