Abstract
Introduction
We aimed to assess metabolite profiles in peri-infarct tissue with magnetic resonance spectroscopy (MRS) and correlate these with early and late clinical recovery.
Methods
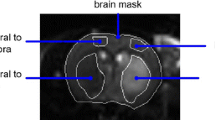
One hundred ten anterior circulation ischemic stroke patients presenting to hospital within 4.5 h of symptom onset and treated with intravenous thrombolysis were studied. Patients underwent computer tomography perfusion (CTP) scanning and subsequently 3-T magnetic resonance imaging (MRI) 24 h after stroke onset, including single-voxel, short-echo-time (30 ms) MRS, and diffusion- and perfusion-weighted imaging (DWI and PWI). MRS voxels were placed in the peri-infarct region in reperfused penumbral tissue. A control voxel was placed in the contralateral homologous area.
Results
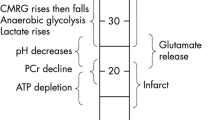
The concentrations of total creatine (5.39 vs 5.85 mM, p = 0.044) and N-acetylaspartic acid (NAA, 6.34 vs 7.13 mM ± 1.57, p < 0.001) were reduced in peri-infarct tissue compared to the matching contralateral region. Baseline National Institutes of Health Stroke Score was correlated with glutamate concentration in the reperfused penumbra at 24 h (r 2 = 0.167, p = 0.017). Higher total creatine was associated with better neurological outcome at 24 h (r 2 = 0.242, p = 0.004). Lower peri-infarct glutamate was a stronger predictor of worse 3-month clinical outcome (area under the curve (AUC) 0.89, p < 0.001) than DWI volume (AUC = 0.79, p < 0.001).
Conclusion
Decreased glutamate, creatine, and NAA concentrations are associated with poor neurological outcome at 24 h and greater disability at 3 months. The significant metabolic variation in salvaged tissue may potentially explain some of the variability seen in stroke recovery despite apparently successful reperfusion.



Similar content being viewed by others
References
Nagakane Y, Christensen S, Brekenfeld C, Ma H, Churilov L, Parsons MW, Levi CR, Butcher KS, Peeters A, Barber PA, Bladin CF, De Silva DA, Fink J, Kimber TE, Schultz DW, Muir KW, Tress BM, Desmond PM, Davis SM, Donnan GA, for the EPITHET Investigators (2011) EPITHET: positive result after reanalysis using baseline diffusion-weighted imaging/perfusion-weighted imaging co-registration. Stroke 42:59–64
O’CollMI VE, Macleod MR, Donnan GA, Horky LL, van der Worp BH, Howells DW (2006) 1,026 experimental treatments in acute stroke. Ann Neurol 59:467–477
Corbett D, Nguemeni C, Gomez-Smith M (2014) How can you mend a broken brain? Neurorestorative approaches to stroke recovery. Cerebrovasc Dis 38(4):233–239
Saur D, Buchert R, Knab R, Weiller C, Röther J (2006) Iomazenil-single-photon emission computed tomography reveals selective neuronal loss in magnetic resonance-defined mismatch areas. Stroke 37(11):2713–2719
Patel TB, Clark JB (1979) Synthesis of N-acetyl-L-aspartate by rat brain mitochondria and its involvement in mitochondrial/cytosolic carbon transport. Biochem J 184:539–546
Baron JC, Yamauchi H, Fujioka M, Endres M (2014) Selective neuronal loss in ischemic stroke and cerebrovascular disease. J Cereb Blood Flow Metab 34:2–18
Guadagno JV, Jones PS, Aigbirhio FI, Wang D, Fryer TD, Day DJ, Antoun N, Nimmo-Smith I, Warburton EA, Baron JC (2008) Selective neuronal loss in rescued penumbra relates to initial hypoperfusion. Brain 131:2666–267
Parsons MW, Miteff F, Bateman GA, Spratt N, Loiselle A, Attia J et al (2009) Acute ischemic stroke: imaging-guided tenecteplase treatment in an extended time window. Neurology 72:915–921
Bivard A, Stanwell P, Levi CR, Parsons MW (2013) Arterial spin labeling identifies tissue salvage and good clinical recovery after acute ischemic stroke. J Neuroimaging 23:391–396
Parsons MW, Pepper EM, Bateman GA, Wang Y, Levi CR (2007) Identification of the penumbra and infarct core on hyperacute noncontrast and perfusion CT. Neurology 68:730–736
Bivard A, Levi C, Spratt N, Parsons M (2013) Perfusion CT in acute stroke: a comprehensive analysis of infarct and penumbra. Radiology 267:543–550
Bivard A, Stanwell P, Spratt N, Davis S, Krishnamurthy V, Levi C, Parsons M Defining acute ischemic stroke tissue pathophysiology with whole brain CT perfusion. Neurad 447
Bivard A, Levi C, Krishnamurthy V, Hislop-Jambrich J, Salazar P, Jackson B, Davis S, Parsons M (2014) Defining acute ischemic stroke tissue pathophysiology with whole brain CT perfusion. J Neuroradiol 41(5):307–315
Bivard A, Spratt N, Levi C, Parsons M (2011) Perfusion computer tomography: imaging and clinical validation in acute ischaemic stroke. Brain 134:3408–3416
Provencher W (1993) Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magn Reson Med 30:672–679
Moffett JR, Ross B, Arun P, Madhavarao CN, Namboodiri AM (2007) N-Acetylaspartate in the CNS: from neurodiagnostics to neurobiology. Prog Neurobiol 81(2):89–131
Hakim AM, Pokrupa RP, Villanueva J, Diksic M, Evans AC, Thompson CJ, Meyer E, Yamamoto YL, Feindel WH (1987) The effect of spontaneous reperfusion on metabolic function in early human cerebral infarcts. Ann Neurol 21(3):279–289
Davis SM, Lees KR, Albers GW, Diener HC, Markabi S, Karlsson G et al (2000) Selfotel in acute ischemic stroke: possible neurotoxic effects of an NMDA antagonist. Stroke 31:347–354
Pellerin L, Magistretti PJ (1994) Glutamate uptake into astrocytes stimulates aerobic glycolysis: a mechanism coupling neuronal activity to glucose utilization. Proc Natl Acad Sci U S A 91:10625–10629
Parsons MW, Li T, Barber PA, Yang Q, Darby DG, Desmond PM, Gerraty RP, Tress BM, Davis SM (2000) Combined (1)H MR spectroscopy and diffusion-weighted MRI improves the prediction of stroke outcome. Neurology 55(4):498–505
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
We declare that all human and animal studies have been approved by the Hunter New England Human Research Ethics Committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. We declare that all patients gave informed consent prior to inclusion in this study.
Conflict of interest
We declare that we have no conflict of interest.
Rights and permissions
About this article
Cite this article
Bivard, A., Yassi, N., Krishnamurthy, V. et al. A comprehensive analysis of metabolic changes in the salvaged penumbra. Neuroradiology 58, 409–415 (2016). https://doi.org/10.1007/s00234-015-1638-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-015-1638-x