Abstract
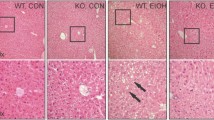
We investigated the responses of mice that are defective in the superoxide-scavenging enzyme SOD1 to thioacetamide (TAA)-induced hepatotoxicity. When a lethal dose of TAA (500 mg/kg) was intraperitoneally injected, the wild-type (WT) mice all died within 36 h, but all of the SOD1-knockout (KO) mice survived. Treatment with an SOD1 inhibitor rendered the WT mice resistant to TAA toxicity. To elucidate the mechanism responsible for this, we examined the acute effects of a sublethal dose of TAA (200 mg/kg) on the livers of WT and KO mice. The extent of TAA-induced liver damage was less in the KO mice, but, instead, lipogenesis was further advanced in the SOD1-KO livers. The levels of proteins modified with acetyllysine, a marker for TAA-mediated injury, were lower in the KO mice than the WT mice upon the TAA treatment. The KO mice, which were under oxidative stress per se, exhibited a lower CYP2E1 activity, and this appeared to result in a decrease in the production of reactive oxygen species (ROS) during TAA metabolism. Both cleaved ATF6, a transcriptional regulator that is activated by endoplasmic reticulum (ER) stress, and CHOP, a death signal mediator, were highly elevated in the WT mice as the result of the TAA treatment and consistent with the liver damage. We conclude that elevated TAA metabolites and reactive oxygen species that are produced by CYP-mediated drug metabolism trigger lipogenesis as well as liver damage via ER stress and determine the fate of the mice.











Similar content being viewed by others
Abbreviations
- NAFLD:
-
Non-alcoholic fatty liver disease
- TAA:
-
Thioacetamide
- TAA-SO:
-
Thioacetamide S-oxide
- TAA-SO2 :
-
Thioacetamide S,S-dioxide
- SOD:
-
Superoxide dismutase
- ROS:
-
Reactive oxygen species
- Prx:
-
Peroxiredoxin
- ALT:
-
Alanine aminotransferase
- ER:
-
Endoplasmic reticulum
- CYP:
-
Cytochrome P450
- KO:
-
Knockout
- WT:
-
Wild type
- i.p.:
-
Intraperitoneally
- PBS:
-
Phosphate-buffered saline
- ATTM:
-
Ammonium tetrathiomolybdate
- H&E:
-
Hematoxylin and eosin
- TG:
-
Triglyceride
- Nrf2:
-
NF-E2-related factor 2
- HNE:
-
4-Hydroxyl 2-nonenal
References
Banerjee A, Kocarek TA, Novak RF (2000) Identification of a ubiquitination-target/substrate-interaction domain of cytochrome P-450 (CYP) 2E1. Drug Metab Dispos 28:118–124
Brown GT, Kleiner DE (2015) Histopathology of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Metabolism. doi:10.1016/j.metabol.2015.11.008
Busuttil RA, Garcia AM, Cabrera C et al (2005) Organ-specific increase in mutation accumulation and apoptosis rate in CuZn-superoxide dismutase-deficient mice. Cancer Res 65:11271–11275. doi:10.1158/0008-5472.CAN-05-2980
Carr RM, Peralta G, Yin X, Ahima RS (2014) Absence of perilipin 2 prevents hepatic steatosis, glucose intolerance and ceramide accumulation in alcohol-fed mice. PLoS ONE 9:e97118. doi:10.1371/journal.pone.0097118
Chiueh CC, Andoh T, Chock PB (2005) Induction of thioredoxin and mitochondrial survival proteins mediates preconditioning-induced cardioprotection and neuroprotection. Ann N Y Acad Sci 1042:403–418
Elchuri S, Oberley TD, Qi W et al (2005) CuZnSOD deficiency leads to persistent and widespread oxidative damage and hepatocarcinogenesis later in life. Oncogene 24:367–380. doi:10.1038/sj.onc.1208207
Elchuri S, Naeemuddin M, Sharpe O et al (2007) Identification of biomarkers associated with the development of hepatocellular carcinoma in CuZn superoxide dismutase deficient mice. Proteomics 7:2121–2129. doi:10.1002/pmic.200601011
Fridovich I (1995) Superoxide radical and superoxide dismutases. Annu Rev Biochem 64:97–112. doi:10.1146/annurev.bi.64.070195.000525
Fujii T, Endo T, Fujii J, Taniguchi N (2002) Differential expression of glutathione reductase and cytosolic glutathione peroxidase, GPX1, in developing rat lungs and kidneys. Free Radic Res 36:1041–1049
Goldstein JL, DeBose-Boyd RA, Brown MS (2006) Protein sensors for membrane sterols. Cell 124:35–46. doi:10.1016/j.cell.2005.12.022
Hadjur S, Ung K, Wadsworth L et al (2001) Defective hematopoiesis and hepatic steatosis in mice with combined deficiencies of the genes encoding Fancc and Cu/Zn superoxide dismutase. Blood 98:1003–1011
Hajovsky H, Hu G, Koen Y et al (2012) Metabolism and toxicity of thioacetamide and thioacetamide S-oxide in rat hepatocytes. Chem Res Toxicol 25:1955–1963. doi:10.1021/tx3002719
Hinson JA, Roberts DW, James LP (2010) Mechanisms of acetaminophen-induced liver necrosis. Handb Exp Pharmacol. doi:10.1007/978-3-642-00663-0_12
Hinz B, Phan SH, Thannickal VJ et al (2012) Recent developments in myofibroblast biology: paradigms for connective tissue remodeling. Am J Pathol 180:1340–1355. doi:10.1016/j.ajpath.2012.02.004
Homma T, Fujii J (2015) Application of glutathione as anti-oxidative and anti-aging drugs. Curr Drug Metab 16:560–571
Homma T, Kurahashi T, Lee J et al (2015) SOD1 deficiency decreases proteasomal function, leading to the accumulation of ubiquitinated proteins in erythrocytes. Arch Biochem Biophys 583:65–72. doi:10.1016/j.abb.2015.07.023
Ingawale DK, Mandlik SK, Naik SR (2014) Models of hepatotoxicity and the underlying cellular, biochemical and immunological mechanism(s): a critical discussion. Environ Toxicol Pharmacol 37:118–133. doi:10.1016/j.etap.2013.08.015
Ishii T, Matsuki S, Iuchi Y et al (2005) Accelerated impairment of spermatogenic cells in SOD1-knockout mice under heat stress. Free Radic Res 39:697–705. doi:10.1080/10715760500130517
Iuchi Y, Okada F, Onuma K et al (2007) Elevated oxidative stress in erythrocytes due to a SOD1 deficiency causes anaemia and triggers autoantibody production. Biochem J 402:219–227. doi:10.1042/BJ20061386
Iuchi Y, Okada F, Tsunoda S et al (2009) Peroxiredoxin 4 knockout results in elevated spermatogenic cell death via oxidative stress. Biochem J 419:149–158. doi:10.1042/BJ20081526
Kang JS, Wanibuchi H, Morimura K et al (2008) Role of CYP2E1 in thioacetamide-induced mouse hepatotoxicity. Toxicol Appl Pharmacol 228:295–300. doi:10.1016/j.taap.2007.11.010
Karuzina II, Archakov AI (1994a) The oxidative inactivation of cytochrome P450 in monooxygenase reactions. Free Radic Biol Med 16:73–97
Karuzina II, Archakov AI (1994b) Hydrogen peroxide-mediated inactivation of microsomal cytochrome P450 during monooxygenase reactions. Free Radic Biol Med 17:557–567
Kaushik S, Cuervo AM (2015) Degradation of lipid droplet-associated proteins by chaperone-mediated autophagy facilitates lipolysis. Nat Cell Biol 17:759–770. doi:10.1038/ncb3166
Koen YM, Sarma D, Hajovsky H et al (2013) Protein targets of thioacetamide metabolites in rat hepatocytes. Chem Res Toxicol 26:564–574. doi:10.1021/tx400001x
Kurahashi T, Konno T, Otsuki N et al (2012) A malfunction in triglyceride transfer from the intracellular lipid pool to apoB in enterocytes of SOD1-deficient mice. FEBS Lett 586:4289–4295. doi:10.1016/j.febslet.2012.09.047
Kurahashi T, Hamashima S, Shirato T et al (2015) An SOD1 deficiency enhances lipid droplet accumulation in the fasted mouse liver by aborting lipophagy. Biochem Biophys Res Commun 467:866–871. doi:10.1016/j.bbrc.2015.10.052
Lee J, Homma T, Kurahashi T et al (2015) Oxidative stress triggers lipid droplet accumulation in primary cultured hepatocytes by activating fatty acid synthesis. Biochem Biophys Res Commun 464:229–235. doi:10.1016/j.bbrc.2015.06.121
Lei XG, Zhu J-H, McClung JP et al (2006) Mice deficient in Cu, Zn-superoxide dismutase are resistant to acetaminophen toxicity. Biochem J 399:455–461. doi:10.1042/BJ20060784
López-Islas A, Chagoya-Hazas V, Pérez-Aguilar B et al (2016) Cholesterol enhances the toxic effect of ethanol and acetaldehyde in primary mouse hepatocytes. Oxid Med Cell Longev 2016:9209825. doi:10.1155/2016/9209825
Low TY, Leow CK, Salto-Tellez M, Chung MCM (2004) A proteomic analysis of thioacetamide-induced hepatotoxicity and cirrhosis in rat livers. Proteomics 4:3960–3974. doi:10.1002/pmic.200400852
Malhotra JD, Kaufman RJ (2007) Endoplasmic reticulum stress and oxidative stress: a vicious cycle or a double-edged sword? Antioxid Redox Signal 9:2277–2293. doi:10.1089/ars.2007.1782
Mallat A, Lotersztajn S (2013) Cellular mechanisms of tissue fibrosis. 5. Novel insights into liver fibrosis. Am J Physiol Cell Physiol 305:C789–C799. doi:10.1152/ajpcell.00230.2013
Miyahara Y, Ikeda S, Muroya T et al (2002) Nepsilon-(carboxymethyl)lysine induces gamma-glutamylcysteine synthetase in RAW264.7 cells. Biochem Biophys Res Commun 296:32–40
Okado-Matsumoto A, Fridovich I (2001) Subcellular distribution of superoxide dismutases (SOD) in rat liver: Cu, Zn-SOD in mitochondria. J Biol Chem 276:38388–38393. doi:10.1074/jbc.M105395200
Pierre N, Barbé C, Gilson H et al (2014) Activation of ER stress by hydrogen peroxide in C2C12 myotubes. Biochem Biophys Res Commun 450:459–463. doi:10.1016/j.bbrc.2014.05.143
Rahman I, Kode A, Biswas SK (2006) Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat Protoc 1:3159–3165. doi:10.1038/nprot.2006.378
Sarma D, Hajovsky H, Koen YM et al (2012) Covalent modification of lipids and proteins in rat hepatocytes and in vitro by thioacetamide metabolites. Chem Res Toxicol 25:1868–1877. doi:10.1021/tx3001658
Serviddio G, Bellanti F, Vendemiale G (2013) Free radical biology for medicine: learning from nonalcoholic fatty liver disease. Free Radic Biol Med 65:952–968. doi:10.1016/j.freeradbiomed.2013.08.174
Shau H, Merino A, Chen L et al (2000) Induction of peroxiredoxins in transplanted livers and demonstration of their in vitro cytoprotection activity. Antioxid Redox Signal 2:347–354. doi:10.1089/ars.2000.2.2-347
Uchiyama S, Shimizu T, Shirasawa T (2006) CuZn-SOD deficiency causes ApoB degradation and induces hepatic lipid accumulation by impaired lipoprotein secretion in mice. J Biol Chem 281:31713–31719. doi:10.1074/jbc.M603422200
Wallace MC, Hamesch K, Lunova M et al (2015) Standard operating procedures in experimental liver research: thioacetamide model in mice and rats. Lab Anim 49:21–29. doi:10.1177/0023677215573040
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334:1081–1086. doi:10.1126/science.1209038
Wang L, Jiang Z, Lei XG (2012) Knockout of SOD1 alters murine hepatic glycolysis, gluconeogenesis, and lipogenesis. Free Radic Biol Med 53:1689–1696. doi:10.1016/j.freeradbiomed.2012.08.570
Wei H, Li Z, Hu S et al (2010) Apoptosis of mesenchymal stem cells induced by hydrogen peroxide concerns both endoplasmic reticulum stress and mitochondrial death pathway through regulation of caspases, p38 and JNK. J Cell Biochem 111:967–978. doi:10.1002/jcb.22785
Yasui H, Hayashi S, Sakurai H (2005) Possible involvement of singlet oxygen species as multiple oxidants in p450 catalytic reactions. Drug Metab Pharmacokinet 20:1–13
Zhou H, Liu R (2014) ER stress and hepatic lipid metabolism. Front Genet 5:1–7. doi:10.3389/fgene.2014.00112
Zhu J-H, Zhang X, Roneker CA et al (2008) Role of copper, zinc-superoxide dismutase in catalyzing nitrotyrosine formation in murine liver. Free Radic Biol Med 45:611–618. doi:10.1016/j.freeradbiomed.2008.05.018
Financial supports
J.L. is a scholarship recipient from the Otsuka Toshimi Scholarship Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shirato, T., Homma, T., Lee, J. et al. Oxidative stress caused by a SOD1 deficiency ameliorates thioacetamide-triggered cell death via CYP2E1 inhibition but stimulates liver steatosis. Arch Toxicol 91, 1319–1333 (2017). https://doi.org/10.1007/s00204-016-1785-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-016-1785-9