Abstract
Rationale
The endogenous cannabinoid system plays a vital role in the control of nausea and emesis. Because of the rapid breakdown and hydrolysis of endocannabinoids, such as anandamide, the therapeutic effects may be enhanced by prolonging their duration of action.
Objective
The present experiment evaluated the potential of various doses of URB597, a fatty acid amide hydrolase (FAAH) inhibitor, alone and in combination with systemic administration of anandamide to modulate the establishment of lithium-induced conditioned taste reactivity responses in rats.
Materials and methods
In experiment 1, on the conditioning day, rats first received an injection of 0.3 mg/kg URB597, 0.15 mg/kg URB597, or vehicle and then received a second injection of anandamide (5 mg/kg) or vehicle, before a 3-min exposure of 0.1% saccharin by intraoral infusion. Immediately after the saccharin exposure, the rats were injected with lithium chloride. On each of three test days, rats received a 3-min intraoral infusion of saccharin solution, and the taste reactivity responses were videotaped and monitored. In experiment 2, the effects of pretreatment with the CB1 antagonist, AM-251, on URB597 and anandamide-induced suppressed aversion was evaluated.
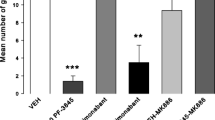
Results
Administration of URB597 alone and in combination with anandamide reduced active rejection reactions elicited by a LiCl-paired saccharin solution; both effects were reversed by pretreatment with AM-251, suggesting that they were CB1 receptor mediated.
Conclusions
The results suggest that prolonging the action of anandamide by pretreatment with the FAAH inhibitor, URB597, suppresses lithium-induced nausea in the rat.




Similar content being viewed by others
References
Arizzi MN, Cervone KM, Aberman JE, Betz A, Liu Q, Lin S, Makriyannis A, Salamone JD (2004) Behavioral effects of inhibition of cannabinoid metabolism: the amidase inhibitor AM374 enhances the suppression of lever pressing produced by exogenously administered anandamide. Life Sci 74:1001–1011
Breslin PAS, Grill HJ, Spector AC (1992) A quantitative comparison of taste reactivity behaviors to sucrose before and after lithium chloride pairings: a unidimensional account of palatability. Behav Neurosci 106:820–836
Darmani NA (2001) Delta(9)-tetrahydrocannabinol and synthetic cannabinoids prevent emesis produced by the cannabinoid CB(1) receptor antagonist/inverse agonist SR 141716A. Neuropsychopharmacology 24:198–203
Darmani NA (2002) The potent emetogenic effects of the endocannabinoid, 2-AG (2-arachidonoylglycerol) are blocked by delta (9)-tetrahydrocannabinol and other cannnabinoids. J Pharmacol Exp Ther 300:34–42
Darmani NA, McClanahan BA, Trinh C, Petrosino S, Valenti M, Di Marzo V (2005) Cisplatin increases brain 2-arachidonoylglycerol (2-AG) and concomitantly reduces intestinal 2-AG and anandamide levels in the least shrew. Neuropharmacology 48:1154–1163
Devane WA, Hanus L, Breuer A, Pertwee RG, Stevenson LA, Griffin G, Gibson D, Mandelbaum A, Etinger A, Mechoulam R (1992) Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258:1946–1949
Di Marzo V, Fontana A, Cadas H, Schinelli S, Cimino G, Schwartz JC, Piomelli D (1994) Formation and inactivation of endogenous cannabinoid anandamide in central neurons. Nature 372:686–691
Eckel LA, Ossenkopp K-P (1996) Area postrema mediates the formation of rapid, conditioned palatability shifts in lithium-treated rats. Behav Neurosci 110:202–212
Fegley D, Gaetani S, Duranti A, Tontini A, Mor M, Tarzia G, Piomelli D (2005) Characterization of the fatty acid amide hydrolase inhibitor cyclohexyl carbamic acid 3′-carbamoyl-biphenyl-3-yl ester (URB597): effects on anandamide and oleoylethanolamide deactivation. J Pharmacol Exp Ther 313:352–358
Gifford AN, Bruneus M, Lin S, Goutopoulos A, Makriyannis A, Volkow ND, Gatley SJ (1999) Potentiation of the action of anandamide on hippocampal slices by the fatty acid amide hydrolase inhibitor, palmitylsulphonyl fluoride (AM 374). Eur J Pharmacol 383:9–14
Grill HJ, Norgren R (1978) The taste reactivity test I. Mimetic responses to gustatory stimuli in neurologically normal rats. Brain Res 143:263–279
Hatcher RA (1924) The mechanism of vomiting. Physiol Rev 4:479–504
Kathuria S, Gaetani S, Fegley D, Valino F, Duranti A, Tontini A, Mor M, Tarzia G, La Rana G, Calignano A, Giustino A, Tattoli M, Palmery M, Cuomo V, Piomelli D (2003) Modulation of anxiety through blockade of anandamide hydrolysis. Nat Med 9:76–81
Kwiatkowska M, Parker LA, Burton P, Mechoulam R (2004) A comparative analysis of the potential of cannabinoids and ondansetron to suppress cisplatin-induced emesis in the Suncus murinus (house musk shrew). Psychopharmacology 174:254–259
Limebeer CL, Parker LA (2000) The antiemetic drug ondansetron interferes with lithium-induced conditioned rejection reactions, but not lithium-induced taste avoidance in rats. J Exp Psychol Anim Behav Process 26:371–384
Limebeer CL, Parker LA, Fletcher PJ (2004) 5,7-dihydroxytryptamine lesions of the dorsal and median raphe nuclei interfere with lithium-induced conditioned gaping, but not conditioned taste avoidance, in rats. Behav Neurosci 118:1391–1399
McLaughlin PJ, Winston KM, Limebeer CL, Parker LA, Makriyannis A, Salamone JD (2005) The cannabinoid CB1 antagonist AM 251 produces food avoidance and behaviors associated with nausea but does not impair feeding efficiency in rats. Psychopharmacology 180:286–293
Ossenkopp K-P, Eckel LA (1995) Toxin-induced conditioned changes in taste reactivity and the role of the chemosensitive area postrema. Neurosci Biobehav Rev 19:99–108
Parker LA (1980) Conditioned suppression of drinking: a measure of the CR elicited by a lithium-conditioned flavor. Learn Motiv 11:538–559
Parker LA (1998) Emetic drugs produce conditioned rejection reactions in the taste reactivity test. J Psychophysiol 12:3–13
Parker LA (2003) Taste avoidance and taste aversion: evidence for two different processes. Learn Behav 31:165–172
Parker LA, Kemp SW (2001) Tetrahydrocannabinol (THC) interferes with conditioned retching in Suncus murinus: an animal model of anticipatory nausea and vomiting (ANV). NeuroReport 12:749–751
Parker LA, Mechoulam R, Schlievert C, Abbott L, Fudge ML, Burton P (2003) Effects of cannabinoids on lithium-induced conditioned rejection reactions in a rat model of nausea. Psychopharmacology 166:156–162
Parker LA, Kwiatkowska M, Burton P, Mechoulam R (2004) Effect of cannabinoids on lithium-induced vomiting in the Suncus murinus (house musk shrew). Psychopharmacology 171:156–161
Piomelli D (2003) The molecular logic of endocannabinoid signalling. Nat Rev Neurosci 4:873–884
Van Sickle MD, Oland LD, Ho W, Hillard CJ, Mackie K, Davison JS, Sharkey KA (2001) Cannabinoids inhibit emesis through CB1 receptors in the brainstem of the ferret. Gastroenterology 121:767–774
Van Sickle MD, Oland LD, Mackie K, Davison JS, Sharkey KA (2003) Delta9-tetrahydrocannabinol selectively acts on CB1 receptors in specific regions of dorsal vagal complex to inhibit emesis in ferrets. Am J Physiol Gastrointest Liver Physiol 285:G566–G576
Acknowledgements
This research was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) operating grants awarded to L.A.P. and K. P.O. and an NSERC postdoctoral fellowship to S.K.C-M. D.P. is funded by grants from the National Institute on Drug Abuse, the University of California Discovery Program, the National Alliance for Research on Schizophrenia and Depression, and Kadmus Pharmaceuticals Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cross-Mellor, S.K., Ossenkopp, KP., Piomelli, D. et al. Effects of the FAAH inhibitor, URB597, and anandamide on lithium-induced taste reactivity responses: a measure of nausea in the rat. Psychopharmacology 190, 135–143 (2007). https://doi.org/10.1007/s00213-006-0589-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0589-7