Abstract
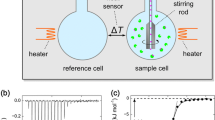
Equilibrium binding constants (Kb) between chemical compounds and target proteins or between interacting proteins provide a quantitative understanding of biological interaction mechanisms. Reported uncertainties of measured experimental parameters are critical for decision-making in many scientific areas, e.g., in lead compound discovery processes and in comparing computational predictions with experimental results. Uncertainties in measured Kb values are commonly represented by a symmetric normal distribution, often quoted in terms of the experimental value plus–minus the standard deviation. However, in general, the distributions of measured Kb (and equivalent Kd) values and the corresponding free energy change ΔGb are all asymmetric to varying degree. Here, using a simulation approach, we illustrate the effect of asymmetric Kb distributions within the realm of isothermal titration calorimetry (ITC) experiments. Further we illustrate the known, but perhaps not widely appreciated, fact that when distributions of any of Kb, Kd and ΔGb are transformed into each other, their degree of asymmetry is changed. Consequently, we recommend that a more accurate way of expressing the uncertainties of Kb, Kd, and ΔGb values is to consistently report 95% confidence intervals, in line with other authors’ suggestions. The ways to obtain such error ranges are discussed in detail and exemplified for a binding reaction obtained by ITC.



Similar content being viewed by others
References
Bates DM, Watts DG (2007) Nonlinear regression analysis and its applications. Wiley, Hoboken
Brautigam CA, Zhao H, Vargas C, Keller S, Schuck P (2016) Integration and global analysis of isothermal titration calorimetry data for studying macromolecular interactions. Nat Protoc 11:882–894
Broecker J, Vargas C, Keller S (2011) Revisiting the optimal cvalue for isothermal titration calorimetry. Anal Biochem 418:307–309
Buurma NJ, Haq I (2007) Advances in the analysis of isothermal titration calorimetry data for ligand–DNA interactions. Methods 42:162–172
Callies O, Daranas AH (2016) Application of isothermal titration calorimetry as a tool to study natural product interactions. Nat Prod Rep 33:881–904
Chaires JB (2008) Calorimetry and thermodynamics in drug design. Annu Rev Biophys 37:135–151
Cheng RKY et al (2017) Structural insight into allosteric modulation of protease-activated receptor 2. Nature 545:112–115
Cimmperman P, Matulis D (2011) Protein thermal denaturation measurements via a fluorescent dye. In: Podjarny A, Dejaegere AP, Kieffer B (eds) RSC biomolecular sciences. Royal Society of Chemistry, pp 247–274
Cimmperman P et al (2008) A quantitative model of thermal stabilization and destabilization of proteins by ligands. Biophys J 95:3222–3231
Ciulli A (2013) Biophysical screening for the discovery of small-molecule ligands. Methods Mol Biol Clifton NJ 1008:357–388
Dam TK, Torres M, Brewer CF, Casadevall A (2008) Isothermal titration calorimetry reveals differential binding thermodynamics of variable region-identical antibodies differing in constant region for a univalent ligand. J Biol Chem 283:31366–31370
Dullweber F, Stubbs MT, Musil Đ, Stürzebecher J, Klebe G (2001) Factorising ligand affinity: a combined thermodynamic and crystallographic study of trypsin and thrombin inhibition†. J Mol Biol 313:593–614
Falconer RJ (2016) Applications of isothermal titration calorimetry—the research and technical developments from 2011 to 2015: review of Isothermal Titration Calorimetry from 2011 to 2015. J Mol Recognit 29:504–515
Gaspari R et al (2016) Kinetic and structural insights into the mechanism of binding of sulfonamides to human carbonic anhydrase by computational and experimental studies. J Med Chem 59:4245–4256
Geschwindner S, Ulander J, Johansson P (2015) Ligand binding thermodynamics in drug discovery: still a hot tip? J Med Chem 58:6321–6335
Guan R, Tyler PC, Evans GB, Schramm VL (2013) Thermodynamic analysis of transition-state features in picomolar inhibitors of human 5′-methylthioadenosine phosphorylase. Biochemistry 52:8313–8322
Hörtner SR et al (2007) Potent inhibitors of tRNA-guanine transglycosylase, an enzyme linked to the pathogenicity of the Shigella Bacterium: charge-assisted hydrogen bonding. Angew Chem Int Ed 46:8266–8269
Huschmann FU et al (2016) Structures of endothiapepsin-fragment complexes from crystallographic fragment screening using a novel, diverse and affordable 96-compound fragment library. Acta Crystallogr Sect F Struct Biol Commun 72:346–355
Jarmoskaite I, AlSadhan I, Vaidyanathan PP, Herschlag D (2020) How to measure and evaluate binding affinities. Elife. https://doi.org/10.7554/eLife.57264
Johnson ML (1983) Evaluation and propagation of confidence intervals in nonlinear, asymmetrical variance spaces. Analysis of ligand-binding data. Biophys J 44:101–106
Kemmer G, Keller S (2010) Nonlinear least-squares data fitting in Excel spreadsheets. Nat Protoc 5:267–281
Krainer G, Keller S (2015) Single-experiment displacement assay for quantifying high-affinity binding by isothermal titration calorimetry. Methods 76:116–123
Krainer G, Broecker J, Vargas C, Fanghänel J, Keller S (2012) Quantifying high-affinity binding of hydrophobic ligands by isothermal titration calorimetry. Anal Chem 84:10715–10722
Krimmer SG, Klebe G (2015) Thermodynamics of protein–ligand interactions as a reference for computational analysis: how to assess accuracy, reliability and relevance of experimental data. J Comput Aided Mol Des 29:867–883
Krishnamurthy VM et al (2007) Thermodynamic parameters for the association of fluorinated benzenesulfonamides with bovine carbonic anhydrase II. Chem Asian J 2:94–105
Ladbury JE, Doyle ML (2004) Biocalorimetry 2: applications of calorimetry in the biological sciences. Wiley, Hoboken
Ladbury JE, Klebe G, Freire E (2010) Adding calorimetric data to decision making in lead discovery: a hot tip. Nat Rev Drug Discov 9:23–27
Lafont V et al (2007) Compensating enthalpic and entropic changes hinder binding affinity optimization. Chem Biol Drug Des 69:413–422
Leavitt S, Freire E (2001) Direct measurement of protein binding energetics by isothermal titration calorimetry. Curr Opin Struct Biol 11:560–566
McDonnell PA et al (2009) Assessing compound binding to the Eg5 motor domain using a thermal shift assay. Anal Biochem 392:59–69
Motulsky H, Christopoulos A (2004) Fitting models to biological data using linear and nonlinear regression: a practical guide to curve fitting. Oxford University Press, Oxford
Myszka DG, Rich RL (2000) Implementing surface plasmon resonance biosensors in drug discovery. Pharm Sci Technol Today 3:310–317
Olaru A, Bala C, Jaffrezic-Renault N, Aboul-Enein HY (2015) Surface plasmon resonance (SPR) biosensors in pharmaceutical analysis. Crit Rev Anal Chem 45:97–105
Pantoliano MW et al (2001) High-density miniaturized thermal shift assays as a general strategy for drug discovery. J Biomol Screen 6:429–440
Patching SG (2014) Surface plasmon resonance spectroscopy for characterisation of membrane protein–ligand interactions and its potential for drug discovery. Biochim Biophys Acta Biomembr 1838:43–55
Pierce MM, Raman CS, Nall BT (1999) Isothermal titration calorimetry of protein-protein interactions. Methods 19:213–221
Pulido NO et al (2015) On the molecular basis of the high affinity binding of basic amino acids to LAOBP, a periplasmic binding protein from Salmonella typhimurium: ENERGETIC BASIS OF LAOBP’S LIGAND RECOGNITION. J Mol Recognit 28:108–116
Rechlin C et al (2017) Price for opening the transient specificity pocket in human aldose reductase upon ligand binding: structural, thermodynamic, kinetic, and computational analysis. ACS Chem Biol. https://doi.org/10.1021/acschembio.7b00062
Ren J et al (2014) Thermodynamic and structural characterization of halogen bonding in protein-ligand interactions: a case study of PDE5 and its inhibitors. J Med Chem 57:3588–3593
Renaud J-P et al (2016) Biophysics in drug discovery: impact, challenges and opportunities. Nat Rev Drug Discov 15:679–698
Rühmann E, Betz M, Heine A, Klebe G (2015) Fragment binding can be either more enthalpy-driven or entropy-driven: crystal structures and residual hydration patterns suggest why. J Med Chem 58:6960–6971
Salim NN, Feig AL (2009) Isothermal titration calorimetry of RNA. Methods 47:198–205
Scheuermann TH, Brautigam CA (2015) High-precision, automated integration of multiple isothermal titration calorimetric thermograms: new features of NITPIC. Methods San Diego Calif 76:87–98
Schnapp G, Klein T, Hoevels Y, Bakker RA, Nar H (2016) Comparative analysis of binding kinetics and thermodynamics of dipeptidyl peptidase-4 inhibitors and their relationship to structure. J Med Chem 59:7466–7477
Smirnovienė J, Smirnovas V, Matulis D (2017) Picomolar inhibitors of carbonic anhydrase: importance of inhibition and binding assays. Anal Biochem 522:61–72
Tellinghuisen J (2003) A study of statistical error in isothermal titration calorimetry. Anal Biochem 321:79–88
Tellinghuisen J (2005) Statistical error in isothermal titration calorimetry: variance function estimation from generalized least squares. Anal Biochem 343:106–115
Tellinghuisen J (2017) Can you trust the parametric standard errors in nonlinear least squares? Yes, with provisos. Biochim Biophys Acta BBA Gen Subj. https://doi.org/10.1016/j.bbagen.2017.12.016
Tellinghuisen J (2018) Critique of methods for estimating heats in isothermal titration calorimetry. Anal Biochem 563:79–86
Typas A, Sourjik V (2015) Bacterial protein networks: properties and functions. Nat Rev Microbiol 13:559–572
Vega S, Abian O, Velazquez-Campoy A (2016) On the link between conformational changes, ligand binding and heat capacity. Biochim Biophys Acta Gen Sub 1860:868–878
Wells RD et al (1980) DNA structure and gene regulation. Prog Nucleic Acid Res Mol Biol 24:167–267
Wiseman T, Williston S, Brandts JF, Lin LN (1989) Rapid measurement of binding constants and heats of binding using a new titration calorimeter. Anal Biochem 179:131–137
Yanchunas J et al (2005) Molecular basis for increased susceptibility of isolates with atazanavir resistance-conferring substitution I50L to other protease inhibitors. Antimicrob Agents Chemother 49:3825–3832
Acknowledgements
This research was funded by grant no. S-LLT-20-2 from the Research Council of Lithuania (DM). MB and MJM aknowledge Fundação para a Ciência e Tecnologia (FCT), Portugal, for the financial support to Projects UIDB/00081/2020 and UIDB/00313/2020, respectively. The authors also acknowledge the COST action ARBRE-MOBIEU CA15126 supported by COST (European Cooperation in Science and Technology).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Special Issue: COST Action CA15126, MOBIEU: Between atom and cell.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paketurytė, V., Petrauskas, V., Zubrienė, A. et al. Uncertainty in protein–ligand binding constants: asymmetric confidence intervals versus standard errors. Eur Biophys J 50, 661–670 (2021). https://doi.org/10.1007/s00249-021-01518-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-021-01518-4