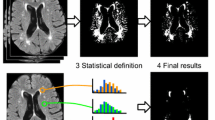
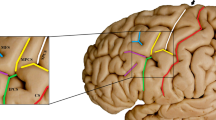
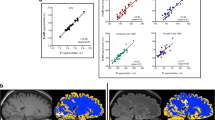
Abstract
Purpose
The central sulcus is an important landmark in the brain. This study aimed to investigate the distinctive signal of the paracentral lobule (PL) on T1-weighted images (T1WIs; the white PL sign) and evaluate its usefulness as a new method of identifying the central sulcus.
Methods
T1WIs of the brain of 96 participants (age, 58.9 ± 17.9 years; range, 8–87 years) scanned at 3-T MR system were retrospectively reviewed. First, we qualitatively analyzed the signal of the cortex of the PL by comparing it with that of the ipsilateral superior frontal gyrus on a 4-point grading score. Second, we compared the cortical signal intensity and gray/white-matter contrast between the PL and superior frontal gyrus. Third, we evaluated the usefulness of the PL signal for identifying the central sulcus.
Results
The PL cortex was either mildly hyperintense (grade 2) or definitely hyperintense (grade 3) in comparison with that of superior frontal cortex in all participants. The signal intensity of the PL cortex was significantly higher than that of the superior frontal cortex (p < 0.001), whereas the gray/white-matter contrast of the PL was weaker than that of the superior frontal gyrus (p < 0.001). The central sulci were identified with 94.3% accuracy (181/192) using the new method.
Conclusion
The white PL sign may be helpful in identifying the central sulcus, and this approach can be recognized as a new method for identification of the central sulcus.







Similar content being viewed by others
References
Naidich TP, Valavanis AG, Kubik S (1995) Anatomic relationships along the low-middle convexity: part I—normal specimens and magnetic resonance imaging. Neurosurgery 36:517–532
Naidich TP, Brightbill TC (1996) Systems for localizing fronto-parietal gyri and sulci on axial CT and MRI. Int J Neuroradiol 2:313–338
Ebeling U, Huber P, Reulen HJ (1986) Localization of the precentral gyrus in the computed tomogram and its clinical application. J Neurol 233:73–76
Meyer JR, Roychowdhury S, Russell EJ, Callahan C, Gitelman D, Mesulam MM (1996) Location of the central sulcus via cortical thickness of the precentral and postcentral gyri on MR. AJNR Am J Neuroradiol 17:1699–1706
Kido DK, LeMay M, Levinson AW, Benson WE (1980) Computed tomographic localization of the precentral gyrus. Radiology 135:373–377
Iwasaki S, Nakagawa H, Fukusumi A, Kichikawa K, Kitamura K, Otsuji H, Uchida H, Ohishi H, Yaguchi K, Sumie H et al (1991) Identification of pre- and postcentral gyri on CT and MR images on the basis of the medullary pattern of cerebral white matter. Radiology 179:207–213
Sobel DF, Gallen CC, Schwartz BJ, Waltz TA, Copeland B, Yamada S, Hirschkoff EC, Bloom FE (1993) Locating the central sulcus: comparison of MR anatomic and magnetoencephalographic functional methods. AJNR Am J Neuroradiol 14:915–925
Yousry TA, Schmid UD, Alkadhi H, Schmidt D, Peraud A, Buettner A, Winkler P (1997) Localization of the motor hand area to a knob on the precentral gyrus. A new landmark. Brain 120(Pt 1):141–157
Wagner M, Jurcoane A, Hattingen E (2013) The U sign: tenth landmark to the central region on brain surface reformatted MR imaging. AJNR Am J Neuroradiol 34:323–326
Kaneko OF, Fischbein NJ, Rosenberg J, Wintermark M, Zeineh MM (2017) The “white gray sign” identifies the central sulcus on 3T high-resolution T1-weighted images. AJNR Am J Neuroradiol 38:276–280
Su S, Yang N, Gaillard F (2019) Invisible cortex sign: a highly accurate feature to localize the inferolateral central sulcus. J Med Imaging Radiat Oncol 63:439–445
Mark IT, Luetmer PH, Rydberg CH, Witte RJ, Geske JR, Johnson MP, Lehman VT (2019) Signal intensity of peri-rolandic cortex identifies the central sulcus on double inversion recovery MRI. J Neurosurg Sci
Rodrigues T, Rodrigues M, Paz D, Costa MD, Santos B, Braga V, Paiva Neto M, Centeno R, Cavalheiro S, Chaddad-Neto F (2015) Is the omega sign a reliable landmark for the neurosurgical team? An anatomical study about the central sulcus region. Arq Neuropsiquiatr 73:934–938
Karaarslan E, Arslan A (2003) Perirolandic cortex of the normal brain: low signal intensity on turbo FLAIR MR images. Radiology 227:538–541
Kamada K, Kakeda S, Ohnari N, Moriya J, Sato T, Korogi Y (2008) Signal intensity of motor and sensory cortices on T2-weighted and FLAIR images: intraindividual comparison of 1.5T and 3T MRI. Eur Radiol 18:2949–2955
Kim EY, Kim DH, Chang JH, Yoo E, Lee JW, Park HJ (2009) Triple-layer appearance of Brodmann area 4 at thin-section double inversion-recovery MR imaging. Radiology 250:515–522
Kakeda S, Korogi Y, Kamada K, Ohnari N, Moriya J, Sato T, Kitajima M, Hasnine H, Hirata N (2008) Signal intensity of the motor cortex on phase-weighted imaging at 3T. AJNR Am J Neuroradiol 29:1171–1175
Dinçer A, Ozyurt O, Erzen C, Pamir MN (2010) Identification of the primary motor cortex: value of T2 echo-planar imaging, diffusion-weighted imaging and quantitative apparent diffusion coefficient measurement at 3 T. Eur Radiol 20:931–940
Spasojević G, Malobabic S, Pilipović-Spasojević O, Djukić-Macut N, Maliković A (2013) Morphology and digitally aided morphometry of the human paracentral lobule. Folia Morphol (Warsz) 72:10–16
Nagaratnam N, Davies D, Chen E (1998) Clinical effects of anterior cerebral artery infarction. J Stroke Cerebrovasc Dis 7:391–397
Fazekas F, Chawluk JB, Alavi A, Hurtig HI, Zimmerman RA (1987) MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol 149:351–356
Steen RG, Reddick WE, Ogg RJ (2000) More than meets the eye: significant regional heterogeneity in human cortical T1. Magn Reson Imaging 18:361–368
Salat DH, Lee SY, van der Kouwe AJ, Greve DN, Fischl B, Rosas HD (2009) Age-associated alterations in cortical gray and white matter signal intensity and gray to white matter contrast. Neuroimage 48:21–28
Stüber C, Morawski M, Schäfer A, Labadie C, Wähnert M, Leuze C, Streicher M, Barapatre N, Reimann K, Geyer S, Spemann D, Turner R (2014) Myelin and iron concentration in the human brain: a quantitative study of MRI contrast. Neuroimage 93(Pt 1):95–106
Nieuwenhuys R, Broere CA (2017) A map of the human neocortex showing the estimated overall myelin content of the individual architectonic areas based on the studies of Adolf Hopf. Brain Struct Funct 222:465–480
Hallgren B, Sourander P (1958) The effect of age on the non-haemin iron in the human brain. J Neurochem 3:41–51.27
Kakeda S, Yoneda T, Ide S, Watanabe K, Hiai Y, Korogi Y (2016) Signal intensity of superficial white matter on phase difference enhanced imaging as a landmark of the perirolandic cortex. Acta Radiol 57:1380–1386
Acknowledgements
We are grateful to the participants of this study.
Funding
Partial financial support was received from the Japan Society for the Promotion of Science KAKENHI grant no.19 K17150.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest/Competing interests
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study, formal consent is not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cho, S., Kurokawa, R., Hagiwara, A. et al. Localization of the central sulcus using the distinctive high signal intensity of the paracentral lobule on T1-weighted images. Neuroradiology 64, 289–299 (2022). https://doi.org/10.1007/s00234-021-02729-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-021-02729-4