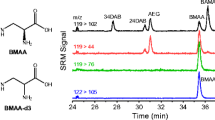
Abstract
The neurotoxin β-N-methylamino-l-alanine (BMAA) has been reported in cyanobacteria and shellfish, raising concerns about widespread human exposure. However, inconsistent results for BMAA analysis have led to controversy. Liquid chromatography–tandem mass spectrometry (LC–MS/MS) is the most appropriate method for analysis of BMAA, but the risk of interference from isomers, other sample components, and the electrospray background is still present. We have investigated differential mobility spectrometry (DMS) as an ion filter to improve selectivity in the hydrophilic interaction liquid chromatographic (HILIC)–MS/MS determination of BMAA. We obtained standards for two BMAA isomers not previously analyzed by HILIC–MS, β-amino-N-methylalanine and 3,4-diaminobutanoic acid, and the typically used 2,4-diaminobutanoic acid and N-(2-aminoethyl)glycine. DMS separation of BMAA from these isomers was achieved and optimized conditions were used to develop a sensitive and highly selective multidimensional HILIC–DMS–MS/MS method. This work revealed current technical limitations of DMS for trace quantitation, and practical solutions were implemented. Accurate control of low levels of DMS carrier gas modifier was essential, but required external metering. The linearity of our optimized method was excellent from 0.01 to 6 μmol L−1. The instrumental LOD was 0.4 pg BMAA injected on-column and the estimated method LOD was 20 ng g−1 dry weight for BMAA in sample matrix. The method was used to analyze cycad plant tissue, a cyanobacterial reference material, and mussel tissues, by use of isotope-dilution quantitation with deuterated BMAA. This confirmed the presence of BMAA and several of its isomers in cycad and mussel tissues, including commercially available mussel tissue reference materials certified for other biotoxins.

Differential Mobility Spectrometry is used to increases the selectivity of BMAA analysis by HILIC–MS/MS







Similar content being viewed by others
References
Vega A, Bell EA (1967) Phytochemistry 6:759–762
Bradley WG, Borenstein AR, Nelson LM, Codd GA, Rosen BH, Stommel EW, Cox PA (2013) Amyotroph Lateral Scler Frontotemporal Degener 14:325–333
Chiu AS, Gehringer MM, Welch JH, Neilan BA (2011) Int J Environ Res Public Health 8:3728–3746
Banack SA, Caller T, Henegan P, Haney J, Murby A, Metcalf JS, Powell J, Cox PA, Stommel E (2015) Toxins 7:322–336
Banack SA, Metcalf JS, Bradley WG, Cox PA (2014) Toxicon 90:167–173
Cox PA, Banack SA, Murch SJ (2003) Proc Natl Acad Sci U S A 100:13380–13383
Faassen EJ, Gillissen F, Lürling M (2012) PLoS One 7:e36667
Faassen EJ (2014) Toxins 6:1109–1138
Kruger T, Oelmuller R, Luckas BJ (2012) Endocytobiosis Cell Res 22:29–36
Jiang L, Aigret B, Borggraeve W, Spacil Z, Ilag L (2012) Anal Bioanal Chem 403:1719–1730
Salomonsson ML, Hansson A, Bondesson U (2013) Anal Methods 5:4865–4874
Jiang L, Kiselova N, Rosen J, Ilag LL (2014) Sci Rep 6931:1–7
McCarron P, Logan AC, Giddings SD, Quilliam MA (2014) Aquat Biosyst 10(5):1–7
Li AF, Fan H, Ma FF, McCarron P, Thomas K, Tang XH, Quilliam MA (2012) Analyst 137:1210–1219
Réveillon D, Abadie E, Séchet V, Brient L, Savar V, Bardouil M, Hess P, Amzil Z (2014) Mar Drugs 12:5441–5467
Combes A, El Abdellaoui S, Sarazin C, Vial J, Mejean A, Ploux O, Pichon V (2013) Anal Chim Acta 771:42–49
Marler TE, Snyder LR, Shaw CA (2010) Toxicon 56:563–568
Guevremont R, Purves RW, Barnett DA, Ells B (2006) FAIMS apparatus and method using carrier gases that contain a trace amount of a dopant species. US Patent no. 7,026,612
Shvartsburg AA (2009) Differential Ion Mobility Spectrometry. CRC Press, Boca Raton
Schneider BB, Nazarov EG, Londry F, Vouros P, Covey TR (2015) Mass Spectrom Rev in press doi:10.1002/mas.21453
Campbell JL, Zhu M, Hopkins WS (2014) J Am Soc Mass Spectrom 25:1583–1591
Kapron JT, Jemal M, Duncan G, Kolakowski B, Purves R (2005) Rapid Commun Mass Spectrom 19:1979–1983
Guevremont R, Kloakowski B (2005) Am Lab 37:11–14
Klaassen T, Szwandt S, Kapron JT, Roemer A (2009) Rapid Commun Mass Spectrom 23:2301–2306
Prasad S, Belford M, Dunyach J, Purves RJ (2014) Am Soc Mass Spectrom 25:2143–2153
Jin W, Jarvis M, Star-Weinstock M, Altemus M (2013) Anal Bioanal Chem 405:9497–9508
Ray JA, Kushnir MM, Yost RA, Rockwood AL, Meikle AW (2015) Performance enhancement in the measurement of 5 endogenous steroids in human serum by LC–MS/MS combined with differential ion mobility spectrometry. Clin Chim Acta 438:330–336
Purves RW, Ozog A, Ambrose SJ, Prasad S, Belford M, Dunyach J-J (2014) J Am Soc Mass Spectrom 25:1274–1284
Pang N, Yan C (2014) Int J Mass Spectrom 362:48–55
Beach DG, Melanson JE, Purves RW (2015) Anal Bioanal Chem 407:2473–2484
Burton IW, Quilliam MA, Walter JA (2005) Anal Chem 77:3123–3131
Hollingdale K, Thomas K, Lewis N, Békri K, McCarron P, Quilliam MA (2015) Anal Bioanal Chem 407:5353–5363
Beach DG, Gabryelski W (2012) J Am Soc Mass Spectrom 23:858–868
Kolakowski BM, McCooeye MA, Mester Z (2006) Rapid Commun Mass Spectrom 20:3319–3329
Schneider BB, Nazarov EG, Londry F, Kang Y, Covey TR (2014) Improved DMS Performance with a Jet Injector Inlet. 62nd ASMS Conference on Mass Spectrometry and Allied Topics, Baltimore, MD
Pagliano E, Mester Z, Meija J (2013) Anal Bioanal Chem 405:2879–2887
Acknowledgments
The authors thank Randy Purves, for input on modifications to enable the introduction of low levels of gas modifiers and for helpful discussions, Sabrina Giddings and Krista Thomas, for technical assistance, and Pearse McCarron for editorial input.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 315 kb)
Rights and permissions
About this article
Cite this article
Beach, D.G., Kerrin, E.S. & Quilliam, M.A. Selective quantitation of the neurotoxin BMAA by use of hydrophilic-interaction liquid chromatography–differential mobility spectrometry–tandem mass spectrometry (HILIC–DMS–MS/MS). Anal Bioanal Chem 407, 8397–8409 (2015). https://doi.org/10.1007/s00216-015-9012-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-9012-8