Abstract
Rationale
Previous studies have shown that alterations in acetylcholine (ACh) receptor subtypes might contribute to cognitive impairment observed in schizophrenia and that choline acetyltransferase activity in the parietal cortex is negatively correlated with the severity of such cognitive impairment. However, clinical data suggest that the acetylcholinesterase (AChE) inhibitors galantamine and donepezil have different effects on negative and cognitive symptoms in schizophrenia. Prepulse inhibition (PPI) deficits—sensory information-processing deficits observed in schizophrenia—may be useful models for studying the efficacy of AChE inhibitors as cognitive enhancers.
Objectives
The present study examined the effects of galantamine and donepezil on PPI deficits induced by an environmental factor and drugs.
Materials and methods
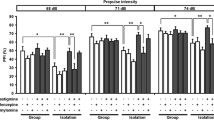
In the isolation-rearing model, 3-week-old male ddY mice were housed either in groups of five or six per cage or isolated in cages of the same size for more than 6 weeks. In the drug-induced model, apomorphine 1 mg/kg and MK-801 0.2 mg/kg were administered to 9- to 10-week-old male ddY mice.
Results
In isolation-reared mice, galantamine attenuated PPI deficits, while donepezil did not. Galantamine and donepezil both attenuated PPI deficits induced by apomorphine, but not by MK-801. The galantamine-induced improvements in PPI deficits were not prevented by the nicotinic ACh receptor antagonists mecamylamine and methyllycaconitine.
Conclusions
These observations suggest that galantamine and donepezil have different effects on the environmentally induced PPI deficits and that these observations may be relevant to the different effects of these drugs observed clinically in schizophrenia.





Similar content being viewed by others
References
Ago Y, Sakaue M, Baba A, Matsuda T (2002) Selective reduction by isolation rearing of 5-HT1A receptor-mediated dopamine release in vivo in the frontal cortex of mice. J Neurochem 83:353–359
Allen TB, McEvoy JP (2002) Galantamine for treatment-resistant schizophrenia. Am J Psychiatry 159:1244–1245
Bitsios P, Giakoumaki SG, Theou K, Frangou S (2006) Increased prepulse inhibition of the acoustic startle response is associated with better strategy formation and execution times in healthy males. Neuropsychologia 44:2494–2499
Bora E, Veznedaroğlu B, Kayahan B (2005) The effect of galantamine added to clozapine on cognition of five patients with schizophrenia. Clin Pharmacol 28:139–141
Braff DL, Light GA (2004) Preattentional and attentional cognitive deficits as targets for treating schizophrenia. Psychopharmacology 174:75–85
Braff DL, Swerdlow NR, Geyer MA (1999) Symptom correlates of prepulse inhibition deficits in male schizophrenic patients. Am J Psychiatry 156:596–602
Cilia J, Reavill C, Hagan JJ, Jones DNC (2001) Long-term evaluation of isolation-reared prepulse inhibition deficits in rats. Psychopharmacology (Berl) 156:327–337
Cilia J, Cluderay JE, Robbins MJ, Reavill C, Southam E, Kew JNC, Jones DNC (2005) Reversal of isolation-rearing-induced PPI deficits by an α7 nicotinic receptor agonist. Psychopharmacology (Berl) 182:214–219
Dajas-Bailador FA, Heimala K, Wonnacott S (2003) The allosteric potentiation of nicotinic acetylcholine receptors by galantamine is transduced into cellular responses in neurons: Ca2+ signals and neurotransmitter release. Mol Pharmacol 64:1217–1226
Drew AE, Derbez AE, Werling LL (2000) Nicotinic receptor-mediated regulation of dopamine transporter activity in rat prefrontal cortex. Synapse 38:10–16
Friedman JI (2004) Cholinergic targets for cognitive enhancement in schizophrenia: focus on cholinesterase inhibitors and muscarinic agonists. Psychopharmacology (Berl) 174:45–53
Friedman JI, Adler DN, Howanitz E, Harvey PD, Brenner G, Temporini H et al (2003) A double blind placebo controlled trail of donepezil adjunctive treatment to risperidone for the cognitive impairment of schizophrenia. Biol Psychiatry 51:349–357
Geyer MA, Krebs-Thomson K, Braff DL, Swerdlow NE (2001) Pharmacological studies of prepulse inhibition models of sensorimotor gating deficits in schizophrenia: a decade in review. Psychopharmacology (Berl) 156:117–154
Graham FK (1975) The more or less startling effects of weak prestimulation. Psychophysiology 12:238–248
Hamada M, Hendrick JP, Ryan GR, Kuroiwa M, Higashi H, Tanaka M, Nairn AC, Greengard P, Nishi A (2005) Nicotine regulates DARPP-32 (dopamine- and cAMP-regulated phosphoprotein of 32 kDa) phosphorylation at multiple sites in neostriatal neurons. J Pharmacol Exp Ther 315:872–878
Hohnadel E, Bouchard K, Terry AV Jr (2007) Galantamine and donepezil attenuate pharmacologically induced deficits in prepulse inhibition in rats. Neuropharmacology 52:542–551
Jones IW, Bolam JP, Wonnacott S (2001) Presynaptic localization of the nicotinic acetylcholine receptor beta2 subunit immunoreactivity in rat nigrostiatal dopaminergic neurons. J Comp Neurol 439:235–247
Jones CK, Eberle EL, Shaw DB, McKinzie DL, Shannon HE (2005) Pharmacologic interactions between the muscarinic cholinergic and dopaminergic systems in the modulation of prepulse inhibition in rats. J Pharmacol Exp Ther 312:1055–1063
Powchik P, Davidson M, Haroutunian V, Gabriel SM, Purohit DP, Perl DP, Harvey PD, Davis KL (1998) Postmortem studies in schizophrenia. Schizophr Bull 24:325–341
Rosse RB, Deutsch SI (2002) Adjuvant galantamine administration improves negative symptoms in a patient with treatment-refractory schizophrenia. Clin Neuropharmacol 25:272–275
Sakaue M, Ago Y, Murakami C, Sowa C, Sakamoto Y, Koyama Y, Baba A, Matsuda T (2001) Involvement of benzodiazepine binding sites in an antiaggressive effect by 5-HT1A receptor activation in isolated mice. Eur J Pharmacol 432:163–166
Sakaue M, Ago Y, Baba A, Matsuda T (2003) The 5-HT1A receptor agonist MKC-242 reverse isolation rearing-induced deficits of prepulse inhibition in mice. Psychopharmacology (Berl) 170:73–79
Samochocki M, Höffle A, Fehrenbacher A, Jostock R, Ludwig J, Christner C, Radina M, Zerlin M, Ullmer C, Pereira EFR, Lübbert H, Albuquerque EX, Maelicke A (2003) Galantamine is an allosterically potentiating ligand of neuronal nicotinic but not of muscarinic acetylcholine receptors. J Pharmacol Exp Ther 305:1024–1036
Schilström B, Ivanov VB, Wiker C, Svensson TH (2007) Galantamine enhances dopaminergic neurotransmission in vivo via allosteric potentiation of nicotinic acetylcholine receptors. Neuropsychopharmacology 32:43–53
Stanhope KJ, Mirza NR, Bickerdike MJ, Bright JL, Harrington NR, Hesselink MB, Kennett GA, Lightowler S, Sherdown MJ, Syed R, Upton RL, Wadsworth G, Weiss SM, Wyatt A (2001) The muscarinic receptor agonist xanomeline has an antipsychotic-like profile in the rat. J Pharmacol Exp Ther 299:782–792
Suemaru K, Yasuda K, Umeda K, Araki H, Shibata K, Choshi T, Hibino S, Gomita Y (2004) Nicotine blocks apomorphine-induced disruption of prepulse inhibition of the acoustic startle in rats: possible involvement of central nicotinic α7 receptors. Br J Pharmacol 142:843–850
Swerdlow NR, Light GA, Cadenhead KS, Sprock J, Hsieh MH, Braff DL (2006) Startle gating deficits in a large cohort of patients with schizophrenia: relationship to medications, symptoms, neurocognition, and level of function. Arch Gen Psychiatry 63:1325–1335
Wang D, Noda Y, Zhou Y, Mouri A, Mizoguchi H, Nitta A, Chen W, Nabeshima T (2007) The allosteric potentiation of nicotinic acetylcholine receptors by galantamine ameliorates the cognitive dysfunction in beta amyloid(25-35) i.c.v.-injected mice: involvement of dopaminergic systems. Neuropsychopharmacology 32:1261–1271
Zhou F-M, Liang Y, Dani JA (2001) Endogenous nicotinic cholinergic activity regulates dopamine release in the striatum. Nature Neurosci 4:1224–1229
Acknowledgments
This study was supported in part by grants from the Ministry of Education, Science, Sports and Culture of Japan, and from Janssen Pharmaceutical K.K.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koda, K., Ago, Y., Kawasaki, T. et al. Galantamine and donepezil differently affect isolation rearing-induced deficits of prepulse inhibition in mice. Psychopharmacology 196, 293–301 (2008). https://doi.org/10.1007/s00213-007-0962-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-007-0962-1