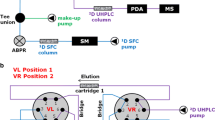
Abstract
In this study, a rapid and sensitive ultra-high performance supercritical fluid chromatography-mass spectrometry (UHPSFC-MS) method has been developed and partially validated for the separation of carotenoids within less than 6 min. Six columns of orthogonal selectivity were examined, and the best separation was obtained by using a 1-aminoanthracene (1-AA) column. The length of polyene chain as well as the number of hydroxyl groups in the structure of the studied carotenoids determines their differences in the physiochemical properties and thus the separation that is achieved on this column. All of the investigated carotenoids were baseline separated with resolution values greater than 1.5. The effects of gradient program, back pressure, and column temperature were studied with respect to chromatographic properties such as retention and selectivity. Electrospray ionization (ESI) and atmospheric pressure chemical ionization (APCI) were compared in both positive and negative mode, using both direct infusion and hyphenated with UHPSFC. The ESI in positive mode provided the highest response. The coefficient of determination (R 2) for all calibration curves were greater than 0.998. Limit of detection (LOD) was in the range of 2.6 and 25.2 ng/mL for α-carotene and astaxanthin, respectively, whereas limit of quantification (LOQ) was in the range of 7.8 and 58.0 ng/mL for α-carotene and astaxanthin, respectively. Repeatability and intermediate precision of the developed UHPSFC-MS method were determined and found to be RSD < 3 % and RSD < 6 %, respectively. The method was applied in order to determine carotenoids in supercritical fluid extracts of microalgae and rosehip.

Ultra-high performance supercritical fluid chromatography-a rapid separation method for the analysis of carotenoids in rosehip and microalgae samples







Similar content being viewed by others
References
Socaciu C. Food colorants : chemical and functional properties. Chemical and functional properties of food components series. Boca Raton, Fla. London: CRC Press; 2008.
Edge R, McGarvey DJ, Truscott TG. The carotenoids as anti-oxidants—a review. J Photochem Photobiol B. 1997;41(3):189–200.
Rao AV, Rao LG. Carotenoids and human health. Pharmacol Res. 2007;55(3):207–16.
Guedes AC, Amaro HM, Malcata FX. Microalgae as sources of carotenoids. Mar Drugs. 2011;9(4):625–44.
Updike AA, Schwartz SJ. Thermal processing of vegetables increases cis isomers of lutein and zeaxanthin. J Agric Food Chem. 2003;51(21):6184–90.
Boon CS, McClements DJ, Weiss J, Decker EA. Factors influencing the chemical stability of carotenoids in foods. Crit Rev Food Sci. 2010;50(6):515–32.
Oliver J, Palou A. Chromatographic determination of carotenoids in foods. J Chromatogr A. 2000;881(1–2):543–55.
Amorim-Carrilho KT, Cepeda A, Fente C, Regal P. Review of methods for analysis of carotenoids. Trac-Trend Anal Chem. 2014;56:49–73.
de Quiros ARB, Costa HS. Analysis of carotenoids in vegetable and plasma samples: a review. J Food Compos Anal. 2006;19(2–3):97–111.
Hodisan T, Socaciu C, Ropan I, Neamtu G. Carotenoid composition of Rosa canina fruits determined by thin-layer chromatography and high-performance liquid chromatography. J Pharm Biomed Anal. 1997;16(3):521–8.
Isaksen M, Francis GW. Reversed-phase thin-layer chromatography of carotenoids. J Chromatogr. 1986;355(1):358–62.
Ligia B. Almeida MVCP. Carotenoids and pro-vitamin A value of white fleshed Brazilian sweet potatoes (Ipomoea batatas Lam.). J Food Compos Anal. 1988;1(4):341–52.
Lane JR, Webb LW, Acuff RV. Concurrent liquid chromatographic separation and photodiode array detection of retinol, tocopherols, all-trans-alpha-carotene, all-trans-beta-carotene and the mono-cis isomers of beta-carotene in extracts of human plasma. J Chromatogr A. 1997;787(1–2):111–8.
Hart DJ, Scott KJ. Development and evaluation of an Hplc method for the analysis of carotenoids in foods, and the measurement of the carotenoid content of vegetables and fruits commonly consumed in the Uk. Food Chem. 1995;54(1):101–11.
Inbaraj BS, Lu H, Hung CF, Wu WB, Lin CL, Chen BH. Determination of carotenoids and their esters in fruits of Lycium barbarum Linnaeus by HPLC-DAD-APCI-MS. J Pharm Biomed Anal. 2008;47(4–5):812–8.
Dietz JM, Sri Kantha S, Erdman Jr JW. Reversed phase HPLC analysis of alpha- and beta-carotene from selected raw and cooked vegetables. Plant Foods Hum Nutr. 1988;38(4):333–41.
Herrero-Martinez JM, Eeltink S, Schoenmakers PJ, Kok WT, Ramis-Ramos G. Determination of major carotenoids in vegetables by capillary electrochromatography. J Sep Sci. 2006;29(5):660–5.
Lakshminarayana R, Raju M, Krishnakantha TP, Baskaran V. Determination of major carotenoids in a few Indian leafy vegetables by high-performance liquid chromatography. J Agric Food Chem. 2005;53(8):2838–42.
Bijttebier S, D’Hondt E, Noten B, Hermans N, Apers S, Voorspoels S. Ultra high performance liquid chromatography versus high performance liquid chromatography: stationary phase selectivity for generic carotenoid screening. J Chromatogr A. 2014;1332:46–56.
Matsubara A, Uchikata T, Shinohara M, Nishiumi S, Yoshida M, Fukusaki E, et al. Highly sensitive and rapid profiling method for carotenoids and their epoxidized products using supercritical fluid chromatography coupled with electrospray ionization-triple quadrupole mass spectrometry. J Biosci Bioeng. 2012;113(6):782–7.
Abrahamsson V, Rodriguez-Meizoso I, Turner C. Determination of carotenoids in microalgae using supercritical fluid extraction and chromatography. J Chromatogr A. 2012;1250:63–8.
Sander LC, Sharpless KE, Craft NE, Wise SA. Development of engineered stationary phases for the separation of carotenoid isomers. Anal Chem. 1994;66(10):1667–74.
Dugo P, Giuffrida D, Herrero M, Donato P, Mondello L. Epoxycarotenoids esters analysis in intact orange juices using two-dimensional comprehensive liquid chromatography. J Sep Sci. 2009;32(7):973–80.
Epler KS, Sander LC, Ziegler RG, Wise SA, Craft NE. Evaluation of reversed-phase liquid-chromatographic columns for recovery and selectivity of selected carotenoids. J Chromatogr. 1992;595(1–2):89–101.
Matsubara A, Bamba T, Ishida H, Fukusaki E, Hirata K. Highly sensitive and accurate profiling of carotenoids by supercritical fluid chromatography coupled with mass spectrometry. J Sep Sci. 2009;32(9):1459–64.
Lesellier E, Gurdale K, Tchapla A. Separation of cis/trans isomers of β-carotene by supercritical fluid chromatography. J Chromatogr A. 1999;844(1–2):307–20.
Li B, Zhao H, Liu J, Liu W, Fan S, Wu G, et al. Application of ultra-high performance supercritical fluid chromatography for the determination of carotenoids in dietary supplements. J Chromatogr A. 2015;1425:287–92.
Smith RM. Supercritical fluid chromatography. K D Bartle (Eds), Theory and Principles of Supercritical Fluid Chromatography, Royal Society of Chemistry; Reprint Edition (1989). 1989.
Smith RM. Supercritical fluids in separation science—the dreams, the reality and the future. J Chromatogr A. 1999;856(1–2):83–115.
Macias-Sanchez MD, Fernandez-Sevilla JM, Fernandez FGA, Garcia MCC, Grima EM. Supercritical fluid extraction of carotenoids from Scenedesmus almeriensis. Food Chem. 2010;123(3):928–35.
West C, Lemasson E, Bertin S, Hennig P, Lesellier E. An improved classification of stationary phases for ultra-high performance supercritical fluid chromatography. J Chromatogr A. 2016;1440:212–28.
Khater S, West C, Lesellier E. Characterization of five chemistries and three particle sizes of stationary phases used in supercritical fluid chromatography. J Chromatogr A. 2013;1319:148–59.
West C, Lesellier E. A unified classification of stationary phases for packed column supercritical fluid chromatography. J Chromatogr A. 2008;1191(1–2):21–39.
Jumaah F, Larsson S, Essen S, Cunico LP, Holm C, Turner C, et al. A rapid method for the separation of vitamin D and its metabolites by ultra-high performance supercritical fluid chromatography-mass spectrometry. J Chromatogr A. 2016;1440:191–200.
Rivera SM, Christou P, Canela-Garayoa R. Identification of carotenoids using mass spectrometry. Mass Spectrom Rev. 2014;33(5):353–72.
Rivera SM, Canela-Garayoa R. Analytical tools for the analysis of carotenoids in diverse materials. J Chromatogr A. 2012;1224:1–10.
Rivera S, Vilaro F, Canela R. Determination of carotenoids by liquid chromatography/mass spectrometry: effect of several dopants. Anal Bioanal Chem. 2011;400(5):1339–46.
Acknowledgments
Research support provided by Kurdistan Regional Government-Human Capacity Development Program (KRG-HCDP), Bokelunds resestipendiefond (94507), the Swedish Research Council FORMAS (239-2013-971), Lund University Antidiabetic Food Centre (LU-AFC, Vinnova), and the Swedish Research Council VR (2010–333).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional information
Parts of this work have been presented at Euroanalysis 2015 in Bordeaux, France.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM. 1
(PDF 333 kb)
Rights and permissions
About this article
Cite this article
Jumaah, F., Plaza, M., Abrahamsson, V. et al. A fast and sensitive method for the separation of carotenoids using ultra-high performance supercritical fluid chromatography-mass spectrometry. Anal Bioanal Chem 408, 5883–5894 (2016). https://doi.org/10.1007/s00216-016-9707-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9707-5