Abstract.
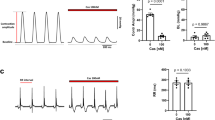
Mibefradil is a tetralol derivative that inhibits cloned and native T-type voltage-operated calcium channels (VOCCs) at an IC50 of 1 µM and with an apparent 15-fold selectivity for T- over L-type VOCCs. Recent electrophysiological studies in Xenopus oocytes and inhibition of noradrenaline release studies in human isolated right atria concluded that mibefradil (0.3–3 µM) can block N-type VOCCs like ω-conotoxin GVIA. We tested this hypothesis in rat and guinea pig isolated driven left atria and rat vas deferens in response to sympathetic nerve stimulation. Mibefradil (3 µM) did not inhibit the inotropic responses to sympathetic electrical field stimulation in atria. In contrast, these responses were blocked by GVIA (10 nM) and tetrodotoxin (TTX 0.1 µM). In rat vas deferens, pretreated with benextramine (BNX 10 µM), contractions considered to be due to transmitter ATP evoked by electrical field stimulation were blocked by: (a) TTX 0.1 µM, (b) P2x receptor desensitisation (αβ-MeATP 100 µM), and (c) GVIA (pIC50 9.04±0.15, n=5). In contrast, mibefradil (0.3–30 µM) had no effect on these N-type VOCC-sensitive sympathetic nerve responses. Potassium (62 mM K+) and αβ-MeATP-induced contractions were unaffected by GVIA (10 nM), but mibefradil inhibited the potassium-induced contractions with a pIC50 6.02±0.08 (n=5), consistent with a T- and L-type VOCC blocking action on the postjunctional smooth muscle. We conclude that mibefradil up to 30 µM does not block N-type VOCCs in these isolated intact tissue assays of sympathetic nerve-muscle transmission. Therefore, at least in these tissues, mibefradil can be used to define T- and L-type VOCC activities at submicromolar and micromolar concentrations in sympathetic nerve-mediated responses.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Xi, Q., Angus, J.A. Evidence against an action of mibefradil at N-type voltage-operated calcium channels. Naunyn-Schmied Arch Pharmacol 364, 430–436 (2001). https://doi.org/10.1007/s002100100470
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002100100470