Abstract
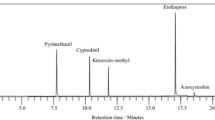
An isocratic LC method for the determination of melamine and its degradation products (ammelide, ammeline, and cyanuric acid), used to increase the apparent protein content of rice protein concentrate, has been developed. Method development involved optimization of different RP columns, aqueous mobile phases, pH, phosphate concentration, and temperature. The optimum separation of these compounds was achieved using a Luna CN column (30 °C), 5 mmol L−1 sodium phosphate (pH 5.0) as mobile phase, 1 mL min−1 flow-rate, UV absorbance-DAD detection at 220 nm, and resorcine as internal standard; this enabled separation of these compounds with baseline resolution (values in the 2.1–10.1 range) in about 8 min. Prior to HPLC, the developed sample preparation procedure consisted in a leaching process using the above mentioned mobile phase. Method validation was carried out in rice protein concentrates in accordance with the European Commission decision 2002/657/EC criteria. For this purpose, eight mandatory performance characteristics for the conventional validation approach were determined: calibration graphs, extraction efficiencies, decision limits, detection capabilities, precision (repeatability and within-laboratory reproducibility), accuracy, selectivity, and robustness. The extraction efficiencies for these compounds were in the range 99–100% and the within-laboratory reproducibility at 1.0, 1.5, and 2.0 detection capabilities concentration levels were smaller than 5, 4, and 3%, respectively. Finally, the proposed method was successfully applied to the analysis of other rice protein concentrates and several animal feed samples.





Similar content being viewed by others
References
European Food Safety Authority (EFSA). www.efsa.europa.eu. Question N° EFSA-Q-2007–093
Pichon V, Chen L, Guenu S, Hennion MC (1995) J Chromatogr A 711:257–267
Ehling S, Tefera S, Ho IP (2007) Food Addit Contam 24:1319–1325
FDA (2007) Interim melamine and analogues safety/risk assessment, May 25
National Toxicology Program. TR-245 Carcinogenesis bioassay of melamine (CAS No. 108–78–1) in F344/N Rats and B6C3F1 mice (feed study). http://ntp.niehs.nih.gov/go/12025
Van der Lee MK, van der Weg G, Tragg WA, Mol HGJ (2008) J Chromatogr A 1186:325–339
Marwah A, Marwah P, Lardy H (2005) J Chromatogr B 824:107–115
Ono S, Funato T, Inoue Y, Munechika T, Yoshimura T, Morita H, Rengakuji S, Shimasaki C (1998) J Chromatogr A 815:197–204
Sugita T, Ishiwata H, Yoshihira K, Maekawa A (1990) Bull Environ Contam Toxicol 44:567–571
Sancho JV, Ibañez M, Grimalt S, Pozo OJ, Hernandez F (2005) Anal Chim Acta 530:237–243
FDA. GC-MS screen for the presence of melamine, ammeline, ammelide and cyanuric acid. Version 2.1. www.fda.gov/cvm/GCMSMelamine.htm
Downes CJ, Mitchell JW, Viotto ES, Eggers NJ (1984) Water Res 18:277–280
www.nutral.com. May 2008
Commission of EC. Council directive 2002/657/EC. Off J Eur Communities L 221 (2002) 8
ISO. Guide to the expression of uncertainty in measurement, 1993; EA-4/16. Guidelines on the expression of uncertainty in quantitative testing, Rev. 00. December 2003; NIST. Technical Note 1297. Guidelines for evaluating and expressing the uncertainty of NIST measurement results, September 1994
Logan BK (1994) Anal Chim Acta 288:111–122
Kim B, Perkins LB, Bushway RJ, Nesbit S, Fan T, Sheridan R, Greene V (2008) JAOAC 91:408–413
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muñiz-Valencia, R., Ceballos-Magaña, S.G., Rosales-Martinez, D. et al. Method development and validation for melamine and its derivatives in rice concentrates by liquid chromatography. Application to animal feed samples. Anal Bioanal Chem 392, 523–531 (2008). https://doi.org/10.1007/s00216-008-2294-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-008-2294-3