Abstract
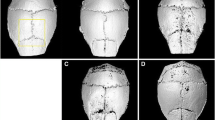
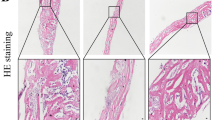
Particle-induced bone loss by osteoclasts is a common cause of aseptic loosening around implants. This study investigates whether caffeic acid phenethyl ester (CAPE), a potent and specific inhibitor of nuclear factor of activated T cells, cytoplasmic, calcineurin-dependent 1 and nuclear factor kappa B, at a low dose reduces bone resorption in a murine calvarial model of polyethylene (PE) particle-induced osteolysis. The effects of particles and CAPE treatment on gastrointestinal tract (GIT) histopathology were also evaluated. Mice were scanned using in vivo animal micro-computed tomography (μCT) as a baseline measurement. PE particles (2.82 × 109 particles/mL) were implanted over the calvariae on day 0. CAPE was administered subcutaneously (1 mg/kg/day) at days 0, 4, 7 and 10. Mice were killed at day 14 and serum was analysed for Type-1 carboxyterminal collagen crosslinks (CTX)-1 and osteoclast-associated receptor (OSCAR) levels. Ex vivo μCT scans were conducted to assess bone volume (BV) change and percentage area of calvarial surface resorbed. Calvarial and GIT tissue was processed for histopathology. By day 14, PE particles significantly induced calvarial bone loss compared with control animals as evidenced by resorption areas adjacent to the implanted PE in three-dimensional μCT images, an increase in percentage of resorbed area (p = 0.0022), reduction in BV (p = 0.0012) and increased Tartrate-resistant acid phosphatase positive cells. Serum CTX-1 (p = 0.0495) and OSCAR levels (p = 0.0006) significantly increased in the PE implant group. CAPE significantly inhibited PE particle-induced calvarial osteolysis, as evidenced by a significant reduction in surface bone resorption (p = 0.0012) and volumetric change (p = 0.0154) compared with PE only, but had no effect on systemic CTX-1. Neither particles nor CAPE had an effect on GIT histopathology.





Similar content being viewed by others
References
Haynes DR, Crotti TN, Potter AE, Loric M, Atkins GJ, Howie DW, Findlay DM (2001) The osteoclastogenic molecules RANKL and RANK are associated with periprosthetic osteolysis. J Bone Joint Surg Br 83:902–911
Crotti TN, Smith MD, Findlay DM, Zreiqat H, Ahern MJ, Weedon H, Hatzinikolous G, Capone M, Holding C, Haynes DR (2004) Factors regulating osteoclast formation in human tissues adjacent to peri-implant bone loss: expression of receptor activator NFkappaB, RANK ligand and osteoprotegerin. Biomaterials 25:565–573
Holding CA, Findlay DM, Stamenkov R, Neale SD, Lucas H, Dharmapatni AS, Callary SA, Shrestha KR, Atkins GJ, Howie DW, Haynes DR (2006) The correlation of RANK, RANKL and TNFalpha expression with bone loss volume and polyethylene wear debris around hip implants. Biomaterials 27:5212–5219
Tuan RS, Lee FY, Konttinen YT, Wilkinson JM, Smith RL (2008) What are the local and systemic biologic reactions and mediators to wear debris, and what host factors determine or modulate the biologic response to wear particles? J Am Acad Orthop Surg 16(Suppl 1):S42–S48
Mikuls TR (2003) Co-morbidity in rheumatoid arthritis. Best Pract Res Clin Rheumatol 17:729–752
Crotti TN, Sharma SM, Fleming JD, Flannery MR, Ostrowski MC, Goldring SR, McHugh KP (2008) PU.1 and NFATc1 mediate osteoclastic induction of the mouse beta3 integrin promoter. J Cell Physiol 215:636–644
Crotti TN, Flannery M, Walsh NC, Fleming JD, Goldring SR, McHugh KP (2006) NFATc1 regulation of the human beta3 integrin promoter in osteoclast differentiation. Gene 372:92–102
Clohisy JC, Roy BC, Biondo C, Frazier E, Willis D, Teitelbaum SL, Abu-Amer Y (2003) Direct inhibition of NF-kappa B blocks bone erosion associated with inflammatory arthritis. J Immunol 171:5547–5553
Yip KH, Zheng MH, Feng HT, Steer JH, Joyce DA, Xu J (2004) Sesquiterpene lactone parthenolide blocks lipopolysaccharide-induced osteolysis through the suppression of NF-kappaB activity. J Bone Miner Res 19:1905–1916
Schwarz EM, Lu AP, Goater JJ, Benz EB, Kollias G, Rosier RN, Puzas JE, O’Keefe RJ (2000) Tumor necrosis factor-alpha/nuclear transcription factor-kappaB signaling in periprosthetic osteolysis. J Orthop Res 18(3):472–480
Gordon AH, O’Keefe RJ, Schwarz EM, Rosier RN, Puzas JE (2005) Nuclear factor-kappaB-dependent mechanisms in breast cancer cells regulate tumor burden and osteolysis in bone. Cancer Res 65:3209–3217
Li Y, Zhang C, Zhou X, Wang H, Mao Y, Wang X (2013) Parthenolide inhibits polyethylene particle-induced mouse calvarial osteolysis in vivo. J Surg Res 12:176–181
Koga T, Inui M, Inoue K, Kim S, Suematsu A, Kobayashi E, Iwata T, Ohnishi H, Matozaki T, Kodama T, Taniguchi T, Takayanagi H, Takai T (2004) Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428:758–763
Kim Y, Sato K, Asagiri M, Morita I, Soma K, Takayanagi H (2005) Contribution of nuclear factor of activated T cells c1 to the transcriptional control of immunoreceptor osteoclast-associated receptor but not triggering receptor expressed by myeloid cells-2 during osteoclastogenesis. J Biol Chem 280:32905–32913
Ishikawa S, Arase N, Suenaga T, Saita Y, Noda M, Kuriyama T, Arase H, Saito T (2004) Involvement of FcRgamma in signal transduction of osteoclast-associated receptor (OSCAR). Int Immunol 16(7):1019–1025
Alias E, Dharmapatni ASSK, Holding AC, Atkins GJ, Findlay DM, Howie DW, Crotti TN, Haynes DR (2012) Polyethylene particles stimulate expression of ITAM-related molecules in peri-implant tissues and when stimulating osteoclastogenesis in vitro. Acta Biomater 8(8):3104–3112
Zawawi MSF, Dharmapatni AA, Cantley MD, McHugh KP, Haynes DR, Crotti TN (2012) Regulation of ITAM adaptor molecules and their receptors by inhibition of calcineurin-NFAT signalling during late stage osteoclast differentiation. Biochem Biophys Res Commun 427(2):404–409
Crotti TN, Dharmapatni AA, Alias E, Zannettino AC, Smith MD, Haynes DR (2012) The immunoreceptor tyrosine-based activation motif (ITAM) -related factors are increased in synovial tissue and vasculature of rheumatoid arthritic joints. Arthr Res Ther 14:R245
Herman S, Muller RB, Kronke G, Zwerina J, Redlich K, Hueber AJ, Gelse H, Neumann E, Muller-Ladner U, Schett G (2008) Induction of osteoclast-associated receptor, a key osteoclast costimulation molecule, in rheumatoid arthritis. Arthr Rheum 58:3041–3050
Zhao S, Guo YY, Ding N, Yang LL, Zhang N (2011) Changes in serum levels of soluble osteoclast-associated receptor in human rheumatoid arthritis. Chin Med J (Engl) 124:3058–3060
Ndongo-Thiam N, de Sallmard G, Kastrup J, Miossec P (2014) Levels of soluble osteoclast-associated receptor (sOSCAR) in rheumatoid arthritis: link to disease severity and cardiovascular risk. Ann Rheum Dis 73(6):1276–1277
Natarajan K, Singh S, Burke TRJR, Grunberger D, Aggarwal BB (1996) Caffeic acid phenethyl ester is a potent and specific inhibitor of activation of nuclear transcription factor NF-kappa B. Proc Natl Acad Sci USA 93(17):9090–9095
Ang ES, Pavlos NJ, Chai LY, Qi M, Cheng TS, Steer JH, Joyce DA, Zheng MH, Xu J (2009) Caffeic acid phenethyl ester, an active component of honeybee propolis attenuates osteoclastogenesis and bone resorption via the suppression of RANKL-induced NF-kappaB and NFAT activity. J Cell Physiol 221:642–649
Marquez N, Sancho R, Macho A, Calzado MA, Fiebich BL, Munoz E (2004) Caffeic acid phenethyl ester inhibits T-cell activation by targeting both nuclear factor of activated T-cells and NF-kappaB transcription factors. J Pharmacol Exp Ther 308:993–1001
Michaluart P, Masferrer JL, Carothers AM, Subbaramaiah K, Zweifel BS, Koboldt C, Mestre JR, Grunberger D, Sacks PG, Tanabe T, Dannenberg AJ (1999) Inhibitory effects of caffeic acid phenethyl ester on the activity and expression of cyclooxygenase-2 in human oral epithelial cells and in a rat model of inflammation. Cancer Res 59:2347–2352
Fitzpatrick LR, Wang J, Le T (2001) Caffeic acid phenethyl ester, an inhibitor of nuclear factor-kappaB, attenuates bacterial peptidoglycan polysaccharide-induced colitis in rats. J Pharmacol Exp Ther 299(3):915–920
Wedemeyer C, Xu J, Neuerburg C, Landgraeber S, Malyar NM, von Knoch F, Gosheger G, von Knoch M, Loer F, Saxler G (2007) Particle-induced osteolysis in three-dimensional micro-computed tomography. Calcif Tissue Int 81:394–402
Zhou X, Zhang C, Wang X, An B, Zhang P, Zhu Z (2012) Berberine inhibits lipopolysaccharide- and polyethylene particle-induced mouse calvarial osteolysis in vivo. J Surg Res 173:e47–e52
von Knoch M, Sprecher C, Barden B, Saxler G, Löer F, Wimmer M (2004) Size and shape of commercially available polyethylene particles for in vitro and in vivo-experiments. Z Orthop Ihre Grenzgeb 142(3):366–370
Merkel KD, Erdmann JM, McHugh KP, Abu-Amer Y, Ross FP, Teitelbaum SL (1999) Tumor necrosis factor-alpha mediates orthopedic implant osteolysis. Am J Pathol 154:203–210
Cantley MD, Bartold PM, Marino V, Reid RC, Fairlie DP, Wyszynski RN, Zilm PS, Haynes DR (2009) The use of live-animal micro-computed tomography to determine the effect of a novel phospholipase A2inhibitor on alveolar bone loss in anin vivomouse model of periodontitis. J Periodontal Res 44:317–322
Perilli E, Le V, Ma B, Salmon P, Reynolds K, Fazzalari NL (2010) Detecting early bone changes using in vivo micro-CT in ovariectomized, zoledronic acid-treated, and sham-operated rats. Osteoporos Int 21:1371–1382
Shen Z, Crotti TN, McHugh KP, Matsuzaki K, Gravallese EM, Bierbaum BE, Goldring SR (2006) The role played by cell-substrate interactions in the pathogenesis of osteoclast-mediated peri-implant osteolysis. Arthr Res Ther 8:R70
Burstone M (1958) Histochemical demonstration of acid phosphatases with naphthol AS-phosphates. J Natl Cancer Inst 21:523–539
Udagawa N, Takahashi N, Akatsu T, Sasaki T, Yamaguchi A, Kodama H, Martin TJ, Suda T (1989) The bone marrow-derived stromal cell lines MC3T3-G2/PA6 and ST2 support osteoclast-like cell differentiation in cocultures with mouse spleen cells. Endocrinology 125:1805–1813
Gibson RJ, Bowen JM, Inglis MR, Cummins AG, Keefe DM (2003) Irinotecan causes severe small intestinal damage, as well as colonic damage, in the rat with implanted breast cancer. J Gastroenterol Hepatol 18(9):1095–1100
Logan RM, Gibson RJ, Bowen JM, Stringer AM, Sonis ST, Keefe DM (2008) Characterisation of mucosal changes in the alimentary tract following administration of irinotecan: implications for the pathobiology of mucositis. Cancer Chemother Pharmacol 62(1):33–41
Kurtz SM, Ok M, Evans M, Edidin AA (1999) Advances in the processing, sterilization, and crosslinking of ultra-high molecular weight polyethylene for total joint arthroplasty. Biomaterials 20(18):1659–1688
Landgraeber S, Jaeckel S, Loer F, Wedemeyer C, Hilken G, Canbay A, Totsch M, von Knoch M (2009) Pan-caspase inhibition suppresses polyethylene particle-induced osteolysis. Apoptosis Int J Program Cell Death 14:173–181
Wedemeyer C, Neuerburg C, Pfeiffer A, Heckelei A, von Knoch F, Hilken G, Brankamp J, Henschke F, von Knoch M, Loer F, Saxler G (2007) Polyethylene particle-induced bone resorption in substance P-deficient mice. Calcif Tissue Int 80(4):268–274
Ha J, Choi HS, Lee Y, Lee ZH, Kim HH (2009) Caffeic acid phenethyl ester inhibits osteoclastogenesis by suppressing NF kappaB and downregulating NFATc1 and c-Fos. Int Immunopharmacol 9:774–780
Ang E, Pavlos NJ, Rea SL, Qi M, Chai T, Walsh JP, Ratajczak T, Zheng MH, Xu J (2009) Proteasome inhibitors impair RANKL-induced NF-kappaB activity in osteoclast-like cells via disruption of p62, TRAF6, CYLD, and IkappaBalpha signaling cascades. J Cell Physiol 220:450–459
Kim N, Takami M, Rho J, Josien R, Choi Y (2002) A novel member of the leukocyte receptor complex regulates osteoclast differentiation. J Exp Med 195:201–209
Nemeth K, Schoppet M, Al-Fakhri N, Helas S, Jessberger R, Hofbauer LC, Goettsch C (2011) The role of osteoclast-associated receptor in osteoimmunology. J Immunol 186(1):13–18
Orban Z, Mitsiades N, Burke TRJ, Tsokos M, Chrousos GP (2000) Caffeic acid phenethyl ester induces leukocyte apoptosis, modulates nuclear factor-kappa B and suppresses acute inflammation. NeuroImmunoModulation 7(2):99–105
Acknowledgments
The work has been supported by a New Appointment Grant, Faculty of Health Sciences, The University of Adelaide. The Malaysian Government (USM) provided support for M. S. F. Z.
Conflict of interest
M. S. F. Zawawi, E. Perilli, R. L. Stansborough, V. Marino, M. D. Cantley, J. Xu, A. A. S. S .K. Dharmapatni, D. R. Haynes, R. J. Gibson and T. N. Crotti state that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
This study was conducted in accordance with ethics approved by The University of Adelaide (M-2001-070) and SA Pathology (106/10) and complied with the National Health and Medical Research Council (Australia) Code of Practice for Animal Care in Research and Teaching (2014).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zawawi, M.S.F., Perilli, E., Stansborough, R.L. et al. Caffeic Acid Phenethyl Ester Abrogates Bone Resorption in a Murine Calvarial Model of Polyethylene Particle-Induced Osteolysis. Calcif Tissue Int 96, 565–574 (2015). https://doi.org/10.1007/s00223-015-9982-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-015-9982-8