Abstract
The sensitivity of protein molecular structures makes them susceptible to aggregation in conditions unfavorable for the maintenance of their native folds. The aggregation of proteins leads to many disorders, but the inhibition of amyloid fibril formation using metal-containing small molecules is gaining popularity. Herein we report the effect of nickel(II) complexes (N1, N2, N3, and N4) bearing thiosemicarbazones on the inhibition of amyloid fibril formation by insulin. The interactions of the complexes with amyloid fibrils were investigated using various biophysical techniques, including light scattering, intrinsic fluorescence assay, thioflavin T (ThT) assay, and Fourier transform-infrared spectroscopy. The results revealed that the phenyl-substituted N3 was an efficient inhibitor of amyloid fibril formation and maintained the insulin in its native structure despite conditions promoting fibrillation.
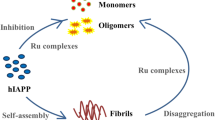
Graphic abstract

Nickel(II) complexes containing indole based thiosemicarbazones were efficient in inhibiting the amyloid fibril formation and maintaining the insulin in its native structure in unfavorable conditions.











Similar content being viewed by others
References
Alam P, Beg AZ, Siddiqi MK, Chaturvedi SK, Rajpoot RK, Ajmal MR, Zaman M, Abdelhameed AS, Khan RH (2017) Ascorbic acid inhibits human insulin aggregation and protects against amyloid induced cytotoxicity. Arch Biochem Biophys 621:54–62
Amini R, Yazdanparast R, Ghaffari SH (2015) Apigenin modulates the expression levels of pro-inflammatory mediators to reduce the human insulin amyloid-induced oxidant damages in SK-N-MC cells. Hum Exp Toxicol 34:642–653
Arora A, Ha C, Park CB (2004) Inhibition of insulin amyloid formation by small stress molecules. FEBS Lett 564:121–125
Balachandran C, Haribabu J, Jeyalakshmi K, Bhuvanesh NSP, Karvembu R, Emi N, Awale S (2018) Nickel(II) bis(isatin thiosemicarbazone) complexes induced apoptosis through mitochondrial signaling pathway and G0/G1 cell cycle arrest in IM-9 cells. J Inorg Biochem 182:208–221
Bekard IB, Dunstan DE (2009) Tyrosine autofluorescence as a measure of bovine insulin fibrillation. Biophys J 97:2521–2531
Das S, Smid SC (2019) Small molecule diketone flavorants diacetyl and 2, 3-pentanedione promote neurotoxicity but inhibit amyloid β aggregation. Toxicol Lett 300:67–72
Gomes LMF, Bataglioli CJ, Storr T (2020) Metal complexes that bind to the amyloid-β peptide of relevance to Alzheimer’s disease. Coord Chem Rev 412:213255
Gong G, Du W, Xu J, Huang X, Yin G (2018) Regulation of heteronuclear Pt–Ru complexes on the fibril formation and cytotoxicity of human islet amyloid polypeptide. J Inorg Biochem 189:7–16
Haribabu J, Jeyalakshmi K, Arun Y, Bhuvanesh NSP, Perumal PT, Karvembu R (2015) Synthesis, DNA/protein binding, molecular docking, DNA cleavage and in vitro anticancer activity of nickel(II) bis(thiosemicarbazone) complexes. RSC Adv 5:46031–46049
Haribabu J, Jeyalakshmi K, Arun Y, Bhuvanesh NSP, Perumal PT, Karvembu R (2017a) Synthesis of Ni(II) complexes bearing indole-based thiosemicarbazone ligands for interaction with biomolecules and some biological applications. J Biol Inorg Chem 22:461–480
Haribabu J, Ranade DS, Bhuvanesh NSP, Kulkarni PP, Karvembu R (2017b) Ru(II)-p-cymene thiosemicarbazone complexes as inhibitors of amyloid β (aβ) peptide aggregation and aβ-induced cytotoxicity. ChemistrySelect 2:11638–11644
Haribabu J, Srividya S, Mahendiran D, Gayathri D, Venkatramu V, Bhuvanesh N, Karvembu R (2020) Synthesis of palladium(II) complexes via Michael addition: Antiproliferative effects through ROS-mediated mitochondrial apoptosis and docking with SARS-CoV-2. Inorg Chem 59:17109–17122
Haribabu J, Alajrawy OI, Jeyalakshmi K, Balachandran C, Krishnan DA, Bhuvanesh N, Aoki S, Natarajan K, Karvembu R (2021) N-substitution in isatin thiosemicarbazones decides nuclearity of Cu(II) complexes–Spectroscopic, molecular docking and cytotoxic studies. Spectrochim Acta A Mol Biomol Spectrosc 246:118963
Iannuzzi C, Borriello M, Portaccio M, Irace G, Sirangelo I (2017) Insights into insulin fibril assembly at physiological and acidic pH and related amyloid intrinsic fluorescence. Int J Mol Sci 18:2551
Ivanova MI, Sievers SA, Sawaya MR, Wall JS, Eisenberg D (2009) Molecular basis for insulin fibril assembly. Proc Natl Acad Sci U S A 106:18990–18995
Jayamani J, Shanmugam G (2014) Gallic acid, one of the components in many plant tissues, is a potential inhibitor for insulin amyloid fibril formation. Eur J Med Chem 85:352–358
Kannan N, Balaji S, Kumar NVA (2017) Structural and elemental characterization of traditional Indian Siddha formulation: Thalagak karuppu. J Ayurveda Integr Med 8:184–189
Kathirvelan D, Haribabu J, Reddy BSR, Balachandran C (2015) Facile and diastereoselective synthesis of 3,2-spiropyrrolidine-oxindoles derivatives, their molecular docking and antiproliferative activities. Bioorg Med Chem Lett 25:389–399
Kaushik NK, Kaushik N, Attri P (2013) Biomedical importance of indoles. Molecules 18:6620–6662
Knechtel AH, Kumar S, Wallin C, Krol S, Warmlander SKTL, Jarvet J, Esposito G, Kirmizialtin S, Graslund A, Hamilton AD, Magzoub M (2020) Designed cell-penetrating peptide inhibitors of amyloid-beta aggregation and cytotoxicity. Cell Rep Phys Sci 1:100014
Kumar A, Nair AGC, Reddy AV, Garg AN (2006) Unique ayurvedic metallic-herbal preparations, chemical characterization. Biol Trace Elem Res 109:231–254
Li S, Shi B, Huang K, Wang Y (2020) Different intracellular signalling sensitivity and cell behaviour of porcine insulin with aging. Peptides 127:170278
Liu H, Qu Y, Wang X (2018) Amyloid β-targeted metal complexes for potential applications in Alzheimer’s disease. Future Med Chem 10:679–701
Luca GD, Galparsoro DF, Sancataldo G, Leone M, Fodera V, Vetri V (2020) Probing ensemble polymorphism and single aggregate structural heterogeneity in insulin amyloid self-assembly. J Colloid Interf Sci 574:229–240
Madav Y, Warikar S, Prabhakar B (2019) Recent therapeutic strategies targeting beta amyloid and tauopathies in Alzheimer’s disease. Brain Res Bull 146:171–184
Morel B, Lara FC (2019) Early mechanisms of amyloid fibril nucleation in model and disease-related proteins. BBA-Proteins Proteom 1867:140264
Nagase T, Iwaya K, Iwaki Y, Kotake F, Uchida R, Oh-I T, Sekine H, Miwa K, Murakami S, Od Kaka T, Kure M, Nemoto Y, Noritake M, Katsura Y (2014) Insulin-derived amyloidosis and poor glycemic control: A case series. Am J Med 127:450–454
Patela P, Parmara K, Das M (2018) Inhibition of insulin amyloid fibrillation by Morin hydrate. Int J Biol Macromol 108:225–239
Paterson BM, Cullinane C, Crouch P, White AP, Barnham KJ, Roselt PD, Noonan W, Binnis D, Hicks RJ, Donnelly PS (2019) Modification of biodistribution and brain uptake of copper bis(thiosemicarbazonato) complexes by the incorporation of amine and polyamine functional groups. Inorg Chem 58:4540–4552
Pettersen EF, Goddard TD, Huang CC, Couch CS, Greenblatt DM, Meng EC, Ferrin TE (2004) UCSF Chimera-a visualization system for exploratory research and analysis. J Comput Chem 25:1605–1612
Rajasekhar K, Samantha S, Bagoban V, Murugan AV, Govindaraju T (2020) Antioxidant berberine-derivative inhibits multifaceted amyloid toxicity. iScience 23:101005–101022
Ranade DS, Bapat AM, Ramteke SN, Joshi BN, Roussel P, Tomas A, Deschamps P, Kulkarani PP (2015) Thiosemicarbazone modification of 3-acetyl coumarin inhibits Aβ peptide aggregation and protect against Aβ-induced cytotoxicity. Eur J Med Chem 121:803–809
Ratha BN, Ghosh A, Brender JR, Gayen N, Ilyas H, Neeraja C, Das KP, Mandal AK, Bhunia A (2016) Inhibition of insulin amyloid fibrillation by a novel amphipathic heptapeptide. J Biol Chem 291:23545–23556
Saithong T, Thilavech T, Adisakwattana S (2018) Cyanidin-3-rutinoside reduces insulin fibrillation and attenuates insulin fibrils-induced oxidative hemolysis of human erythrocytes. Int J Biol Macromol 113:259–268
Singh AK, Bhattarcharya S, Halder K, Dasgupta S, Roy A (2020) Restriction of microwave-induced amyloid fibrillar growth by gold nanoparticles. Int J Biol Macromol 151:212–219
Swaminathan S (2016) Iron, hormesis, and protection in acute kidney injury. Kidney Int 90(1):16–17
Turkkan E, Sayin U, Erbilen N, Pehivanoglu S, Erdogan G, Tasdemir HU, Saf AO, Guler L, Akgemci EG (2017) Anticancer, antimicrobial, spectral, voltammetric and DFT studies with Cu(II) complexes of 2-hydroxy-5-methoxyacetophenone thiosemicarbazone and its N(4)-substituted derivatives. J Organomet Chem 831:23–35
Valensina D, Gabbiani C, Messoric L (2012) Metal compounds as inhibitors of β-amyloid aggregation. Perspectives for an innovative metallotherapeutics on Alzheimer’s disease. Coord Chem Rev 256:2357–2366
Wang H, Zhang J, Dou F, Chen Z (2020) A near-infrared fluorescent probe quinaldine red lights up the β-sheet structure of amyloid proteins in mouse brain. Biosens Bioelectron 153:112048
Zaman M, Khan AN, Wahiduzzaman ZSM, Khan RH (2019) Protein misfolding, aggregation and mechanism of amyloid cytotoxicity: an overview and therapeutic strategies to inhibit aggregation. Int J Biol Macromol 134:1022–1037
Zhang Y, Yu X, Zhu J, Liu S, Liu X, Dong Q, Chai J, Liu R (2018) Intravenous immunoglobulin improves glucose control and β-cell function in human IAPP transgenic mice by attenuating islet inflammation and reducing IAPP oligomers. Int Immunopharmcol 54:145–152
Acknowledgements
K.G. and S.S thanks Dr. MGR Educational and Research Institute for giving enough infrastructure to complete this work. J.H. thanks the Fondo Nacional de Ciencia y Tecnologia (FONDECYT) (3200391 and 11170840).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gomathi, K., Haribabu, J., Saranya, S. et al. Effective inhibition of insulin amyloid fibril aggregation by nickel(II) complexes containing heterocyclic thiosemicarbazones. Eur Biophys J 50, 1069–1081 (2021). https://doi.org/10.1007/s00249-021-01566-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-021-01566-w