Abstract
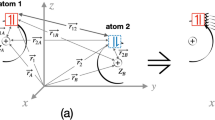
A recent paper (Phys. Chem. Chem. Phys. 2020, 22:22,459) shows that the energy components of the extended transition state energy decomposition analysis (ETS-EDA) are path functions, and therefore, they are not uniquely defined. In this work, we apply the ETS-EDA to analyse all possible dissociation paths of the water tetramer to four free water molecules. Our results confirm that the energy components of the ETS-EDA are path functions. However, they also show that differences among energy components obtained for the different paths are relatively small, and therefore, we conclude that the information obtained from an ETS-EDA can be used to discuss the nature of chemical bonds and analyse the origin of isomerization energies and energy barriers. However, if a given process can be attained by means of different and chemically reasonable paths, we recommend to perform the ETS-EDA of a given reaction for all different paths to confirm that energy components of the ETS-EDA do not differ very much from one path to another.



Similar content being viewed by others
5. References
Vaughan AL (1931) Mass spectrograph analyses, and critical potentials for the production of ions by electron impact, in nitrogen and carbon monoxide. Phys Rev 38:1687–1695
Mathur D (2004) Structure and dynamics of molecules in high charge states. Phys Rep 391:1–118
Fletcher JD, Parkes MA, Price SD (2015) Bond-forming reactions of N22+ with C2H4, C2H6, C3H4 and C3H6. Int J Mass Spectrom 377:101–108
Phipps MJS, Fox T, Tautermann CS, Skylaris C-K (2015) Energy decomposition analysis approaches and their evaluation on prototypical protein–drug interaction patterns. Chem Soc Rev 44:3177–3211
Szalewicz K, Jeziorski B (1979) Symmetry-adapted double-perturbation analysis of intramolecular correlation effects in weak intermolecular interactions. Mol Phys 38:191–208
Jeziorski B, Moszynski R, Szalewicz K (1994) Perturbation theory approach to intermolecular potential energy surfaces of van der Waals complexes. Chem Rev 94:1887–1930
Kitaura K, Morokuma K (1976) A new energy decomposition scheme for molecular interactions within the Hartree-Fock approximation. Int J Quantum Chem 10:325–340
Morokuma K (1977) Why do molecules interact? The origin of electron donor-acceptor complexes, hydrogen bonding and proton affinity. Acc Chem Res 10:294–300
Ziegler T, Rauk A (1977) On the calculation of bonding energies by the Hartree Fock slater method. Theor Chim Acta 46:1–10
Ziegler T, Rauk A (1979) A theoretical study of the ethylene-metal bond in complexes between copper(1+), silver(1+), gold(1+), platinum(0) or platinum(2+) and ethylene, based on the Hartree-Fock-Slater transition-state method. Inorg Chem 18:1558–1565
Bickelhaupt FM, Baerends EJ. Rev. Comput. Chem. In: Lipkowitz KB, Boyd DB, eds. Vol. 15. New York: Wiley-VCH; 2000, 1–86.
Frenking G, Bickelhaupt FM (2014) The EDA perspective of chemical bonding. In: Frenking G, Shaik S (eds) The chemical bond. Wiley, Weinheim, pp 121–157
Bickelhaupt FM, Houk KN (2017) Analyzing reaction rates with the distortion/interaction-activation strain model. Angew Chem Int Ed 56:10070–10086
Fernández I (2018) Understanding the reactivity of fullerenes through the activation strain model. Eur J Org Chem 2018:1394–1402
Zhao L, von Hopffgarten M, Andrada DM, Frenking G (2018) Energy decomposition analysis. WIREs Comput Mol Sci 8:e1345
Andrés J, Ayers PW, Boto RA, Carbó-Dorca R, Chermette H, Cioslowski J, Contreras-García J, Cooper DL, Frenking G, Gatti C et al (2019) Nine questions on energy decomposition analysis. J Comput Chem 40:2248–2283
Andrada DM, Foroutan-Nejad C (2020) Energy components in energy decomposition analysis (EDA) are path functions; why does it matter? Phys Chem Chem Phys 22:22459–22464
Pérez JF, Hadad CZ, Restrepo A (2008) Structural studies of the water tetramer. Int J Quantum Chem 108:1653–1659
Vítek A, Kalus R, Paidarová I (2010) Structural changes in the water tetramer. A combined Monte Carlo and DFT study. Phys Chem Chem Phys 12:13657–13666
Ceponkus J, Uvdal P, Nelander B (2012) Water tetramer, pentamer, and hexamer in inert matrices. J Phys Chem A 116:4842–4850
Cruzan JD, Braly LB, Liu K, Brown MG, Loeser JG, Saykally RJ (1996) Quantifying hydrogen bond cooperativity in water: VRT spectroscopy of the water tetramer. Science 271:59–62
Baerends EJ, Autschbach J, Bérces A, Bickelhaupt FM, Bo C, de Boeij PL, Boerrigter PM, Cavallo L, Chong DP, Deng L et al (2017) ADF2017.01, SCM, Vrje Universiteit Amsterdam, Amsterdam, The Netherlands. http://www.scm.com
te Velde G, Bickelhaupt FM, Baerends EJ, Fonseca Guerra C, van Gisbergen SJA, Snijders JG, Ziegler T (2001) Chemistry with ADF. J Comput Chem 22:931–967
Becke AD (1988) Density-functional exchange-energy approximation with correct asymptotic-behavior. Phys Rev A 38:3098–3100
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Grimme S, Antony J, Ehrlich S, Krieg H (2010) A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J Chem Phys 132:154104
Grimme S, Ehrlich S, Goerigk L (2011) Effect of the damping function in dispersion corrected density functional theory. J Comput Chem 32:1456–1465
Becke AD, Johnson ER (2007) Exchange-hole dipole moment and the dispersion interaction revisited. J Chem Phys 127:154108
Van Lenthe E, Baerends EJ (2003) Optimized slater-type basis sets for the elements 1–118. J Comput Chem 24:1142–1156
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566
Solà M, Lledós A, Duran M, Bertrán J, Ventura ON (1990) Ab initio study of substituent effect on the addition of hydrogen fluoride to fluoroethylenes. J Comput Chem 11:170–180
Glendening ED, Streitwieser A (1994) Natural energy decomposition analysis: an energy partitioning procedure for molecular interactions with application to weak hydrogen bonding, strong ionic, and moderate donor–acceptor interactions. J Chem Phys 100:2900–2909
Poater J, Fradera X, Solà M, Duran M, Simon S (2003) On the electron-pair nature of the hydrogen bond in the framework of the atoms in molecules theory. Chem Phys Lett 369:248–255
Song H-J, Xiao H-M, Dong H-S (2006) Cooperative effects, strengths of hydrogen bonds, and intermolecular interactions in circular cis, trans-cyclotriazane clusters (n = 3–8). J Chem Phys 125:074308
Guevara-Vela JM, Chávez-Calvillo R, García-Revilla M, Hernández-Trujillo J, Christiansen O, Francisco E, Martín Pendás Á, Rocha-Rinza T (2013) Hydrogen-bond cooperative effects in small cyclic water clusters as revealed by the interacting quantum atoms approach. Chem Eur J 19:14304–14315
Fonseca Guerra C, Zijlstra H, Paragi G, Bickelhaupt FM (2011) Telomere structure and stability: covalency in hydrogen bonds, not resonance assistance, causes cooperativity in guanine quartets. Chem Eur J 17:12612–12622
Nochebuena J, Cuautli C, Ireta J (2017) Origin of cooperativity in hydrogen bonding. Phys Chem Chem Phys 19:15256–15263
Wolters LP, Smits NWG, Guerra CF (2015) Covalency in resonance-assisted halogen bonds demonstrated with cooperativity in N-halo-guanine quartets. Phys Chem Chem Phys 17:1585–1592
Paragi G, Fonseca GC (2017) Cooperativity in the self-assembly of the guanine nucleobase into quartet and ribbon structures on surfaces. Chem Eur J 23:3042–3050
Bergmann ED (1971) Aromaticity, pseudo-aromaticity and anti-aromaticity. In: Bergmann ED, Pullman B (eds) Proceedings of the international symposium, Jerusalem, 1970. The Israel Academy of Science and Humanities, Jerusalem, p 392
El-Hamdi M, Tiznado W, Poater J, Solà M (2011) An analysis of the isomerization energies of 1,2-/1,3-Diazacyclobutadiene, Pyrazole/Imidazole, and Pyridazine/Pyrimidine with the turn-upside-down approach. J Org Chem 76:8913–8921
El Bakouri O, Solà M, Poater J (2016) Planar versus three-dimensional X62−, X2Y42−, and X3Y32− (X, Y = B, Al, Ga) metal clusters: an analysis of their relative energies through the turn-upside-down approach. Phys Chem Chem Phys 18:21102–21110; Erratum, íbid (2018) 20:3845–3846
Wolters LP, Bickelhaupt FM (2015) The activation strain model and molecular orbital theory. WIREs Comput Mol Sci 5:324–343
Fernández I, Bickelhaupt FM (2014) The activation strain model and molecular orbital theory: understanding and designing chemical reactions. Chem Soc Rev 43:4953–4967
Vermeeren P, van der Lubbe SCC, Fonseca Guerra C, Bickelhaupt FM, Hamlin TA (2020) Understanding chemical reactivity using the activation strain model. Nat Protoc 15:649–667
Acknowledgements
This work has been supported by the Ministerio de Economía y Competitividad (MINECO) of Spain (Projects CTQ2017-85341-P, PID2019-106830GB-I00, and MDM-2017-0767) and the Generalitat de Catalunya (projects 2017SGR39 and 2017SGR348). Excellent service by the Supercomputer center of the Consorci de Serveis Universitaris de Catalunya (CSUC) is gratefully acknowledged. This work is dedicated to Prof. Ramon Carbó-Dorca as a proof of our admiration for his brilliant contributions to chemistry.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published as part of the special collection of articles "Festschrift in honour of Prof. Ramon Carbó-Dorca".
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Solà, M., Duran, M. & Poater, J. The energy components of the extended transition state energy decomposition analysis are path functions: the case of water tetramer. Theor Chem Acc 140, 33 (2021). https://doi.org/10.1007/s00214-021-02730-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-021-02730-3