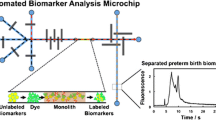
Abstract
Preterm birth (PTB) is defined as birth before the 37th week of pregnancy and results in 15 million early deliveries worldwide every year. Presently, there is no clinical test to determine PTB risk; however, a panel of nine biomarkers found in maternal blood serum has predictive power for a subsequent PTB. A significant step in creating a clinical diagnostic for PTB is designing an automated method to extract and purify these biomarkers from blood serum. Here, microfluidic devices with 45 μm × 50 μm cross-section channels were 3D printed with a built-in polymerization window to allow a glycidyl methacrylate monolith to be site-specifically polymerized within the channel. This monolith was then used as a solid support to attach antibodies for PTB biomarker extraction. Using these functionalized monoliths, it was possible to selectively extract a PTB biomarker, ferritin, from buffer and a human blood serum matrix. This is the first demonstration of monolith formation in a 3D printed microfluidic device for immunoaffinity extraction. Notably, this work is a crucial first step toward developing a 3D printed microfluidic clinical diagnostic for PTB risk.




Similar content being viewed by others
References
World Health Organization. Preterm birth. In: Fact sheets. WHO. 2018. http://www.who.int/mediacentre/factsheets/fs363/en/. Accessed 3 Jul 2018.
Centers for Disease Control and Prevention. Preterm birth. In: Maternal and infant health. CDC. 2018. https://www.cdc.gov/reproductivehealth/maternalinfanthealth/pretermbirth.htm. Accessed 3 Jul 2018.
Behrman RE, Butler AS. Preterm birth: causes, consequences, and prevention. Washington DC: National Academies Press; 2007.
Blencowe H, Cousens S, Oestergaard MZ, Chou D, Moller AB, Narwal R, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet. 2012;379:2162–72.
Roberto R, Sudhansu KD, Susan JF. Preterm labor: one syndrome, many causes. Science. 2014;345:760–5.
Esplin MS, Merrell K, Goldenberg R, Lai Y, Iams JD, Mercer B, et al. Proteomic identification of serum peptides predicting subsequent spontaneous preterm birth. Am J Obstet Gynecol. 2011;204:391.e8.
Sahore V, Sonker M, Nielsen AV, Knob R, Kumar S, Woolley AT. Automated microfluidic devices integrating solid-phase extraction, fluorescent labeling, and microchip electrophoresis for preterm birth biomarker analysis. Anal Bioanal Chem. 2018;410:933–41.
Sonker M, Parker EK, Nielsen AV, Sahore V, Woolley AT. Electrokinetically operated microfluidic devices for integrated immunoaffinity monolith extraction and electrophoretic separation of preterm birth biomarkers. Analyst. 2018;143:224–31.
Nielsen AV, Nielsen JB, Sonker M, Knob R, Sahore V, Woolley AT. Microchip electrophoresis separation of a panel of preterm birth biomarkers. Electrophoresis. 2018;39:2300–7.
Chin CD, Linder V, Sia SK. Commercialization of microfluidic point-of-care diagnostic devices. Lab Chip. 2012;13:2118–34.
Sonker M, Sahore V, Woolley AT. Recent advances in microfluidic sample preparation and separation techniques for molecular biomarker analysis: a critical review. Anal Chim Acta. 2017;986:1–11.
Núñez O, Nakanishi K, Tanaka N. Preparation of monolithic silica columns for high-performance liquid chromatography. J Chromatogr A. 2008;1191:231–52.
Knob R, Sahore V, Sonker M, Woolley AT. Advances in monoliths and related porous materials for microfluidics. Biomicrofluidics. 2016;10:032901–19.
Andjelković U, Tufegdžić S, Popović M. Use of monolithic supports for high-throughput protein and peptide separation in proteomics. Electrophoresis. 2017;38:2851–69.
Svec F, Lv Y. Advances and recent trends in the field of monolithic columns for chromatography. Anal Chem. 2015;87:250–73.
Tetala KKR, Vijayalakshmi MA. A review on recent developments for biomolecule separation at analytical scale using microfluidic devices. Anal Chim Acta. 2016;906:7–21.
Buchmeiser MR. Polymeric monolithic materials: syntheses, properties, functionalization and applications. Polymer. 2007;48:2187–98.
Masini JC, Svec F. Porous monoliths for on-line sample preparation: a review. Anal Chim Acta. 2017;964:24–44.
Yang W, Sun X, Wang HY, Woolley AT. Integrated microfluidic device for serum biomarker quantitation using standard addition or a calibration curve. Anal Chem. 2009;81:8230–5.
Yang W, Yu M, Sun X, Woolley AT. Microdevices integrating affinity columns and capillary electrophoresis for multibiomarker analysis in human serum. Lab Chip. 2010;10:2527–33.
Svec F. Porous polymer monoliths: amazingly wide variety of techniques enabling their preparation. J Chromatogr A. 2010;1217:902–24.
Dziomba S, Araya-Farias M, Smadja C, Taverna M, Carbonnier B, Tran NT. Solid supports for extraction and preconcentration of proteins and peptides in microfluidic devices: a review. Anal Chim Acta. 2017;955:1–26.
Pandey C, Augustine S, Kumar S, Kumar S, Nara S, Srivastave S, et al. Microfluidics based point-of-care diagnostics. Biotechnol J. 2018;13:1700047.
Nge PN, Rogers CI, Woolley AT. Advances in microfluidic materials, functions, integration, and applications. Chem Rev. 2013;113:2550–83.
Erickson D, Li D. Integrated microfluidic devices. Anal Chim Acta. 2004;507:11–26.
Hu J, Cui X, Gong Y, Xu X, Gao B, Wen T, et al. Portable microfluidic and smartphone-based devices for monitoring of cardiovascular diseases at the point of care. Biotechnol Adv. 2016;34:305–20.
Yazdi AA, Popma A, Wong W, Nguyen T, Pan Y, Xu J. 3D printing: an emerging tool for novel microfluidics and lab-on-a-chip applications. Microfluid Nanofluid. 2016;20:50.
Berman B. 3-D printing: the new industrial revolution. Bus Horiz. 2012;55:155–62.
Stansbury JW, Idacavage MJ. 3D printing with polymers: challenges among expanding options and opportunities. Dent Mater. 2016;32:54–64.
Beauchamp MJ, Nordin GP, Woolley AT. Moving from millifluidic to truly microfluidic sub-100-μm cross-section 3D printed devices. Anal Bioanal Chem. 2017;409:4311–9.
Shallan AI, Smejkal P, Corban M, Guijt RM, Breadmore MC. Cost-effective three-dimensional printing of visibly transparent microchips within minutes. Anal Chem. 2014;86:3124–30.
Anciaux SK, Geiger M, Bowser MT. 3D printed micro free-flow electrophoresis device. Anal Chem. 2016;88:7675–82.
Anderson KB, Lockwood SY, Martin RS, Spence DM. A 3D printed fluidic device that enables integrated features. Anal Chem. 2013;85:5622–6.
Sochol RD, Sweet E, Glick CC, Venkatesh S, Avetisyan A, Ekman KG, et al. 3D printed microfluidic circuitry via multijet based additive manufacturing. Lab Chip. 2016;16:668–78.
Lee KG, Park KJ, Seok S, Shin S, Kim DH, Park JY, et al. 3D printed modules for integrated microfluidic devices. RSC Adv. 2014;4:32876–80.
Lee JM, Zhang M, Yeong WY. Characterization and evaluation of 3D printed microfluidic chip for cell processing. Microfluid Nanofluid. 2016;20:1–15.
Gong H, Beauchamp M, Perry S, Woolley AT, Nordin GP. Optical approach to resin formulation for 3D printed microfluidics. RSC Adv. 2015;5:106621–32.
Gong H, Bickham BP, Woolley AT, Nordin GP. Custom 3D printer and resin for 18 μm × 20 μm microfluidic flow channels. Lab Chip. 2017;17:2899–909.
Beauchamp MJ, Gong H, Woolley AT, Nordin GP. 3D printed microfluidic features using dose control in X, Y, and Z dimensions. Micromachines. 2018;9:326-1-326-12.
Sahore V, Kumar S, Rogers CI, Sonker M, Jensen JK, Woolley AT. Pressure-actuated microfluidic devices for electrophoretic separation of pre-term birth biomarkers. Anal Bioanal Chem. 2016;408:599–607.
Gong H, Woolley AT, Nordin GP. 3D printed high density, reversible, chip-to-chip microfluidic interconnects. Lab Chip. 2018;18:639–47.
Gong H, Woolley AT, Nordin GP. High density 3D printed microfluidic valves, pumps, and multiplexers. Lab Chip. 2016;16:2450–8.
Funding
The authors acknowledge support from the National Institutes of Health (R01 EB006124 and R15 GM123405-01A1). A.V.N. and M.J.B. thank the BYU Department of Chemistry and Biochemistry for financial support from a Roland K. Robins Fellowship, and H.M.A acknowledges Taif University for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Research involving human participants
Human blood serum samples were obtained from commercial sources; human subjects work has been approved under IRB no. E18401.
Additional information
Published in the topical collection Ultrasmall Sample Biochemical Analysis with guest editors Ryan Kelly and Ying Zhu.
Electronic supplementary material
ESM 1
(PDF 4001 kb)
Rights and permissions
About this article
Cite this article
Parker, E.K., Nielsen, A.V., Beauchamp, M.J. et al. 3D printed microfluidic devices with immunoaffinity monoliths for extraction of preterm birth biomarkers. Anal Bioanal Chem 411, 5405–5413 (2019). https://doi.org/10.1007/s00216-018-1440-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-1440-9