Abstract
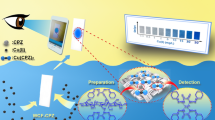
Diamino-oxidase (DAO), horseradish peroxidase (HRP), and tetramethylbenzidine (TMB) have been immobilized into cellulose to obtain circular cellulose test supports (CCTSs) for the determination of cadaverine (Cad) and putrescine (Put). During the enzymatic reaction, TMB is oxidized and a blue spot is obtained. This color (RGB coordinates) is measured with a smartphone and a commercial application. The highest sensitivity is provided by the component R and a linear response is observed for low biogenic amine (BA) concentrations, but a second-order polynomial response better fits the experimental results for a wider concentration range. This has been successfully explained with a model developed to explain the RGB values obtained in this type of analytical system. Optimization studies enable CCTSs to be obtained for Put and Cad determination, which could be used (kept at 4 °C) for at least 45 days if a stabilizer (StabilCoat™ or StabilGuard™) is added during its synthesis. In these conditions, the R coordinate follows the model up to at least 4 × 10−4 M Put and/or Cad (both analytes give the same response). The method permits the Put and Cad determination from 5 × 10−5 M up to 4 × 10−4 M (RSD = 3%, n = 3). The CCTSs have been applied to Put + Cad determination in a tuna sample without any interference by other biogenic amines. The concentration found statistically agrees with that obtained using a HPLC-MS-validated method.

Graphical abstract





Similar content being viewed by others
References
Ladero V, Calles-Enríquez M, Fernández M, Álvarez MA. Toxicological effects of dietary biogenic amines. Curr Nutr Food Sci. 2010;6:145–56. https://doi.org/10.2174/157340110791233256.
Bulushi IA, Poole S, Deeth HC, Dykes GA. Biogenic amines in fish: roles in intoxication, spoilage, and nitrosamine formation—a review. Crit Rev Food Sci Nutr. 2009;49:369–77. https://doi.org/10.1080/10408390802067514.
Choi MH, Kim KR, Kim YT, Chung BC. Increased polyamine concentrations in the hair of cancer patients. Clin Chem. 2001;47:143–4. https://doi.org/10.1093/clinchem/47.1.143.
Ernestus RI, Röhn G, Schröder R, Els T, Klekner Á, Paschen E, et al. Polyamine metabolism in brain tumours: diagnostic relevance of quantitative biochemistry. J Neurol Neurosurg Psychiatry. 2001;71:88–92. https://doi.org/10.1136/jnnp.71.1.88.
Levêque J, Foucher F, Bansard JY, Havouis R, Grall JY, Moluinoux JP. Polyamine profiles in tumor, normal tissue of the homologous breast, blood, and urine of breast cancer sufferers. Breast Cancer Res Treat. 2000;60:99–105. https://doi.org/10.1023/A:1006319818530.
Önal A, Tekkeli SEK, Önal C. A review of the liquid chromatographic methods for the determination of biogenic amines in foods. Food Chem. 2013;138:509–15. https://doi.org/10.1016/j.foodchem.2012.10.056.
Erim FB. Recent analytical approaches to the analysis of biogenic amines in food samples. Trends Anal Chem. 2013;52:239–47. https://doi.org/10.1016/j.trac.2013.05.018.
Ordóñez JL, Troncoso AM, García-Parrilla MDC, Callejón RM. Recent trends in the determination of biogenic amines in fermented beverages - a review. Anal Chim Acta. 2016;939:10–25. https://doi.org/10.1016/j.aca.2016.07.045.
Lerke PA, Porcuna MH, Hin HB. Screening test for histamine in fish. J Food Sci. 1983;48:155–7. https://doi.org/10.1111/j.1365-2621.1983.tb14812.x.
Tombelli S, Mascini M. Electrochemical biosensors for biogenic amines: a comparison between different approaches. Anal Chim Acta. 1998;358:277–84. https://doi.org/10.1016/S0003-2670(97)00606-5.
Leonardo S, Campàs M. Electrochemical enzyme sensor arrays for the detection of the biogenic amines histamine, putrescine and cadaverine using magnetic beads as immobilisation supports. Microchim Acta. 2016;183:1881–90. https://doi.org/10.1007/s00604-016-1821-8.
Yang X, Feng B, He X, Li F, Ding Y, Fei J. Carbon nanomaterial based electrochemical sensors for biogenic amines. Microchim Acta. 2013;180:935–56. https://doi.org/10.1007/s00604-013-1015-6.
Kivirand K, Rinken T. Biosensors for biogenic amines: the present state of art mini-review. Anal Lett. 2011;44:2821–33. https://doi.org/10.1080/00032719.2011.565445.
Lee JI, Jang JH, Yu MJ, Kim YW. Construction of a bifunctional enzyme fusion for the combined determination of biogenic amines in foods. J Agric Food Chem. 2013;61:9118–24. https://doi.org/10.1021/jf403044m.
Navarro J, Sanz-Vicente I, Lozano R, de Marcos S, Galbán J. Analytical possibilities of putrescine and cadaverine enzymatic colorimetric determination in tuna based on diamine oxidase: a critical study of the use of ABTS. Talanta. 2020;208:120392. https://doi.org/10.1016/j.talanta.2019.120392.
Chen YL, Jonathan Y. Enzyme immobilization on cellulose matrixes. J Bioact Compat Polym. 2016:1–15. https://doi.org/10.1177/0883911516637377.
Hall M, Eldridge DB, Saunders RD, Fairclough DL, Bateman RC. A rapid dipstick test for histamine in tuna. Food Biotechnol. 1995;9:39–57. https://doi.org/10.1080/08905439509549884.
Hall M, Sykes PA, Fairclough DL, Lucchese J, Rogers P, Staruszkiewicz W, et al. A test strip for diamines in tuna. JAOAC Int. 1999;82:1102–8.
Calabria D, Caliceti C, Zangheri M, Mirasoli M, Simoni P, Roda A. Smartphone–based enzymatic biosensor for oral fluid L-lactate detection in one minute using confined multilayer paper reflectometry. Biosens Bioelectron. 2017;94:124–30. https://doi.org/10.1016/j.bios.2017.02.053.
ITE-FQ080-02: determination of biogenic amines by HPLC-MS/MS. Public Health Laboratory. Government of Aragón (Spain). https://gobierno.aragon.es/estaticos/GobiernoAragon/Departamentos/SanidadBienestarSocialFamilia/Sanidad/Profesionales/13_SaludPublica/Cartera_Servicios_Laboratorio_20190507.pdf
Pietrangeli P, Federico R, Mondovì B, Morpurgo L. Substrate specificity of copper-containing plant amine oxidases. J Inorg Biochem. 2007;101:997–1004. https://doi.org/10.1016/j.jinorgbio.2007.03.014.
Connah D, Westland S, Thomson MGA. Recovering spectral information using digital camera systems. Color Technol. 2001;117:309–12. https://doi.org/10.1111/j.1478-4408.2001.tb00080.x.
Goldman J. Quantitative analysis on thin-layer chromatograms. Theory of absorption and fluorescent densitometry. J Chromatogr. A1973. 78:7–19. https://doi.org/10.1016/S0021-9673(01)99031-4.
Acknowledgments
J Navarro thanks to the Government of Aragon (DGA) for a grant. The authors are also very grateful to Dr. Francisco Palacios and MSc Cristina Asensio from Laboratorio de Salud Pública de Aragón (LSPA, DGA) for their invaluable help. This work was the winner of a Poster Prize awarded for excellent presentation of particularly significant innovative analytical research at X CIE/XXVI RNE. This award was sponsored by Analytical and Bioanalytical Chemistry.
Funding
This work was supported by the MINECO of Spain (project CTQ2016-76846R) and by research groups funding DGA-FEDER (group E25_17R).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal interest
The research does not involve human participants and/or animals.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1369 kb)
Rights and permissions
About this article
Cite this article
Sanz-Vicente, I., López-Molinero, Á., de Marcos, S. et al. Smartphone-interrogated test supports for the enzymatic determination of putrescine and cadaverine in food. Anal Bioanal Chem 412, 4261–4271 (2020). https://doi.org/10.1007/s00216-020-02677-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-020-02677-7