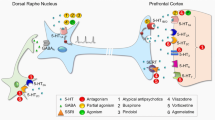
Abstract
Rationale
Fas-associated death domain (FADD) is an adaptor of death receptors that can also induce anti-apoptotic actions through its phosphorylated form (p-FADD). Activation of monoamine receptors, indirect targets of classic anti-depressant drugs (ADs), reduced FADD and increased p-FADD and p-FADD/FADD ratio in brain.
Objectives
To ascertain whether ADs, which indirectly regulate monoamine receptors, modulate FADD protein forms to promote anti-apoptotic actions.
Methods
The effects of selected norepinephrine transporter (NET), serotonin transporter (SERT), monoamine oxidase (MAO) inhibitors, atypical ADs, and electroconvulsive shock (ECS) or behavioral procedures (forced swim test, FST) on FADD forms and pro-survival FADD-like interleukin-1β-converting enzyme-inhibitory protein (FLIP-L) and phosphoprotein enriched in astrocytes of 15 kDa (p-PEA-15) contents were assessed in rat brain cortex by western blot analysis.
Results
Acute NET (e.g., nisoxetine) but not SERT (e.g., fluoxetine) inhibitors decreased cortical FADD (up to 37 %) and increased p-FADD/FADD ratio (up to 1.9-fold). Nisoxetine effects were prevented by α2-antagonist RX-821002, suggesting the involvement of presynaptic α2-autoreceptors. Immobility time in the FST correlated with increases of pro-apoptotic FADD and decreases of anti-apoptotic p-FADD. The MAO-A/B inhibitor phenelzine decreased FADD (up to 33 %) and increased p-FADD (up to 65 %) and p-FADD/FADD (up to 2.4-fold). Other MAO inhibitors (clorgyline, Ro 41-1049, rasagiline), atypical ADs (ketamine and mirtazapine), or ECS did not modulate cortical FADD. Chronic (14 days) desipramine and fluoxetine, but not phenelzine, increased p-FADD (up to 59 %), p-FADD/FADD ratio (up to 1.8-fold), and pro-survival p-PEA-15 (up to 46 %) in rat brain cortex.
Conclusions
Multifunctional FADD protein, through an increased p-FADD/FADD ratio, could participate in the mechanisms of anti-apoptotic actions induced by ADs.








Similar content being viewed by others
References
Alappat EC, Feig C, Boyerinas B, Vokland J, Samuels M, Murmann AE et al (2005) Phosphorylation of FADD at serine 194 by CKIalpha regulates its nonapoptotic activities. Mol Cell 19:321–332
Al-Nuaimi SK, Mackenzie EM, Baker GB (2012) Monoamine oxidase inhibitors and neuroprotection: a review. Am J Ther 19:436–448
Álvaro-Bartolomé M, García-Sevilla JA (2015) The neuroplastic index p-FADD/FADD and phosphoprotein PEA-15, interacting at GABAA receptor, are upregulated in brain cortex during midazolam-induced hypnosis in mice. Eur Neuropsychopharmacol 25:2131–2144
Álvaro-Bartolomé M, La Harpe R, Callado LF, Meana JJ, García-Sevilla JA (2011) Molecular adaptations of apoptotic pathways and signaling partners in the cerebral cortex of human cocaine addicts and cocaine-treated rats. Neuroscience 196:1–15
Antypa N, Calati R, Serretti A (2014) The neuropsychological hypothesis of antidepressant drug action revisited. CNS Neurol Disord Drug Targ 13:1722–1739
Artigas F (2015) Developments in the field of antidepressants, where do we go now? Eur Neuropsychopharmacol 25:657–670
Baghai TC, Blier P, Baldwin DS, Bauer M, Goodwin GM, Fountoulakis KN et al (2011) General and comparative efficacy and effectiveness of antidepressants in the acute treatment of depressive disorders: a report by the WPA section of pharmacopsychiatry. Eur Arch Psychiatry Clin Neurosci 261:207–245
Baker GB, Wong JT, Yeung JM, Coutts RT (1991) Effects of the antidepressant phenelzine on brain levels of gamma-aminobutyric acid (GABA). J Affect Disord 21:207–211
Belmaker RH, Agam G (2008) Major depressive disorder. N Engl J Med 358:55–68
Bhojani MS, Chen G, Ross BD, Beer DG, Rehemtulla A (2005) Nuclear localized phosphorylated FADD induces cell proliferation and is associated with aggressive lung cancer. Cell Cycle 4:1478–1481
Bortolozzi A, Castañé A, Semakova J, Santana N, Alvarado G, Cortés R et al (2012) Selective siRNA-mediated suppression of 5-HT1A autoreceptors evokes strong anti-depressant-like effects. Mol Psychiatry 17:612–623
Callado LF, Meana JJ, Grijalba B, Pazos A, Sastre M, García-Sevilla JA (1998) Selective increase of α2A-adrenoceptor agonist binding sites in brains of depressed suicide victims. J Neurochem 70:1114–1123
Carradori S, Silvestri R (2015) New frontiers in selective human MAO-B inhibitors. J Med Chem 58:6717–6732
Cottingham C, Wang Q (2012) α2-Adrenergic receptor dysregulation in depressive disorders: implications for the neurobiology of depression and antidepressant therapy. Neurosci Biobehav Rev 36:2214–2225
Cottingham C, Jones A, Wang Q (2012) Desipramine selectively potentiates norepinephrine-elicited ERK1/2 activation through the α2-adrenergic receptor. Biochem Biophys Res Comm 420:161–165
Detke MJ, Lucki I (1996) Detection of serotonergic and noradrenergic antidepressants in the rat forced swimming test: the effects of water depth. Behav Brain Res 73:43–46
Dhanasekaran DN, Reddy EP (2008) JNK signaling in apoptosis. Oncogene 27:6245–6251
Drzyzga ŁR, Marcinowska A, Obuchowicz E (2009) Antiapoptotic and neurotrophic effects of antidepressants: a review of clinical and experimental studies. Brain Res Bull 79:248–257
Dutta A, McKie S, Deakin JF (2015) Ketamine and other potential glutamate antidepressants. Psychiatry Res 225:1–13
Esteban S, Lladó J, Sastre-Coll A, García-Sevilla JA (1999) Activation and desensitization by cyclic antidepressant drugs of α2-autoreceptors, α2-heteroreceptors and 5-HT1A-autoreceptors regulating monoamine synthesis in rat brain in vivo. Naunyn-Schmiedeberg's Arch Pharmacol 360:135–143
Finberg JPM (2014) Update on the pharmacology of selective inhibitors of MAO-A and MAO-B: focus on modulation of CNS monoamine neurotransmitter release. Pharmacol Ther 143:133–152
Fink KB, Göthert M (2007) 5-HT receptor regulation of neurotransmitter release. Pharmacol Rev 59:360–417
Flint J, Kendler KS (2014) The genetics of major depression. Neuron 81:484–503
Galluzzi L, Blomgren K, Kroemer G (2009) Mitochondrial membrane permeabilization in neuronal injury. Nature Rev Neurosci 10:481–494
García-Fuster MJ, García-Sevilla JA (2015) Monoamine receptor agonists, acting preferentially at presynaptic autoreceptors and heteroreceptors, downregulate the cell fate adaptor FADD in rat brain cortex. Neuropharmacology 89:204–214
García-Fuster MJ, Miralles A, García-Sevilla JA (2007) Effects of opiate drugs on Fas-associated protein with death domain (FADD) and effector caspases in the rat brain: regulation by the ERK1/2 MAP kinase pathway. Neuropsychopharmacology 32:399–411
García-Fuster MJ, Ramos-Miguel A, Rivero G, La Harpe R, Meana JJ, García-Sevilla JA (2008a) Regulation of the extrinsic and intrinsic apoptotic pathways in the prefrontal cortex of short- and long-term human opiate abusers. Neuroscience 157:105–119
García-Fuster MJ, Ramos-Miguel A, Miralles A, García-Sevilla JA (2008b) Opioid receptor agonists enhance the phosphorylation state of Fas-associated death domain (FADD) protein in the rat brain: functional interactions with casein kinase Ialpha, Galphai proteins, and ERK1/2 signaling. Neuropharmacology 55:886–8991
García-Fuster MJ, Parks GS, Clinton SM, Watson SJ, Akil H, Civelli O (2012) The melanin-concentrating hormone (MCH) system in an animal model of depression-like behavior. Eur Neuropsychopharmacol 22:607–613
García-Fuster MJ, Díez-Alarcia R, Ferrer-Alcón M, La Harpe R, Meana JJ, García-Sevilla JA (2014) FADD adaptor and PEA-15/ERK1/2 partners in major depression and schizophrenia postmortem brains: basal contents and effects of psychotropic treatments. Neuroscience 277:541–551
García-Sevilla JA, Ventayol P, Pérez V, Rubovszky G, Puigdemont D, Ferrer-Alcón M et al (2004) Regulation of platelet α2A-adrenoceptors, Gi proteins and receptor kinases in major depression: effects of mirtazapine treatment. Neuropsychopharmacology 29:580–588
González MM, Aston-Jones G (2008) Light deprivation damages monoamine neurons and produces a depressive behavioral phenotype in rats. Proc Natl Acad Sci U S A 105:4898–4903
González-Maeso J, Rodríguez-Puertas R, Meana JJ, García-Sevilla JA, Guimón J (2002) Neurotransmitter receptor-mediated activation of G-proteins in brains of suicide victims with mood disorders: selective supersensitivity of α2A-adrenoceptors. Mol Psychiatry 7:755–767
Greig FH, Nixon GF (2014) Phosphoprotein enriched in astrocytes (PEA)-15: a potential therapeutic target in multiple disease states. Pharmacol Ther 143:265–274
Grieve SM, Korgaonkar MS, Koslow SH, Gordon E, Williams LM (2013) Widespread reductions in gray matter volume in depression. Neuroimage Clin 3:332–339
Harlan J, Chen Y, Gubbins E, Mueller R, Roch JM, Walter K et al (2006) Variants in Apaf-1 segregating with major depression promote apoptosome function. Mol Psychiatry 11:76–85
Hayasaka Y, Purgato M, Magni LR, Ogawa Y, Takeshima N, Cipriani A et al (2015) Dose equivalents of antidepressants: evidence-based recommendations from randomized controlled trials. J Affect Disord 180:179–184
Hieronymus F, Emilsson JF, Nilsson S, Eriksson E (2016) Consistent superiority of selective serotonin reuptake inhibitors over placebo in reducing depressed mood in patients with major depression. Mol Psychiatry 21:523–530
Hjorth S, Tao R (1991) The putative 5-HT1B receptor agonist CP-93,129 suppresses rat hippocampal 5-HT release in vivo: comparison with RU 24969. Eur J Pharmacol 209:249–252
Hwang EY, Jeong MS, Park SY, Jang SB (2014) Evidence of complex formation between FADD and c-FLIP death effector domains for the death inducing signaling complex. BMB Rep 47:488–493
Jama A, Cecchi M, Calvo N, Watson SJ, Akil H (2008) Inter-individual differences in novelty-seeking behavior in rats predict differential responses to desipramine in the forced swim test. Psychopharmacology (Berl) 198:333–340
Jantas D, Krawczyk S, Lason W (2014) The predominant protective effect of tianeptine over other antidepressants in models of neuronal apoptosis: the effect blocked by inhibitors of MAPK/ERK1/2 and PI3-K/Akt pathways. Neurotox Res 25:208–225
Jin Y, Lim CM, Kim SW, Park JY, Seo JS, Han PL et al (2009) Fluoxetine attenuates kainic acid-induced neuronal cell death in the mouse hippocampus. Brain Res 1281:108–116
Kataoka T (2005) The caspase-8 modulator c-FLIP. Crit Rev Immunol 25:31–58
Kristensen AS, Andersen J, Jørgensen TN, Sørensen L, Eriksen J, Loland CJ et al (2011) SLC6 neurotransmitter transporters: structure, function, and regulation. Pharmacol Rev 63:585–640
Kupfer DJ, Frank E, Phillips ML (2012) Major depressive disorder: new clinical, neurobiological, and treatment perspectives. Lancet 379:1045–1055
Langer SZ (2015) α2-Adrenoceptors in the treatment of major neuropsychiatric disorders. Trends Pharmacol Sci 36:196–202
Lillethorup TP, Iversen P, Fontain J, Wegener G, Doudet DJ, Landau AM (2015) Electroconvulsive shocks decrease α2-adrenoceptor binding in the Flinders rat model of depression. Eur Neuropsychopharmacol 25:404–412
Lucassen PJ, Fuchs E, Czéh B (2004) Antidepressant treatment with tianeptine reduces apoptosis in the hippocampal dentate gyrus and temporal cortex. Biol Psychiatry 55:789–796
McKernan DP, Dinan TG, Cryan JF (2009) “Killing the blues”: a role for cellular suicide (apoptosis) in depression and the antidepressant response? Prog Neurobiol 88:246–263
McManus DJ, Baker GB, Martin IL, Greenshaw AJ, McKenna KF (1992) Effects of the antidepressant/antipanic drug phenelzine on GABA concentrations and GABA-transaminase activity in rat brain. Biochem Pharmacol 43:2486–2489
Meana JJ, Barturen F, García-Sevilla JA (1992) Alpha 2-adrenoceptors in the brain of suicide victims: increased receptor density associated with major depression. Biol Psychiatry 31:471–490
Menargues A, Obach R, García-Sevilla JA (1990) Modulation by antidepressant drugs of CNS postsynaptic α2-adrenoceptors mediating mydriasis in the rat. Naunyn Schmiedeberg's Arch Pharmacol 341:101–107
Meyer JH, Ginovart N, Boovariwala A, Sagrati S, Hussey D, Garcia A et al (2006) Elevated monoamine oxidase a levels in the brain: an explanation for the monoamine imbalance of major depression. Arch Gen Psychiatry 63:1209–1216
Miguel-Hidalgo JJ, Whittom A, Villarreal A, Soni M, Meshram A, Pickett JC et al (2014) Apoptosis-related proteins and proliferation markers in the orbitofrontal cortex in major depressive disorder. J Affect Disord 158:62–70
Nutt DJ (2002) The neuropharmacology of serotonin and noradrenaline in depression. Int Clin Psycopharmacol 17, Suppl. 1, S1–S12
O’Donnell JM, Shelton RC (2011) Drug therapy of depression and anxiety disorders. In Goodman and Gilman’s The Pharmacological Basis of Therapeutics, 12th edition (Editor, L.L. Brunton). New York, McGraw Hill Medical. Chapter 15:397–415
Popov N, Matthies H (1969) Some effects of monoamine oxidase inhibitors on the metabolism of gamma-aminobutyric acid in rat brain. J Neurochem 16:899–907
Rajkowska G, Miguel-Hidalgo JJ, Wei J, Dilley G, Pittman SD, Meltzer HY et al (1999) Morphometric evidence for neuronal and glial prefrontal cell pathology in major depression. Biol Psychiatry 45:1085–1098
Ramos-Miguel A, García-Fuster MJ, Callado LF, La Harpe R, Meana JJ, García-Sevilla JA (2009) Phosphorylation of FADD at serine 194 is increased in the prefrontal cortex of opiate abusers: relation to mitogen activated protein kinase, phosphoprotein enriched in astrocytes of 15 kDa, and Akt signaling pathways involved in neuroplasticity. Neuroscience 161:23–38
Ramos-Miguel A, Álvaro-Bartolomé M, García-Fuster MJ, García-Sevilla JA (2012) Role of multifunctional FADD (Fas-associated death domain) adaptor in drug addiction (Chapter 7). In: Addictions—From Pathophysiology to Treatment. In InTech Open. Edited by David Belin. ISBN: 978-953-51-0783-5
Renganathan H, Vaidyanathan H, Knapinska A, Ramos JW (2005) Phosphorylation of PEA-15 switches its binding specificity from ERK/MAPK to FADD. Biochem J 390:729–735
Rivero G, Gabilondo AM, García-Sevilla JA, La Harpe R, Callado LF, Meana JJ (2014) Increased α2- and β1-adrenoceptor densities in postmortem brain of subjects with depression: differential effect of antidepressant treatment. J Affect Disord 167:343–350
Screaton RA, Kiessling S, Sansom OJ, Millar CB, Maddison K, Bird A et al (2003) Fas-associated death domain protein interacts with methyl-CpG binding domain protein 4: a potential link between genome surveillance and apoptosis. Proc Natl Acad Sci U S A 100:5211–5216
Shih JC, Chen K, Ridd MJ (1999) Monoamine oxidase: from genes to behavior. Annu Rev Neurosci 22:197–217
Sibille E, French B (2013) Biological substrates underpinning diagnosis of major depression. Int J Neuropsychopharmacol 16:1893–1909
Starke K (2001) Presynaptic autoreceptors in the third decade: focus on α2-adrenoceptors. J Neurochem 78:685–693
Stewart JW (2007) Treating depression with atypical features. J Clin Psychiatry 68:25–29
Sulzmaier F, Opoku-Ansah J, Ramos JW (2012) Phosphorylation is the switch that turns PEA-15 from tumor suppressor to tumor promoter. Small GTPases 3:173–177
Tanner DC, Campbell A, O'Banion KM, Noble M, Mayer-Pröschel M (2015) cFLIP is critical for oligodendrocyte protection from inflammation. Cell Death Differ 22:1489–1501
Taoufik E, Valable S, Müller GJ, Roberts ML, Divoux D, Tinel A et al (2007) FLIPL protects neurons against in vivo ischemia and in vitro glucose deprivation-induced cell death. J Neurosci 27:6633–6646
Thomas DN, Nutt DJ, Holman RB (1992) Effects of acute and chronic electroconvulsive shock on noradrenaline release in the rat hippocampus and frontal cortex. Br J Pharmacol 106:430–434
Trencia A, Perfetti A, Cassese A, Vigliotta G, Miele C, Oriente F et al (2003) Protein kinase B/Akt binds and phosphorylates PED/PEA-15, stabilizing its antiapoptotic action. Mol Cell Biol 23:4511–4521
Urigüen L, Arteta D, Díez-Alarcia R, Ferrer-Alcón M, Díaz A, Pazos A et al (2008) Gene expression patterns in brain cortex of three different animal models of depression. Genes Brain Behav 7:649–658
Van Waes V, Ehrlich S, Beverley JA, Steiner H (2015) Fluoxetine potentiation of methylphenidate-induced gene regulation in striatal output pathways: potential role for 5-HT1B receptor. Neuropharmacology 89:77–86
Won E, Ham B-J (2016) Imaging genetics studies on monoaminergic genes in major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry 64:311–319
Yang D, Chen M, Russo-Neustadt A (2012) Antidepressants are neuroprotective against nutrient deprivation stress in rat hippocampal neurons. Eur J Neurosci 36:2573–2587
Zhao Y-J, Du M-Y, Huang X-Q, Lui S, Chen Z-Q, Liu J et al (2014) Brain grey matter abnormalities in medication-free patients with major depressive disorder: a meta-analysis. Psychol Med 44:2927–2937
Acknowledgments
This research was funded by Grants SAF2011-29918 (to J.A.G.-S.) and SAF2014-55903-R (to M.J.G.-F.) from Ministerio de Economía y Competitividad (MINECO, Madrid, Spain) and Fondo Europeo de Desarrollo Regional (FEDER). The study was supported in part by Project 2012/011 from Delegación del Gobierno para el Plan Nacional sobre Drogas (Ministerio de Sanidad, Servicios Sociales e Igualdad, Madrid) and by Fundación Alicia Koplowitz (Madrid) both to M. J. G.-F. The authors would like to thank Antonio Crespo for the skillful technical assistance. M. J. G.-F. is a Ramón y Cajal researcher (MINECO-UIB). J. A. G.-S. is a member of the Institut d’Estudis Catalans (Barcelona, Catalonia, Spain).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiments were carried out in accordance with standard ethical guidelines (European Communities Council Directive 86/609/EEC and Guidelines for the Care and Use of Mammals in Neuroscience and Behavioral Research, National Research Council 2003) and approved by the Local Bioethical Committee (UIB). All efforts were made to minimize the number of rats used and their suffering.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
García-Fuster, M.J., García-Sevilla, J.A. Effects of anti-depressant treatments on FADD and p-FADD protein in rat brain cortex: enhanced anti-apoptotic p-FADD/FADD ratio after chronic desipramine and fluoxetine administration. Psychopharmacology 233, 2955–2971 (2016). https://doi.org/10.1007/s00213-016-4342-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4342-6