Abstract
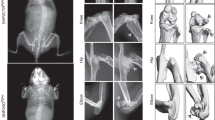
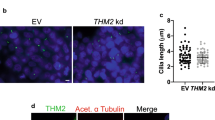
An autosomal-recessive inactivating mutation R272Q in the human intestinal cell kinase (ICK) gene caused profound multiplex developmental defects in human endocrine-cerebro-osteodysplasia (ECO) syndrome. ECO patients exhibited a wide variety of skeletal abnormalities, yet the underlying mechanisms by which ICK regulates skeletal development remained largely unknown. The goal of this study was to understand the structural and mechanistic basis underlying skeletal anomalies caused by ICK dysfunction. Ick R272Q knock-in transgenic mouse model not only recapitulated major ECO skeletal defects such as short limbs and polydactyly but also revealed a deformed spine with defective intervertebral disk. Loss of ICK function markedly reduced mineralization in the spinal column, ribs, and long bones. Ick mutants showed a significant decrease in the proliferation zone of long bones and the number of type X collagen-expressing hypertrophic chondrocytes in the spinal column and the growth plate of long bones. These results implicate that ICK plays an important role in bone and cartilage development by promoting chondrocyte proliferation and maturation. Our findings provided new mechanistic insights into the skeletal phenotype of human ECO and ECO-like syndromes.






Similar content being viewed by others
References
Kronenberg HM (2003) Developmental regulation of the growth plate. Nature 423:332–336
Olsen BR, Reginato AM, Wang W (2000) Bone development. Annu Rev Cell Dev Biol 16:191–220
Serra R (2008) Role of intraflagellar transport and primary cilia in skeletal development. Anat Rec (Hoboken) 291:1049–1061
Badano JL, Mitsuma N, Beales PL, Katsanis N (2006) The ciliopathies: an emerging class of human genetic disorders. Annu Rev Genomics Hum Genet 7:125–148
Lahiry P, Wang J, Robinson JF, Turowec JP, Litchfield DW, Lanktree MB, Gloor GB, Puffenberger EG, Strauss KA, Martens MB, Ramsay DA, Rupar CA, Siu V, Hegele RA (2009) A multiplex human syndrome implicates a key role for intestinal cell kinase in development of central nervous, skeletal, and endocrine systems. Am J Hum Genet 84:134–147
Oud MM, Bonnard C, Mans DA, Altunoglu U, Tohari S, Ng AY, Eskin A, Lee H, Rupar CA, de Wagenaar NP, Wu KM, Lahiry P, Pazour GJ, Nelson SF, Hegele RA, Roepman R, Kayserili H, Venkatesh B, Siu VM, Reversade B, Arts HH (2016) A novel ICK mutation causes ciliary disruption and lethal endocrine-cerebro-osteodysplasia syndrome. Cilia 5:8
Paige TS, Kunova BM, Varecha M, Balek L, Barta T, Trantirek L, Jelinkova I, Duran I, Vesela I, Forlenza KN, Martin JH, Hampl A, Bamshad M, Nickerson D, Jaworski ML, Song J, Ko HW, Cohn DH, Krakow D, Krejci P (2016) An inactivating mutation in intestinal cell kinase, ICK, impairs hedgehog signalling and causes short rib-polydactyly syndrome. Hum Mol Genet 25:3998–4011
Chaya T, Omori Y, Kuwahara R, Furukawa T (2014) ICK is essential for cell type-specific ciliogenesis and the regulation of ciliary transport. EMBO J 33:1227–1242
Moon H, Song J, Shin JO, Lee H, Kim HK, Eggenschwiller JT, Bok J, Ko HW (2014) Intestinal cell kinase, a protein associated with endocrine-cerebro-osteodysplasia syndrome, is a key regulator of cilia length and Hedgehog signaling. Proc Natl Acad Sci USA 111:8541–8546
Fu Z, Kim J, Vidrich A, Sturgill TW, Cohn SM (2009) Intestinal cell kinase, a MAP kinase-related kinase, regulates proliferation and G1 cell cycle progression of intestinal epithelial cells. Am J Physiol Gastrointest Liver Physiol 297:G632–G640
Bolick DT, Chen T, Alves LOA, Tong Y, Wu D, Joyner LT 2nd, Oria RB, Guerrant RL, Fu Z (2014) Intestinal cell kinase is a novel participant in intestinal cell signaling responses to protein malnutrition. PLoS ONE 9:e106902
Tong Y, Park SH, Wu D, Xu W, Guillot SJ, Jin L, Li X, Wang Y, Lin CS, Fu Z (2017) An essential role of intestinal cell kinase in lung development is linked to the perinatal lethality of human ECO syndrome. FEBS Lett 591:1247–1257
Rigueur D, Lyons KM (2014) Whole-mount skeletal staining. Methods. Mol Biol 1130:113–121
Xiao L, Ding M, Zhang Y, Chordia M, Pan D, Shimer A, Shen FH, Glover D, Jin L, Li X (2017) A novel modality for functional imaging in acute intervertebral disc herniation via tracking leukocyte infiltration. Mol Imaging Biol
Jin L, Liu Q, Scott P, Zhang D, Shen FH, Balian G, Li X (2014) Annulus fibrosus cell characteristics are a potential source of intervertebral disc pathogenesis. PLoS ONE 9:e96519
Xiao L, Ding M, Fernandez A, Zhao P, Jin L, Li X (2017) Curcumin alleviates lumbar radiculopathy by reducing neuroinflammation, oxidative stress and nociceptive factors. Eur Cell Mater 33:279–293
Chen T, Wu D, Moskaluk CA, Fu Z (2013) Distinct expression patterns of ICK/MAK/MOK protein kinases in the intestine implicate functional diversity. PLoS ONE 8:e79359
Karsenty G, Wagner EF (2002) Reaching a genetic and molecular understanding of skeletal development. Dev Cell 2:389–406
Horton WA (2003) Skeletal development: insights from targeting the mouse genome. Lancet 362:560–569
Breur GJ, Farnum CE, Padgett GA, Wilsman NJ (1992) Cellular basis of decreased rate of longitudinal growth of bone in pseudoachondroplastic dogs. J Bone Joint Surg Am 74:516–528
Michigami T (2013) Regulatory mechanisms for the development of growth plate cartilage. Cell Mol Life Sci 70:4213–4221
Sivakamasundari V, Lufkin T (2012) Bridging the gap: understanding embryonic intervertebral disc development. Cell Dev Biol 1:103
Yamanaka Y, Tamplin OJ, Beckers A, Gossler A, Rossant J (2007) Live imaging and genetic analysis of mouse notochord formation reveals regional morphogenetic mechanisms. Dev Cell 13:884–896
McCann MR, Tamplin OJ, Rossant J, Séguin CA (2012) Tracing notochord-derived cells using a Noto-cre mouse: implications for intervertebral disc development. Dis Model Mech 5:73–82
Moore ER, Jacobs CR (2017) The primary cilium as a signaling nexus for growth plate function and subsequent skeletal development. J Orthop Res
Acknowledgements
We are grateful for the partial financial support from NIH R01AR064792 (XL), DK082614 (ZF), and CA195273 (ZF). We appreciate the technical assistance of the Research Histology Core at University of Virginia. The funders have no role in the study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Mengmeng Ding, Li Jin, Lin Xie, So Hyun Park, Yixin Tong, Di Wu, A. Bobby Chhabra, Zheng Fu, Xudong Li declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All procedures involving animals were performed in accordance with ethical standards in an animal protocol that was approved by the Institutional Animal Care and Use Committee.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ding, M., Jin, L., Xie, L. et al. A Murine Model for Human ECO Syndrome Reveals a Critical Role of Intestinal Cell Kinase in Skeletal Development. Calcif Tissue Int 102, 348–357 (2018). https://doi.org/10.1007/s00223-017-0355-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-017-0355-3